Impact Factor ISSN: 1449-1907
Int J Med Sci 2021; 18(8):1753-1759. doi:10.7150/ijms.54954 This issue Cite
Research Paper
Carboxypeptidase A4 negatively correlates with p53 expression and regulates the stemness of breast cancer cells
1. Department of Breast Surgery, National Cancer Center/National Clinical Research Center for Cancer/Cancer Hospital, Chinese Academy of Medical Sciences and Peking Union Medical College, Beijing 100021, China.
2. State Key Laboratory of Molecular Oncology, National Cancer Center/National Clinical Research Center for Cancer/Cancer Hospital, Chinese Academy of Medical Sciences and Peking Union Medical College, Beijing, 100021, P.R. China.
3. Department of Pancreatic and Gastric Surgery, National Cancer Center/National Clinical Research Center for Cancer/Cancer Hospital, Chinese Academy of Medical Sciences and Peking Union Medical College, Beijing 100021, P.R. China.
4. Department of oncology, second medical centre of Chinese PLA General Hospital, Beijing, P.R. China.
5. Department of Pharmaceutical Sciences, University of Michigan, Ann Arbor, MI 48109.
6. Department of Anesthesiology, Beijing Obstetrics and Gynecology Hospital, Capital Medical University, Beijing, P.R. China.
7. Institute of Medical Information, Chinese Academy of Medical Sciences and Peking Union Medical College, Beijing 100020, P.R. China.
*Contributed equally to this work.
Abstract
Background: Triple-negative breast cancer (TNBC) is an aggressive cancer subtype lacking effective treatment options, and p53 is the most frequently mutated or deleted gene. Carboxypeptidase A4 (CPA4) is an extracellular metallocarboxypeptidase, which was closely associated with aggressiveness. Although a recent study indicated that CPA4 could induce epithelial‑mesenchymal transition in breast cancer cells, no studies investigated its stemness-related function and the correlation between CPA4 and p53 in TNBC. In this study, we aimed to investigate the CPA4 levels in breast cancer tissues and analyze its association with p53, and study its roles in cancer stemness maintenance.
Methods: CPA4 mRNA level and its prognostic value were analyzed by using online database UALCAN (
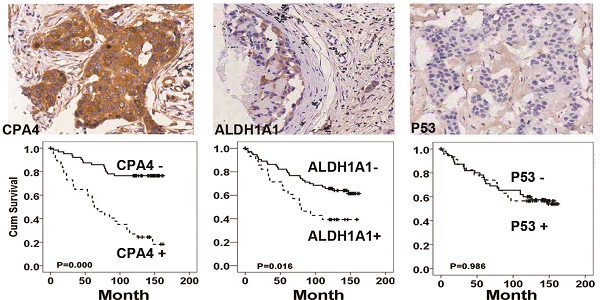
Results: Analysis of the UALCAN datasets identified that CPA4 mRNA levels were elevated in TNBC, especially in the TP53-mutant subgroup. Furthermore, high levels of CPA4 mRNA were significantly associated with unfavourable overall survival OS in breast cancer patients. Immunohistochemistical analysis demonstrated that CPA4 levels were elevated in 32.1% of breast cancer samples (45/140), and the positive rates of ALDH1A1 and p53 in the breast cancer tissues were 25% (35/140) and 50% (70/140), respectively. Statistical analysis revealed high levels of CPA4 was significantly associated with TNBC phenotype. Correlation analysis indicated that CPA4 over-expression was positively associated with ALDH1A1 (P<0.01) and negatively correlated with p53 (P<0.05). In Kaplan-Meier survival analysis, either high CPA4 or ALDH1A1 levels was significantly correlated with poor survival in breast cancer patients. Functional studies demonstrated that down-regulation of CPA4 significantly inhibited TNBC cell proliferation, colony-formation assays in soft agar and sphere formation in serum-free medium.
Conclusion: This study demonstrated for the first time that CPA4 was negatively correlates with p53 expression and inhibition of CPA4 could reduce the number of breast cancer cells with stemness property. It might be a potential target for the TNBC treatment.
Keywords: triple negative breast cancer, CPA4, P53, biomarker, stemness

 Global reach, higher impact
Global reach, higher impact