Impact Factor ISSN: 1449-1907
Int J Med Sci 2026; 23(6):2006-2015. doi:10.7150/ijms.127617 This issue Cite
Research Paper
Association Between Post-COVID-19 Herpes Zoster Reactivation and Peripheral Nervous System Disorders: Multinational Real-World Evidence from TriNetX
1. School of Medicine, College of Medicine, Fu Jen Catholic University, New Taipei City, Taiwan
2. Department of Neurology, Fu Jen Catholic University Hospital, Fu Jen Catholic University, New Taipei City, Taiwan
3. Division of Nephrology, Department of Internal Medicine, Fu Jen Catholic University Hospital, Fu Jen Catholic University, New Taipei City 24352, Taiwan.
4. Department of Research, Taipei Tzu Chi Hospital, Buddhist Tzu Chi Medical Foundation, New Taipei City, Taiwan.
5. School of Biomedical Sciences, Queensland University of Technology, Brisbane, Australia.
6. Division of Nephrology, Department of Internal Medicine, Cardinal-Tien Hospital, School of Medicine, College of Medicine, Fu Jen Catholic University, New Taipei City 24205, Taiwan.
7. Division of Nephrology, Department of Medicine, Taipei Tzu Chi Hospital, Buddhist Tzu Chi Medical Foundation, New Taipei City, Taiwan.
Received 2025-10-31; Accepted 2026-4-14; Published 2026-4-23
Abstract
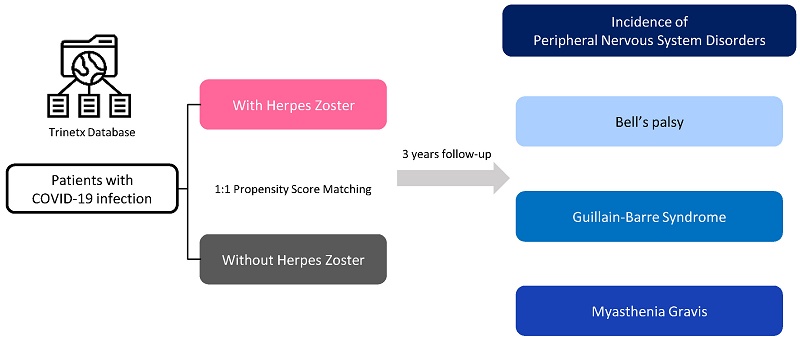
Background: The increased incidence of herpes zoster (HZ) following coronavirus disease 2019 (COVID-19) suggests a period of immune dysregulation, but the associated long-term neuro-immunological risks remain unclear.
Methods: Utilizing de-identified electronic health records from the TriNetX Global Collaborative Network, we established 1:1 propensity score-matched cohorts of COVID-19 survivors, comparing HZ-exposed and unexposed groups over a three-year follow-up. Primary endpoints included the incidence of peripheral nervous system disorders: Bell's palsy, Guillain-Barré syndrome (GBS), and myasthenia gravis (MG). Analyses included Cox proportional hazards models, landmark analyses, and multiple sensitivity analyses.
Results: HZ reactivation was associated with increased three-year risks of all outcomes. The risk of Bell's palsy was elevated early and remained sustained (hazard ratio 3.625, 95% confidence interval 3.151-4.170). In contrast, the risks of GBS (hazard ratio 1.858, 95% confidence interval 1.243-2.779) and MG (hazard ratio 1.640, 95% confidence interval 1.178-2.284) showed delayed increases emerging after the first year. These associations remained consistent across sensitivity analyses and were more pronounced in individuals with metabolic comorbidities. COVID-19 vaccination was not associated with an increased risk of these outcomes, although subgroup findings should be interpreted with caution due to limited event counts.
Conclusions: Post-COVID-19 HZ is associated with an increased risk of peripheral nervous system disorders, highlighting the need for symptom-based neurological awareness during both early and delayed post-infectious periods.
Keywords: autoimmune diseases, Bell's palsy, COVID-19, Guillain-Barré syndrome, herpes zoster, myasthenia gravis, peripheral nervous system, SARS-CoV-2
Introduction
Coronavirus disease 2019 (COVID-19) profoundly impacts the peripheral nervous system (PNS) [1]. A prominent complication is increased herpes zoster (HZ) reactivation [2]. HZ represents clinical reactivation of latent varicella-zoster virus (VZV) within sensory ganglia, producing characteristic vesicular rash and neuronal damage [3].
Observational studies confirm substantially elevated HZ risk in COVID-19 survivors versus uninfected controls (reported hazard ratios [HRs]: 1.59-2.16) [4, 5]. SARS-CoV-2 likely induces immunological perturbations—including lymphopenia and impaired VZV-specific cell-mediated immunity [2, 6, 7] —while Th17/IL-17 pathway activation may further predispose to VZV reactivation [8].
HZ's neurotropism frequently causes direct PNS pathology, including cranial nerve involvement [2, 3]. Ramsay Hunt syndrome (Bell's palsy variant) exemplifies VZV-mediated facial paralysis [9, 10]. Post-COVID HZ may thus compound SARS-CoV-2's direct neuroinflammatory effects [11, 12].
COVID-19 independently elevates risks of Guillain-Barré syndrome (GBS; post-infectious demyelination) [13, 14], myasthenia gravis (MG; molecular mimicry) [15, 16] and Bell's palsy [17]. Although literature links COVID-19 to HZ, GBS, and MG [16-18], HZ's mediating role remains unexplored [16, 18, 19].
Given HZ's immune dysregulation and neural inflammation, we hypothesize that post-COVID HZ reactivation exacerbates latent immune dysfunction, increasing subsequent autoimmune/inflammatory PNS disorder risk. This propensity score-matched (PSM) cohort study therefore evaluates Bell's palsy, GBS, and MG incidence in COVID-19 survivors with versus without subsequent HZ over a three-year follow-up.
Materials and Methods
Study design and data source
We conducted a retrospective cohort study using the TriNetX federated global research network (Global Collaborative Network), which provides access to de-identified electronic health records (EHRs) from 144 international healthcare organizations. The database encompasses structured patient dossiers including demographics, clinical diagnoses (International Classification of Diseases, Tenth Revision, Clinical Modification [ICD-10-CM]), procedures, medications, and laboratory results. This study followed the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) reporting guidelines and the tenets of the Declaration of Helsinki. Institutional Review Board (IRB) approval was obtained from Taipei Tzu Chi Hospital (No: 14-IRB140), with a waiver of informed consent due to the use of de-identified retrospective data.
Study population
Adult patients (aged ≥ 18 years) with a confirmed COVID-19 diagnosis between January 1, 2020, and December 31, 2022, were identified. COVID-19 was defined by a positive SARS-CoV-2 RNA laboratory result (TriNetX code: 9088) or ICD-10-CM code U07.1. The index date was defined as the date of the first recorded COVID-19 diagnosis or positive laboratory test. From the initial pool of COVID-19 survivors, we identified an exposed group (COVID-19 + HZ; n = 110,054) comprising patients with a new diagnosis of HZ (ICD-10-CM B02 and subtypes) occurring between day 1 and 3 years following the index date, and an unexposed group (COVID-19 only; n = 14,849,703) comprising patients without HZ during the follow-up. Patients with a history of HZ within one year prior to or on the index date were excluded.
Outcome measures
The primary outcomes were the 3-year incidence of three PNS disorders: Bell's palsy (ICD-10 G51.0), GBS (ICD-10 G61.0), and MG (ICD-10 G70.0, G70.00, G70.01). To ensure all outcomes were incident events, patients with a documented history of the specific outcome prior to the index date were excluded from the respective analysis. Follow-up commenced one day after the index date and continued for up to 1,095 days, terminating at the occurrence of the outcome, death, or loss to follow-up.
Propensity score matching
To address potential confounding and the computational constraints of the large-scale dataset, a two-stage 1:1 PSM strategy was employed using a greedy nearest-neighbor algorithm. The primary analysis matched the overall cohort on essential covariates, including age, sex, White race, diabetes mellitus, and hypertension. A complementary validation was conducted on the age-restricted cohort (50-59 years) to allow for a broader adjustment of clinical and laboratory parameters. Covariate balance was defined by a standardized mean difference (SMD) < 0.1.
Statistical analysis
Following PSM, the analysis involved two distinct approaches. First, absolute risks, relative risks (RRs), and odds ratios (ORs) were calculated for the PNS outcomes (Supplementary Table S2). Second, a time-to-event analysis was performed using Kaplan-Meier survival curves (log-rank test) and Cox proportional hazards models to estimate HRs and 95% confidence intervals (CIs). The proportionality assumption of the Cox model was formally evaluated. Statistical significance was predetermined at a two-sided p-value < 0.05.
Sensitivity analysis
To ensure the reliability of the findings, several sensitivity analyses were conducted. These included comparisons between the age-restricted cohort (50-59 years) and the overall cohort to assess generalizability. Temporal stability and sequencing were evaluated using landmark analyses and time-window analyses, with additional exclusion of individuals whose outcomes occurred prior to or concurrent with HZ onset. Clinical sub-analyses were performed by excluding individuals with immunosuppression, human immunodeficiency virus (HIV) infection (ICD-10: B20), or influenza (ICD-10: J09-J11), and by evaluating the influence of vaccination. To address potential detection bias, diagnostic frequency, nerve conduction velocity (NCV) utilization, and hospitalization burden were assessed. Finally, additional analyses examined the impact of anti-HZ therapy and calculated E-values to evaluate the influence of unmeasured confounding.
Results
Cohort characteristics and baseline balance
The baseline characteristics of the age-restricted cohort (50-59 years) before and after PSM are presented in Table 1. Prior to matching, the COVID+HZ group exhibited higher proportions of females (64.4% vs. 53.1%), White race (68.5% vs. 57.8%), hypertension (27.2% vs. 16.0%), and diabetes mellitus (14.3% vs. 8.0%). After 1:1 PSM, all covariates achieved balance with standardized mean differences (SMD) < 0.10. Similar baseline patterns and post-match balance were observed in the overall cohort (18-89 years) (Supplementary Table S1).
Baseline characteristics of COVID-19 patients aged 50-59 years with vs without subsequent herpes zoster reactivation, before and after propensity score matching
| Characteristic | Before Matching | After Matching | ||||||
|---|---|---|---|---|---|---|---|---|
| Mean±SD | Patient Count | % of Cohort | Std. Dif. | Mean±SD | Patient Count | % of Cohort | Std. Dif. | |
| Demographics | ||||||||
| Age at Index | 54.56±2.98 vs 54.45±2.94 | 16,419 vs 2,251,793 | 100.00% vs 100.00% | 0.0365 | 54.56±2.98 vs 54.57±2.97 | 16,418 vs 16,418 | 100.00% vs 100.00% | 0.0043 |
| Female | 10,577 vs 1,195,574 | 64.42% vs 53.09% | 0.2316 | 10,576 vs 10,518 | 64.42% vs 64.06% | 0.0074 | ||
| Male | 5,452 vs 1,020,437 | 33.20% vs 45.32% | 0.2499 | 5,452 vs 5,498 | 33.21% vs 33.49% | 0.0059 | ||
| White | 11,245 vs 1,302,467 | 68.49% vs 57.84% | 0.2221 | 11,245 vs 11,346 | 68.49% vs 69.11% | 0.0133 | ||
| Black or African American | 1,981 vs 328,104 | 12.06% vs 14.57% | 0.0738 | 1,981 vs 2,000 | 12.07% vs 12.18% | 0.0035 | ||
| Unknown Race | 1,559 vs 400,073 | 9.49% vs 17.77% | 0.2428 | 1,559 vs 1,521 | 9.50% vs 9.26% | 0.0079 | ||
| Asian | 775 vs 90,773 | 4.72% vs 4.03% | 0.0337 | 774 vs 720 | 4.71% vs 4.38% | 0.0158 | ||
| Diagnoses | ||||||||
| Hypertensive diseases | 4,463 vs 360,007 | 27.18% vs 15.99% | 0.2747 | 4,462 vs 4,542 | 27.18% vs 27.66% | 0.0109 | ||
| Diabetes mellitus | 2,356 vs 179,437 | 14.35% vs 7.97% | 0.2037 | 2,355 vs 2,364 | 14.34% vs 14.40% | 0.0016 | ||
| Ischemic heart diseases | 819 vs 70,241 | 4.99% vs 3.12% | 0.0949 | 819 vs 779 | 4.99% vs 4.75% | 0.0113 | ||
| Cerebrovascular diseases | 301 vs 30,403 | 1.83% vs 1.35% | 0.0386 | 301 vs 321 | 1.83% vs 1.96% | 0.0089 | ||
| Laboratory data | ||||||||
| Creatinine | 1.04±1.52 vs 1.06±2.39 | 7,153 vs 610,113 | 43.56% vs 27.09% | 0.0106 | 1.04±1.52 vs 1.03±2.03 | 7,152 vs 7,230 | 43.56% vs 44.04% | 0.0055 |
| Glucose | 118.49±56.72 vs 117.88±54.70 | 7,008 vs 606,536 | 42.68% vs 26.94% | 0.0109 | 118.49±56.72 vs 118.94±56.56 | 7,008 vs 7,098 | 42.69% vs 43.23% | 0.0079 |
| Calcium | 9.36±0.56 vs 9.34±0.57 | 6,905 vs 575,087 | 42.05% vs 25.54% | 0.0395 | 9.36±0.56 vs 9.36±0.56 | 6,904 vs 6,963 | 42.05% vs 42.41% | 0.0032 |
| Urea nitrogen | 16.72±10.11 vs 16.37±9.56 | 6,776 vs 554,166 | 41.27% vs 24.61% | 0.0362 | 16.72±10.11 vs 16.26±9.57 | 6,775 vs 6,839 | 41.27% vs 41.66% | 0.0468 |
| Hematocrit | 40.08±6.84 vs 39.95±8.14 | 6,449 vs 556,603 | 39.28% vs 24.72% | 0.0173 | 40.08±6.84 vs 40.13±7.03 | 6,448 vs 6,492 | 39.27% vs 39.54% | 0.0083 |
| Hemoglobin | 13.35±1.93 vs 13.51±1.99 | 6,427 vs 552,799 | 39.14% vs 24.55% | 0.0814 | 13.35±1.93 vs 13.41±1.95 | 6,426 vs 6,460 | 39.14% vs 39.35% | 0.0309 |
| Alanine aminotransferase | 30.57±181.75 vs 30.55±59.50 | 6,164 vs 506,798 | 37.54% vs 22.51% | 0.0002 | 30.58±181.77 vs 31.00±101.08 | 6,163 vs 6,147 | 37.54% vs 37.44% | 0.0029 |
| Aspartate aminotransferase | 27.21±44.32 vs 30.09±86.37 | 6,054 vs 492,589 | 36.87% vs 21.88% | 0.0421 | 27.21±44.33 vs 30.27±79.43 | 6,053 vs 6,052 | 36.87% vs 36.86% | 0.0476 |
| Albumin | 4.14±0.47 vs 4.12±0.53 | 5,942 vs 481,307 | 36.19% vs 21.37% | 0.0358 | 4.14±0.47 vs 4.13±0.51 | 5,941 vs 5,984 | 36.19% vs 36.45% | 0.0189 |
| Alkaline phosphatase | 89.05±47.77 vs 89.57±62.66 | 5,862 vs 477,000 | 35.70% vs 21.18% | 0.0093 | 89.05±47.77 vs 91.05±62.52 | 5,862 vs 5,881 | 35.70% vs 35.82% | 0.0360 |
| Bilirubin, total | 0.56±0.88 vs 0.63±1.08 | 5,768 vs 468,463 | 35.13% vs 20.80% | 0.0685 | 0.56±0.88 vs 0.60±0.78 | 5,767 vs 5,799 | 35.13% vs 35.32% | 0.0407 |
| Leukocytes | 53.61±432.80 vs 26.08±278.58 | 5,718 vs 488,168 | 34.83% vs 21.68% | 0.0756 | 52.84±428.90 vs 25.09±267.03 | 5,717 vs 5,746 | 34.82% vs 35.00% | 0.0777 |
| Protein | 7.08±0.67 vs 7.12±0.77 | 5,526 vs 455,044 | 33.66% vs 20.21% | 0.0593 | 7.08±0.67 vs 7.12±0.65 | 5,526 vs 5,571 | 33.66% vs 33.93% | 0.0580 |
| Platelet mean | 9.73±1.33 vs 9.77±1.46 | 4,074 vs 370,944 | 24.81% vs 16.47% | 0.0292 | 9.73±1.33 vs 9.77±1.33 | 4,073 vs 4,051 | 24.81% vs 24.67% | 0.0271 |
| Cholesterol | 191.68±45.71 vs 189.25±45.49 | 3,830 vs 286,158 | 23.33% vs 12.71% | 0.0532 | 191.68±45.71 vs 188.68±45.18 | 3,829 vs 3,838 | 23.32% vs 23.38% | 0.0662 |
| Cholesterol in HDL | 50.39±20.71 vs 49.37±19.92 | 3,826 vs 291,274 | 23.30% vs 12.94% | 0.0499 | 50.39±20.71 vs 50.26±20.13 | 3,826 vs 3,847 | 23.30% vs 23.43% | 0.0060 |
| Cholesterol in LDL | 110.41±38.26 vs 109.73±38.03 | 3,799 vs 287,023 | 23.14% vs 12.75% | 0.0179 | 110.42±38.26 vs 108.51±37.87 | 3,798 vs 3,818 | 23.13% vs 23.25% | 0.0500 |
| Triglyceride | 147.07±114.44 vs 147.15±132.31 | 3,779 vs 291,274 | 23.02% vs 12.94% | 0.0006 | 147.07±114.44 vs 147.55±143.95 | 3,779 vs 3,824 | 23.02% vs 23.29% | 0.0037 |
| Hemoglobin A1c | 6.74±1.92 vs 6.62±1.81 | 3,087 vs 243,192 | 18.80% vs 10.80% | 0.0667 | 6.74±1.92 vs 6.70±1.83 | 3,086 vs 3,015 | 18.80% vs 18.36% | 0.0237 |
| Magnesium | 1.93±0.31 vs 1.94±0.31 | 1,198 vs 96,288 | 7.30% vs 4.28% | 0.0301 | 1.93±0.31 vs 1.93±0.31 | 1,198 vs 1,140 | 7.30% vs 6.94% | 0.0070 |
| Phosphate | 3.63±1.08 vs 3.60±1.10 | 968 vs 70,013 | 5.90% vs 3.11% | 0.0291 | 3.63±1.08 vs 3.63±1.10 | 967 vs 883 | 5.89% vs 5.38% | 0.0006 |
| C-reactive protein | 17.84±42.43 vs 25.35±52.21 | 940 vs 73,185 | 5.72% vs 3.25% | 0.1578 | 17.86±42.45 vs 21.74±49.91 | 939 vs 905 | 5.72% vs 5.51% | 0.0837 |
| 0-10 mg/L | 703 vs 48,303 | 4.3% vs 2.1% | 0.121 | 702 vs 654 | 4.3% vs 4.0% | 0.015 | ||
| 10-20 mg/L | 158 vs 12,871 | 1.0% vs 0.6% | 0.045 | 158 vs 155 | 1.0% vs 0.9% | 0.002 | ||
| 20-30 mg/L | 66 vs 6,561 | 0.4% vs 0.3% | 0.019 | 66 vs 61 | 0.4% vs 0.4% | 0.005 | ||
| 30-60 mg/L | 87 vs 9,163 | 0.5% vs 0.4% | 0.018 | 87 vs 81 | 0.5% vs 0.5% | 0.005 | ||
| Calcidiol | 34.66±16.88 vs 33.97±17.20 | 793 vs 50,493 | 4.83% vs 2.24% | 0.0403 | 34.66±16.88 vs 35.75±18.52 | 793 vs 724 | 4.83% vs 4.41% | 0.0616 |
| Urate | 5.82±1.93 vs 5.82±2.02 | 553 vs 36,237 | 3.37% vs 1.61% | 0.0002 | 5.82±1.93 vs 5.84±1.97 | 552 vs 495 | 3.36% vs 3.02% | 0.0082 |
| Medications | ||||||||
| Antilipemic agents | 2,521 vs 214,098 | 15.35% vs 9.51% | 0.1779 | 2,521 vs 2,601 | 15.36% vs 15.84% | 0.0134 | ||
| Antiarrhythmics | 1,916 vs 155,165 | 11.67% vs 6.89% | 0.1653 | 1,916 vs 1,650 | 11.67% vs 10.05% | 0.0521 | ||
| Diuretics | 1,843 vs 153,195 | 11.22% vs 6.80% | 0.1549 | 1,843 vs 1,788 | 11.22% vs 10.89% | 0.0107 | ||
| Beta blockers | 1,669 vs 144,071 | 10.16% vs 6.40% | 0.1370 | 1,669 vs 1,642 | 10.17% vs 10.00% | 0.0055 | ||
| Calcium channel blockers | 1,310 vs 112,927 | 7.98% vs 5.01% | 0.1205 | 1,310 vs 1,320 | 7.98% vs 8.04% | 0.0022 | ||
| ACE inhibitors | 1,230 vs 113,727 | 7.49% vs 5.05% | 0.1008 | 1,230 vs 1,254 | 7.49% vs 7.64% | 0.0055 | ||
| Angiotensin II inhibitor | 1,069 vs 88,724 | 6.51% vs 3.94% | 0.1157 | 1,069 vs 1,114 | 6.51% vs 6.79% | 0.0110 | ||
Abbreviations: COVID-19, coronavirus disease 2019; ACE, angiotensin-converting enzyme; SD, standard deviation; St. Diff, standardized difference; values < 0.1 typically indicate good balance.
Bell's palsy
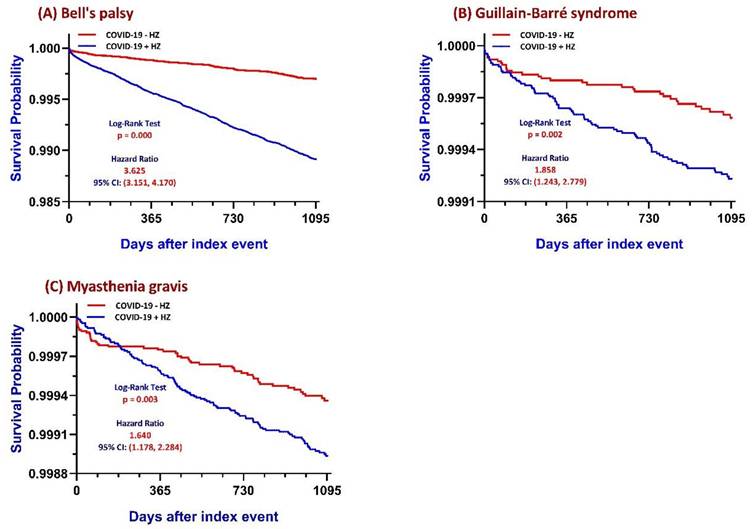
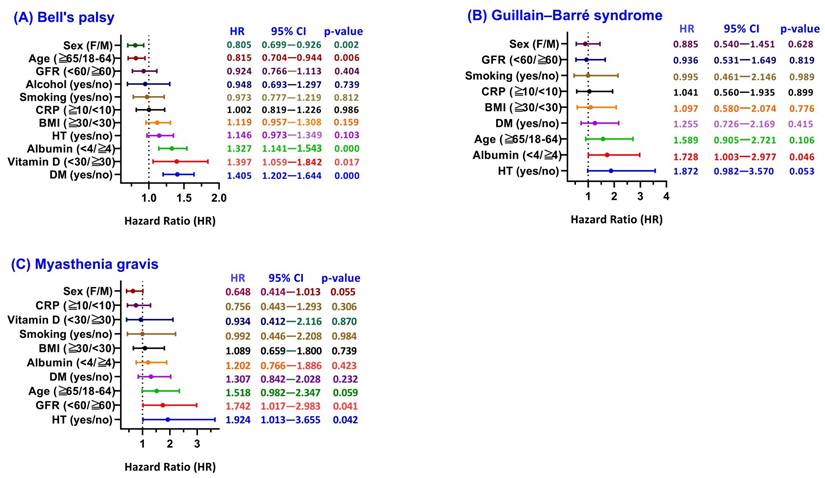
Among COVID-19 survivors, the COVID+HZ group (n = 109,010) demonstrated a higher 3-year cumulative incidence of Bell's palsy compared to the COVID-HZ group (1.00% vs. 0.22%; absolute risk difference 0.008, 95% CI 0.007-0.009; p < 0.001). Time-to-event analysis yielded a Cox HR of 3.625 (95% CI 3.151-4.170; log-rank p < 0.001) (Figure 2A; Supplementary Table S2). Subgroup analysis (Figure 3A) revealed that metabolic vulnerabilities were associated with further elevated HRs, including diabetes (HR 1.405, 95% CI 1.202-1.644; p < 0.001), vitamin D deficiency (HR 1.397, 95% CI 1.059-1.842; p = 0.017), and hypoalbuminemia (HR 1.327, 95% CI 1.141-1.543; p < 0.001). Conversely, lower HRs were observed in older adults (≥ 65 years: HR 0.815, 95% CI 0.704-0.944; p = 0.006) and females (HR 0.805, 95% CI 0.699-0.926; p = 0.002).
Flowchart of patient selection and cohort construction. This diagram illustrates the identification of adult COVID-19 survivors and their subsequent categorization into Herpes Zoster (HZ)-exposed and HZ-unexposed cohorts over a 3-year follow-up period. Exclusion criteria and final cohort sizes (n) after propensity score matching are indicated within the respective steps.
Kaplan-Meier survival curves comparing COVID-19 patients with and without Herpes Zoster (HZ) reactivation. Cumulative incidence of freedom from (A) Bell's palsy, (B) Guillain-Barré syndrome, and (C) myasthenia gravis over a 3-year follow-up period is shown for the HZ-exposed and matched control cohorts. P values were calculated using the log-rank test. Abbreviations: HZ, herpes zoster; COVID-19, coronavirus disease 2019; COVID+ HZ, COVID-19 survivors with HZ reactivation; COVID- HZ, COVID-19 survivors without HZ reactivation.
Subgroup analysis of Peripheral Nervous System outcomes: hazard ratios associated with Herpes Zoster (HZ). Forest plots showing hazard ratios (HRs) for (A) Bell's palsy, (B) Guillain-Barré syndrome, and (C) Myasthenia gravis, comparing HZ-exposed versus HZ-unexposed COVID-19 survivors stratified by baseline factors. Data are presented as adjusted HRs with 95% confidence intervals (CIs). Abbreviations: HZ, herpes zoster; COVID-19, coronavirus disease 2019; GFR, glomerular filtration rate; CRP, C-reactive protein; BMI, body mass index; HT, hypertension; DM, diabetes mellitus; HR, hazard ratio; CI, confidence interval.
Guillain-Barré syndrome and myasthenia gravis
The 3-year GBS incidence was 0.07% (79/109,918) in the COVID+HZ group versus 0.03% (34/109,975) in the COVID-HZ group (RR 2.33, 95% CI 1.56-3.48; Supplementary Table S2). Kaplan-Meier curves showed early separation (log-rank χ² = 9.406, p = 0.002; Figure 2B), with a Cox HR of 1.858 (95% CI 1.243-2.779). For MG, subsequent HZ was associated with a higher 3-year incidence (0.10% vs. 0.05%; risk difference 0.001, 95% CI 0.000-0.001; p < 0.001), with a Cox HR of 1.640 (95% CI 1.178-2.284; Figure 2C). Subgroup analyses (Figure 3B, 3C) identified higher risks associated with hypertension (GBS HR 1.872; MG HR 1.924) and reduced kidney function (MG HR 1.742, 95% CI 1.017-2.983).
Comprehensive sensitivity and robustness analyses
Landmark analysis (Supplementary Table S3) showed distinct temporal patterns, with an immediate elevation in Bell's palsy risk (HR approximately 3.6-3.8 across time points), whereas associations for GBS and MG were not significant at 1 year but became significant during longer follow-up periods. Time-window-based analysis showed the HR for Bell's palsy peaked within the first 6 months (HR 5.02, 95% CI 3.629-6.942) (Supplementary Table S9). To ensure temporal sequence, a sensitivity analysis excluding individuals whose PNS outcomes occurred prior to or concurrent with their first HZ episode was performed (Supplementary Table S10); the risk for Bell's palsy remained significant (HR 1.428, 95% CI 1.203-1.696), while associations for GBS and MG were attenuated. E-value analysis yielded values of 6.72 for Bell's palsy, 3.12 for GBS, and 2.66 for MG (Supplementary Table S14).
The exclusion of pre-index immunosuppression or radiotherapy resulted in HRs of 3.74 for Bell's palsy and 2.51 for GBS (Supplementary Table S4). After excluding individuals with HIV or influenza, HRs remained significant: 4.17 (95% CI 3.52-4.95) for Bell's palsy, 2.32 (95% CI 1.38-3.91) for GBS, and 1.53 (95% CI 1.04-2.26) for MG (Supplementary Table S6). Vaccination-stratified analyses (Supplementary Tables S5A and S5B) showed that Bell's palsy risk persisted in both vaccinated (HR 3.685, 95% CI 2.674-5.077) and unvaccinated (HR 4.138, 95% CI 3.514-4.873) cohorts. Additional sensitivity analysis for HZ vaccination status demonstrated consistent risk for Bell's palsy (Supplementary Table S13).
Detection bias and treatment impact
Diagnostic frequency analysis: the average number of Bell's palsy-related diagnoses per patient was 1.291 ± 1.34 in the COVID+HZ group versus 1.521 ± 1.62 in the COVID-HZ group, with diagnostic ratios consistently exceeding 1.0 across all studied outcomes (Supplementary Table S7). Regarding the neurologic testing workout, the COVID+HZ cohort demonstrated significantly higher NCV utilization (mean 0.054 ± 0.45 vs. 0.022 ± 0.28; p < 0.001; Supplementary Table S8). Furthermore, the HZ-exposed group exhibited a higher 3-year hospitalization burden (mean 2.532 vs. 1.446, p < 0.0001; Supplementary Table S11). Finally, the impact of anti-HZ therapy on the 3-year risk of Bell's palsy among patients with concurrent infections showed no statistically significant reduction (HR 1.378, 95% CI 0.773-2.457; p = 0.275; Supplementary Table S12).
Discussion
Principal findings
This large-scale, longitudinal study provides evidence that HZ reactivation following COVID-19 is associated with a clinically meaningful increase in risk of subsequent neurological outcomes. Our 3-year analysis demonstrates a substantial relative risk increase across all primary outcomes: Bell's palsy showed the strongest association (HR 3.625), followed by GBS (HR 1.858) and MG (HR 1.640). While the absolute risks observed are relatively low (ranging from 0.07% to 1.00%), the observed associations may still be clinically relevant, particularly given the severity of these conditions, including the life-threatening nature of GBS [20] and the chronic management required for MG [21], as well as the large population of COVID-19 survivors.
Notably, the analysis revealed a distinct temporal divergence in risk profiles. Bell's palsy demonstrated an immediate and sustained risk elevation starting within the first six months, whereas the hazards for GBS and MG exhibited a delayed trajectory, becoming statistically prominent primarily after the first year. This temporal shift suggests a transition from acute viral-mediated injury [5] to a more chronic, secondary immune dysregulation [20, 22].
Immediate vs. latent pathophysiological mechanisms
The distinct temporal patterns observed in our analysis provide important clues to the underlying mechanisms. The immediate increase in risk of Bell's palsy, occurring within the early post-HZ period, supports a mechanism related to direct viral neurotropism rather than a delayed immune-mediated process [11, 23]. VZV reactivation within the geniculate ganglion may trigger acute neuritis and inflammatory edema [24, 25], which, within the anatomical confines of the fallopian canal [26], can result in facial nerve compression and ischemia [27, 28]. Subgroup analyses further suggest that metabolic vulnerabilities, such as diabetes [10, 29] and vitamin D deficiency [30], may amplify this risk by impairing host immune responses and facilitating localized viral reactivation.
In contrast, the delayed emergence of GBS and MG, typically occurring beyond the first year, is more consistent with secondary immune-mediated mechanisms rather than direct viral invasion. Following HZ reactivation, a latency period may be required for the development of autoreactive immune responses [31, 32], such as molecular mimicry and epitope spreading [33-35]. In GBS, this may involve immune-mediated injury to peripheral myelin [20], whereas in MG, disruption of neuromuscular transmission may occur through autoantibody-mediated mechanisms [22].
This temporal-mechanistic linkage is further supported by prior TriNetX analyses (e.g., Lu et al., Chien et al.), which demonstrated that HZ reactivation following COVID-19 serves as a systemic marker of post-viral immunodysregulation and is associated with an increased risk of subsequent multi-system complications, including cardiorenal and autoimmune sequelae [36, 37]. Prior studies using artificial intelligence (AI)-based models have also highlighted the role of systemic inflammation in determining COVID-19 severity, which may contribute to subsequent immune dysregulation [38]. The magnitude of systemic inflammation during acute COVID-19 may further influence this process by lowering the threshold for both VZV reactivation [6, 8] and subsequent neurological complications [39-41].
Together, these findings support a unified model in which HZ reactivation reflects a state of persistent immune dysregulation, predisposing individuals to both early neurotropic effects and delayed autoimmune-mediated complications.
Clinical significance and stratified surveillance strategy
To translate these findings into clinical practice, we propose a stratified surveillance strategy tailored to high-risk populations, specifically those with advanced age or metabolic comorbidities. Given the temporal divergence observed, monitoring should be biphasic. During the acute phase (0-6 months), clinicians should prioritize cranial nerve assessments to ensure the timely administration of corticosteroids or antivirals for Bell's palsy, which may prevent irreversible axonal loss. For the latent phase (6-36 months), vigilance should shift toward detecting autoimmune markers or progressive neuromuscular symptoms. We specifically recommend strategic clinical checkpoints at 3, 12, and 24 months post-HZ. During these visits, patients should be screened for "red-flag" indicators such as progressive distal weakness, exercise-induced fatigue, or bulbar symptoms. Such a structured approach ensures that high-severity, low-frequency neuro-immunological sequelae are not overlooked during routine post-viral follow-up.
Validation and internal consistency
The reliability of our findings is supported by several comprehensive sensitivity analyses. To establish stringent temporal precedence, we analyzed outcomes occurring prior to or on the same date as the HZ episode (Supplementary Table S10); the risk for Bell's palsy remained significant (HR 1.428; p < 0.0001), while GBS and MG risks decreased, suggesting those primary risks were driven by concurrent or temporally complex disease processes [28, 31, 41]. To address potential detection bias, we verified that both groups averaged more than one ICD-10 entry per patient (1.291 vs. 1.521; Supplementary Table S7) and the COVID+HZ cohort demonstrated significantly higher NCV utilization (mean 0.054 vs. 0.022; p < 0.001; Supplementary Table S8), reflecting increased neurologic evaluation rather than administrative artifacts.
Notably, excluding individuals with prior immunosuppression or HIV infection had minimal impact on the results, reinforcing that the observed relationship persists in immunocompetent populations and is driven by VZV's inherent neurotropic capacity to reactivate during immune perturbation [42]. Regarding vaccination, exploratory analyses showed that while COVID-19 vaccination did not fully mitigate the elevated neuro-immunological risk in HZ-exposed patients, the risks remained consistently high across both vaccinated and unvaccinated cohorts (HRs approximately 2.67-2.91), arguing against vaccine-induced injury as a primary driver [4]. Furthermore, excluding individuals with a history of HZ vaccination did not significantly alter the HRs, indicating that the profound immune perturbation following COVID-19 may overcome the protection typically afforded by prior VZV-specific immunization. Additionally, the HZ-exposed group exhibited a markedly higher hospitalization burden (mean 2.532 vs. 1.446; p < 0.0001; Supplementary Table S11), underscoring that post-COVID HZ is associated with broader systemic morbidity [36, 43]. Finally, anti-HZ therapy showed no statistically significant reduction in Bell's palsy risk (HR 1.378; p = 0.275; Supplementary Table S12), suggesting standard treatment alone may be insufficient to fully offset these risks.
Limitations
Several limitations warrant consideration. First, as an observational cohort study, these findings demonstrate associations rather than definitive causation. While E-value analysis (Supplementary Table S14) suggests that an unmeasured confounder would need a very high effect size to nullify our results, residual confounding may remain. In particular, the primary PSM model included a limited number of covariates, and additional factors such as COVID-19 severity, socioeconomic status, and detailed immune status may not have been fully captured. Second, our reliance on ICD-10 codes without clinical adjudication (e.g., antibody titers for MG or cerebrospinal fluid analysis for GBS) carries a risk of misclassification. However, prior studies have demonstrated acceptable validity of ICD-10 coding for HZ, with reported positive predictive values exceeding 80% [44], and improved diagnostic accuracy for GBS when supported by objective testing [45]. While our NCV utilization data helps mitigate this concern, the potential for overdiagnosis of Bell's palsy in EHR databases must still be acknowledged.
Third, inherent limitations of EHR databases, including potential underdiagnosis and incomplete clinical capture, may affect case ascertainment, as discussed in prior evaluations of the TriNetX platform [46]. Fourth, while emerging AI-based diagnostic approaches [38] may provide more granular insights into systemic inflammation, current EHR structures limit our ability to incorporate detailed laboratory or electrophysiological data. Additionally, our vaccine subgroup analysis was limited by low event counts for GBS and MG, and these results should be interpreted with caution. Finally, the geographic distribution of participating healthcare organizations in TriNetX may restrict the generalizability of these findings to uninsured or non-U.S. populations.
Conclusions
HZ reactivation following COVID-19 is associated with an increased risk of Bell's palsy, GBS, and MG, reflecting distinct temporal patterns suggestive of immediate neurotropic effects and delayed immune-mediated processes. Post-COVID HZ may serve as a clinically relevant marker of neuro-immunological vulnerability, particularly among individuals with metabolic comorbidities. Importantly, COVID-19 vaccination was not associated with an increased risk of these neurological outcomes in our analysis, although subgroup findings should be interpreted with caution, given limited event counts. These findings support a symptom-triggered neurological surveillance approach, with attention to early cranial nerve involvement and delayed neuromuscular manifestations. Further prospective studies are warranted to confirm these associations and clarify the underlying mechanisms.
Abbreviations
COVID-19: coronavirus disease 2019; PNS: peripheral nervous system; HZ: herpes zoster; VZV: varicella-zoster virus; HR: hazard ratio; GBS: Guillain-Barré syndrome; MG: myasthenia gravis; EHR: electronic health record; ICD-10-CM: International Classification of Diseases, Tenth Revision, Clinical Modification; STROBE: Strengthening the Reporting of Observational Studies in Epidemiology; IRB: Institutional Review Board; PSM: propensity score matching; SMD: standardized mean difference; RR: relative risk; OR: odds ratio; CI: confidence interval; HIV: human immunodeficiency virus; NCV: nerve conduction velocity; AI: artificial intelligence.
Supplementary Material
Supplementary tables.
Acknowledgements
Special thanks to the Taipei Tzu Chi Hospital Research Department for their full support in administrative resources.
Author contributions
Yu-Chen Cheng, Chien-Lin Lu, Yi-Chou Hou, and Kuo-Cheng Lu: conceptualization; Yu-Chen Cheng and Chien-Lin Lu: literature search; Joshua Wang, Yu-Chen Cheng, and Kuo-Cheng Lu: data collection; Kuo-Cheng Lu and Joshua Wang: data analysis; Kuo-Cheng Lu: statistical analysis; Yu-Chen Cheng: manuscript preparation; Yu-Chen Cheng, Joshua Wang, and Kuo-Cheng Lu: manuscript editing; Yu-Chen Cheng, Joshua Wang, and Kuo-Cheng Lu: manuscript review.
Ethical approval
Ethical approval was granted by the Institutional Review Board of Taipei Tzu Chi Hospital (Approval Number: 14-IRB140), which waived the requirement for informed consent due to the retrospective nature of the study. All methods adhered to relevant guidelines and regulations.
Data availability statement
Due to licensing and privacy restrictions, the de-identified, aggregate-level data used in this study from the TriNetX Global Health Research Network are not publicly available. TriNetX provides access to data sourced from a global network of healthcare organizations. Researchers may request access through the TriNetX website (https://trinetx.com) or by contacting Privacy@TriNetX.com. Data are also available from the corresponding author upon reasonable request.
Competing Interests
The authors have declared that no competing interest exists.
References
1. Pang Z, Tang A, He Y, Fan J, Yang Q, Tong Y. et al. Neurological complications caused by SARS-CoV-2. Clin Microbiol Rev. 2024;37:e0013124
2. Almutairi N, Almutairi AN, Almazyad M, Alwazzan S. Herpes zoster in the era of COVID 19: A prospective observational study to probe the association of herpes zoster with COVID 19 infection and vaccination. Dermatol Ther. 2022;35:e15521
3. Ferreira A, Romao TT, Macedo YS, Pupe C, Nascimento OJM, Fellow of the American Academy of N. COVID-19 and herpes zoster co-infection presenting with trigeminal neuropathy. Eur J Neurol. 2020;27:1748-50
4. Wang F, Gao Y, Wagner AL, Lu Y. A systematic review and meta-analysis of herpes zoster occurrence/recurrence after COVID-19 infection and vaccination. J Med Virol. 2024;96:e29629
5. Chen YC, Ho CH, Liu TH, Wu JY, Huang PY, Tsai YW. et al. Long-term risk of herpes zoster following COVID-19: A retrospective cohort study of 2 442 686 patients. J Med Virol. 2023;95:e28745
6. Rahangdale RR, Tender T, Balireddy S, Pasupuleti M, Hariharapura RC. Interplay between stress and immunity triggers herpes zoster infection in COVID-19 patients: a review. Can J Microbiol. 2022;68:303-14
7. Czech T, Nishimura Y. Characteristics of herpes zoster infection in patients with COVID-19: a systematic scoping review. Int J Dermatol. 2022;61:1087-92
8. Yu X, Li L, Chan MTV, Wu WKK. Bioinformatic analyses suggest augmented interleukin-17 signaling as the mechanism of COVID-19-associated herpes zoster. Environ Sci Pollut Res Int. 2021;28:65769-75
9. Freire de Castro R, Crema D, Neiva FC, Pinto R, Suzuki FA. Prevalence of herpes zoster virus reactivation in patients diagnosed with Bell's palsy. J Laryngol Otol. 2022;136:975-8
10. Neckel N, Nahles S, Heiland M, Audebert H, Zdunczyk A, Guntinas-Lichius O. et al. Risk Factors Associated with Bell's Palsy: A Real-World Analysis of 281,600 Patients. Eur J Neurol. 2025;32:e70336
11. Wang A, Xie W, Zhang J. The synergistic role of viral infection and immune response in the pathogenesis of facial palsy. J Neurovirol. 2025;31:208-18
12. Gupta S, Jawanda MK. Surge of Bell's Palsy in the era of COVID-19: Systematic review. Eur J Neurol. 2022;29:2526-43
13. Luijten LWG, Leonhard SE, van der Eijk AA, Doets AY, Appeltshauser L, Arends S. et al. Guillain-Barre syndrome after SARS-CoV-2 infection in an international prospective cohort study. Brain. 2021;144:3392-404
14. Fragiel M, Miro O, Llorens P, Jimenez S, Pinera P, Burillo G. et al. Incidence, clinical, risk factors and outcomes of Guillain-Barre in Covid-19. Ann Neurol. 2021;89:598-603
15. Tereshko Y, Gigli GL, Pez S, De Pellegrin A, Valente M. New-onset Myasthenia Gravis after SARS-CoV-2 infection: case report and literature review. J Neurol. 2023;270:601-9
16. Croitoru CG, Cuciureanu DI, Hodorog DN, Grosu C, Cianga P. Autoimmune myasthenia gravis and COVID-19. A case report-based review. J Int Med Res. 2023;51:3000605231191025
17. Gupta S, Jawanda MK, Taneja N, Taneja T. A systematic review of Bell's Palsy as the only major neurological manifestation in COVID-19 patients. J Clin Neurosci. 2021;90:284-92
18. Mizuno T, Suzuki J, Takahashi S, Imai H, Itagaki H, Akaba T. et al. Relationship between shingles and coronavirus disease 2019: a self-controlled case series study. Int J Epidemiol. 2025 54
19. Meidaninikjeh S, Sabouni N, Taheri M, Borjkhani M, Bengar S, Majidi Zolbanin N. et al. SARS-CoV-2 and Guillain-Barre Syndrome: Lessons from Viral Infections. Viral Immunol. 2022;35:404-17
20. Shahrizaila N, Lehmann HC, Kuwabara S. Guillain-Barre syndrome. Lancet. 2021;397:1214-28
21. Gilhus NE, Verschuuren JJ. Myasthenia gravis: subgroup classification and therapeutic strategies. Lancet Neurol. 2015;14:1023-36
22. Huijbers MG, Marx A, Plomp JJ, Le Panse R, Phillips WD. Advances in the understanding of disease mechanisms of autoimmune neuromuscular junction disorders. Lancet Neurol. 2022;21:163-75
23. Haginomori S, Ichihara T, Mori A, Kanazawa A, Kawata R, Tang H. et al. Varicella-zoster virus-specific cell-mediated immunity in Ramsay Hunt syndrome. Laryngoscope. 2016;126:E35-9
24. Furuta Y, Takasu T, Fukuda S, Sato-Matsumura KC, Inuyama Y, Hondo R. et al. Detection of varicella-zoster virus DNA in human geniculate ganglia by polymerase chain reaction. J Infect Dis. 1992;166:1157-9
25. Furuta Y, Ohtani F, Kawabata H, Fukuda S, Bergstrom T. High prevalence of varicella-zoster virus reactivation in herpes simplex virus-seronegative patients with acute peripheral facial palsy. Clin Infect Dis. 2000;30:529-33
26. Grewal DS. Bell's Palsy-Tertiary Ischemia: An Etiological Factor in Residual Facial Palsy. Indian J Otolaryngol Head Neck Surg. 2018;70:374-9
27. Singh A, Deshmukh P. Bell's Palsy: A Review. Cureus. 2022;14:e30186
28. Gilden D, Cohrs RJ, Mahalingam R, Nagel MA. Varicella zoster virus vasculopathies: diverse clinical manifestations, laboratory features, pathogenesis, and treatment. Lancet Neurol. 2009;8:731-40
29. Psillas G, Dimas GG, Sarafidou A, Didangelos T, Perifanis V, Kaiafa G. et al. Evaluation of Effects of Diabetes Mellitus, Hypercholesterolemia and Hypertension on Bell's Palsy. J Clin Med. 2021 10
30. Foroughi E, Mozafar M, Haghighi S, Baerz MM, Torabi KS, Bafti AH. et al. Serum vitamin D levels in patients with Bell's palsy: a systematic review and meta-analysis. BMC Neurol. 2025;25:262
31. Anderson TC, Leung JW, Harpaz R, Dooling KL. Risk of Guillain-Barre syndrome following herpes zoster, United States, 2010-2018. Hum Vaccin Immunother. 2021;17:5304-10
32. Saha A, Batra P, Vilhekar KY, Chaturvedi P. Post-varicella myasthenia gravis. Singapore Med J. 2007;48:e177-80
33. Laman JD, Huizinga R, Boons GJ, Jacobs BC. Guillain-Barre syndrome: expanding the concept of molecular mimicry. Trends Immunol. 2022;43:296-308
34. Olson JK, Eagar TN, Miller SD. Functional activation of myelin-specific T cells by virus-induced molecular mimicry. J Immunol. 2002;169:2719-26
35. Miller SD, Vanderlugt CL, Begolka WS, Pao W, Yauch RL, Neville KL. et al. Persistent infection with Theiler's virus leads to CNS autoimmunity via epitope spreading. Nat Med. 1997;3:1133-6
36. Lu CL, Wang J, Chang YC, Lu KC. Cardiorenal outcomes after herpes zoster reactivation in COVID-19 survivors from a global TriNetX study. Sci Rep. 2025;15:30036
37. Chien MH, Wang J, Lu KC, Lu CL. Herpes Zoster Reactivation Following COVID-19 and the Risk of Renal, Infectious, and Autoimmune Complications: A Global Propensity-Matched Cohort Study. Biomedicines. 2025 13
38. Parczewski M, Kufel J, Aksak-Was B, Piwnik J, Chober D, Puzio T. et al. Artificial neural network based prediction of the lung tissue involvement as an independent in-hospital mortality and mechanical ventilation risk factor in COVID-19. J Med Virol. 2023;95:e28787
39. Hanganu AR, Niculae CM, Dulamea AO, Moisa E, Constantin R, Neagu G. et al. The outcome and risk factors associated with central and peripheral nervous system involvement in hospitalized COVID-19 patients: a retrospective cohort study. Front Neurol. 2023;14:1338593
40. Fisher SS, Lindaas A, Muthuri SG, Lloyd PC, Gruber JF, Richey MM. et al. Risk of neurologic or immune-mediated adverse events after COVID-19 diagnosis in the United States. PLoS One. 2025;20:e0333704
41. Assini A, Gandoglia I, Damato V, Rikani K, Evoli A, Del Sette M. Myasthenia gravis associated with anti-MuSK antibodies developed after SARS-CoV-2 infection. Eur J Neurol. 2021;28:3537-9
42. Steiner I, Kennedy PG, Pachner AR. The neurotropic herpes viruses: herpes simplex and varicella-zoster. Lancet Neurol. 2007;6:1015-28
43. Lu CL, Wang J, Ho CL, Wu YJ, Lu KC, Yang CC. Risk of hematologic malignancies following herpes zoster after COVID-19: a global cohort study. Front Med (Lausanne). 2025;12:1651614
44. Kim YS, Seo HM, Bang CH, Lee JH, Park YG, Kim YJ. et al. Validation of Herpes Zoster Diagnosis Code in the Electronic Medical Record: A Retrospective, Multicenter Study. Ann Dermatol. 2018;30:253-5
45. Hsieh CY, Chen PT, Shao SC, Lin SJ, Liao SC, Lai EC. Validating ICD-10 Diagnosis Codes for Guillain-Barre Syndrome in Taiwan's National Health Insurance Claims Database. Clin Epidemiol. 2024;16:733-42
46. Hochberg AR, Gomella PT, Im B, Ghosh A, Shah S, Thompson RAM. et al. Is the TriNetX Database a Good Tool for Investigation of Real-World Management of Von Hippel-Lindau? J Kidney Cancer VHL. 2024;11:28-38
Author contact
Corresponding author: Kuo-Cheng Lu, Division of Nephrology, Department of Medicine, Taipei Tzu Chi Hospital, Buddhist Tzu Chi Medical Foundation, New Taipei City, Taiwan. Email: kuochenglucom.

 Global reach, higher impact
Global reach, higher impact