Impact Factor ISSN: 1449-1907
Int J Med Sci 2026; 23(5):1748-1760. doi:10.7150/ijms.130295 This issue Cite
Research Paper
Clinical effects of glucagon-like peptide-1 receptor agonist in type 2 diabetes with low body mass index: findings from large-scale emulated target trials
1. Division of Nephrology, Department of Internal Medicine, Shin-Kong Wu Ho-Su Memorial Hospital, Taipei, Taiwan.
2. Department of Digital Medicine, Shin-Kong Wu Ho-Su Memorial Hospital, Taipei, Taiwan.
3. School of Medicine, College of Medicine, Fu Jen Catholic University, New Taipei City, Taiwan.
4. Department of Medicine, Chang Gung University, Taoyuan, Taiwan
5. Division of Nephrology, Department of Medicine, Taipei Tzu Chi Hospital, Buddhist Tzu Chi Medical Foundation, New Taipei City, Taiwan.
6. Division of Nephrology, Department of Internal Medicine, Fu Jen Catholic University Hospital, Fu Jen Catholic University, New Taipei City, Taiwan.
Received 2025-12-20; Accepted 2026-3-20; Published 2026-3-30
Abstract
Aims: Chronic kidney disease (CKD) is a common and serious complication of type 2 diabetes, yet the effectiveness of glucagon-like peptide-1 receptor agonists (GLP-1 RAs) in non-obese or mildly overweight individuals remains uncertain. This study evaluated renal, cardiovascular, and systemic outcomes associated with GLP-1 RA therapy in adults with type 2 diabetes and body mass index (BMI) ≤ 30 kg/m².
Materials and Methods: We conducted a real-world, target trial emulation, retrospective cohort study using the TriNetX US Collaborative Network. Adults with type 2 diabetes and BMI ≤30 kg/m² initiating GLP-1 RAs or dipeptidyl peptidase-4 inhibitors (DPP-4i) between 2016 and 2023 were identified. After exclusions, 23,103 GLP-1 RA and 44,156 DPP-4i users remained; 1:1 propensity score matching yielded two balanced cohorts of 20,928 patients. Outcomes—including major adverse kidney events (MAKE), progression to dialysis, cardiovascular events, hospitalization, and sepsis—were assessed over up to four years. Cox regression and Kaplan-Meier analyses estimated hazard ratios.
Results: GLP-1 RA initiation was associated with lower risks of MAKE (14.8% vs. 16.8%; HR 0.93, p = 0.005) and progression to dialysis (HR 0.78, p < 0.001). Cardiovascular outcomes and all-cause mortality were similar between groups. GLP-1 RAs significantly reduced hospitalization (HR 0.84, p < 0.001) and sepsis (HR 0.88, p = 0.001). Benefits were consistent across BMI strata and clinical subgroups, with no evidence of effect modification.
Conclusions: In adults with type 2 diabetes and BMI ≤ 30 kg/m², GLP-1 RAs confer clinically meaningful kidney protection and reduce hospitalization and sepsis, despite neutral cardiovascular effects. These findings support the use of GLP-1 RAs in non-obese or mildly overweight diabetic populations.
Keywords: Cardiovascular outcomes, GLP-1 receptor agonists, kidney outcomes, Type 2 diabetes
Introduction
Chronic kidney disease (CKD) is a major complication of type 2 diabetes and affects roughly one-quarter to one-third of patients in several Asian regions. Reported prevalence ranges from 24-35% in Thailand and India to about 31-33% in China, and studies from South Asia describe a rapid decline in kidney function once CKD develops [1-3]. A substantial portion of this burden occurs in individuals with relatively low body mass index (BMI). This “lean diabetes” pattern—common in many Asian countries—is characterized by limited adiposity but pronounced metabolic risk and is linked to earlier onset of CKD and faster loss of eGFR [4, 5]. Similar lower-BMI phenotypes are now described outside Asia as well, indicating that many patients with type 2 diabetes differ from the predominantly overweight profiles represented in most Western trials.
Glucagon-like peptide-1 receptor agonists (GLP-1 RAs) have been shown in large cardiovascular outcome trials to reduce major adverse cardiovascular events (MACE) by roughly 13-15% in high-risk type 2 diabetes populations, as demonstrated in LEADER, SUSTAIN-6, and REWIND [6-8]. Renal endpoints from these trials and related meta-analyses consistently show reduced progression of albuminuria (approximately 21-26%) and modest protection against composite kidney outcomes, including sustained eGFR decline and kidney failure [9-11]. Experimental and translational work suggests several kidney-specific actions of GLP-1 RAs, including effects on tubular sodium handling, tubuloglomerular feedback, and pathways related to renal inflammation and fibrosis, many of which appear to occur independently of weight change [12-15].
Whether these kidney benefits extend to individuals with lower BMI is less certain. Most randomized trials included patients who were overweight or obese, and individuals with BMI below 30 kg/m² constitute a minority of participants. Observational data from Taiwan suggest that the cardiovascular effects of GLP-1 RAs may differ by BMI, with clearer benefit at BMI ≥ 25 kg/m², while kidney outcomes appear less affected by adiposity [16]. Meta-analyses also indicate potential variation in cardiovascular response by BMI but little evidence that renal effects differ substantially across weight categories [17-19]. These observations raise an important question regarding the effectiveness of GLP-1 RAs in the growing number of patients with type 2 diabetes who are non-obese or only mildly overweight.
To examine this issue, we conducted a real-world, target trial emulation comparing GLP-1 RAs with dipeptidyl peptidase-4 inhibitors (DPP-4i) in adults with type 2 diabetes and BMI ≤ 30 kg/m². Using a new-user, active-comparator design with broad covariate adjustment through propensity score matching, we evaluated major adverse kidney events (MAKE), progression to dialysis, cardiovascular outcomes, hospitalization, and sepsis. Our aim was to determine whether GLP-1 RAs provide kidney and systemic benefits in patients who fall outside the typical BMI profile of prior randomized trials.
Materials and Methods
Data sources
TriNetX is a global health research platform that accelerates medical discoveries by securely connecting healthcare organizations, pharma companies, and academic researchers. It aggregates and anonymizes real-world patient data from electronic healthy records (EHRs) and insurance claims in a continuously updated, cloud-based database. Researchers use TriNetX's intuitive tools to define patient cohorts, quickly assess study feasibility, and conduct robust analyses using real data. The platform streamlines clinical trial recruitment, optimizes study design, and offers advanced analytics to visualize treatment patterns and predict health trends. Its specialized networks foster collaboration across institutions, helping tackle challenges like rare disease research. With strong privacy measures compliant with Health Insurance Portability and Accountability Act and General Data Protection Regulation, TriNetX enables comprehensive, compliant research and drives meaningful advancements in patient care and evidence-based medicine.
Ethics statement
This study was approved by the Institutional Review Board of Shin Kong Wu Ho-Su Memorial Hospital (IRB number: 20250907R) and the patients' informed consent was waived because as TriNetX provides access to a de-identified database for patient selection.
Study design and study population
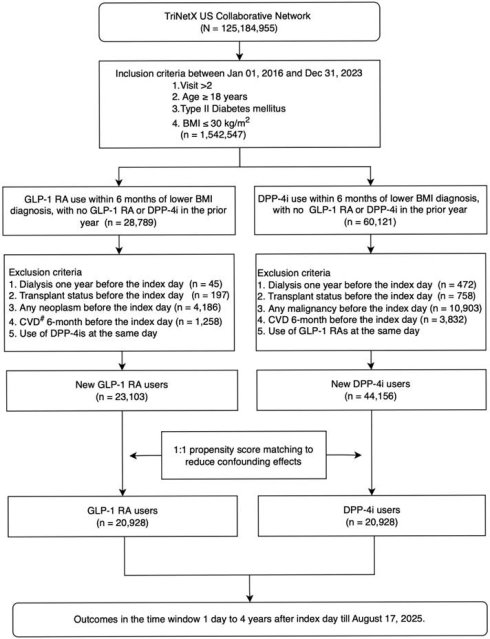
This retrospective cohort study, as illustrated in Figure 1, utilized a new-user, intention-to-treat, and active-comparator design to evaluate outcomes associated with GLP-1 RA and DPP-4 inhibitor (DPP-4i) use among adults with type 2 diabetes mellitus based on target trial emulation guidelines [20]. The analysis was based on the TriNetX US Collaborative Network, comprising a large population of 125,184,955 individuals. Patients were eligible if they were aged 18 years or older, had a diagnosis of type 2 diabetes, and a recorded BMI of 30 kg/m² or below, and had at least two clinical encounters recorded in the database between January 1, 2016, and December 31, 2023. The requirement for at least two visits was applied as a data-density criterion to ensure adequate baseline clinical information and active engagement with the healthcare system, and was not used as a requirement for survival until the end of the study period. This criterion was applied before cohort assignment and equally to both treatment groups and therefore was unlikely to introduce differential selection-related bias or immortal time bias. The index date was defined as the date of each patient's first prescription of either a GLP-1 RA or a DPP-4i within 6 months after documentation of BMI ≤ 30 kg/m2, provided that no use of either drug class occurred in the preceding year. BMI ≤ 30 kg/m2 was defined based on the most recent recorded BMI measurement prior to treatment initiation and was not required to be sustained over time. Longitudinal weight trajectories before cohort entry could not be fully characterized due to platform-level data constraints.
The schema of patient enrollment in the study. Abbreviation: eGFR, estimated glomerular filtration rate.
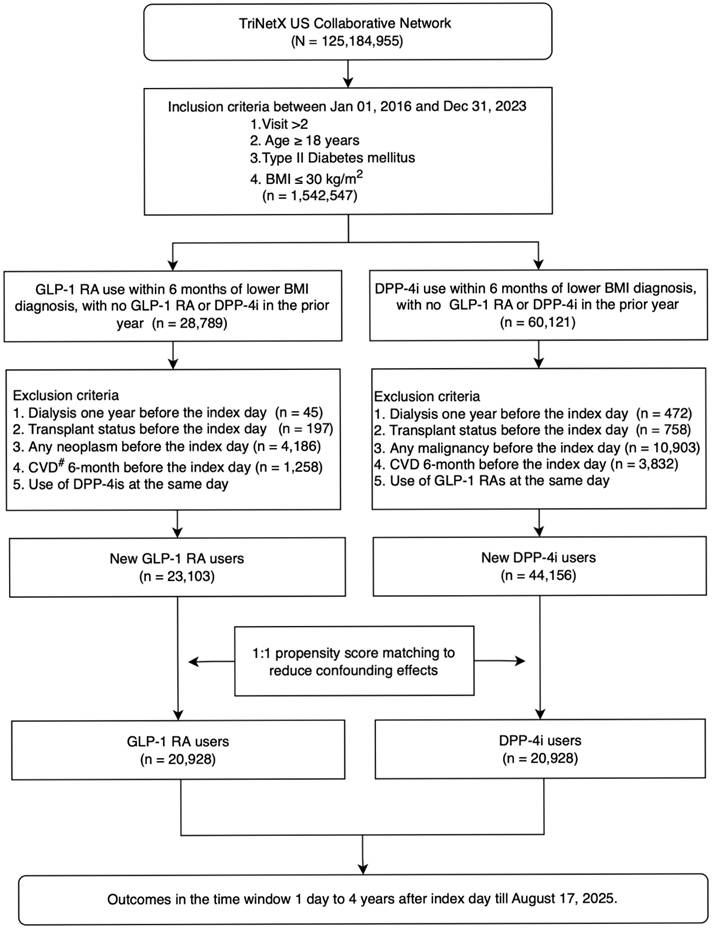
Follow-up for each patient began at the index date and continued until the occurrence of an outcome event, loss to follow-up, or the end of the study period. Two mutually exclusive new-user cohorts were identified: GLP-1 RA users (n = 28,789) and DPP-4i users (n = 60,121), from which patients meeting specific exclusion criteria—such as history of dialysis, transplant, neoplasms or malignancy, recent cardiovascular disease, or concurrent use of the comparator drug class—were removed. This process yielded 23,103 new GLP-1 RA users and 44,156 new DPP-4i users.
To control for confounding, 1:1 propensity score matching (PSM) was performed, resulting in two balanced groups, each comprising 20,928 patients. Outcomes were assessed in a time window from 1 day up to 4 years following the index date, through August 17, 2025. Cohort definitions are described in A Supplementary Method S1.
Covariates
Baseline covariates included demographic variables, comorbidities, medication use, and laboratory measurements recorded within 1 year before the index date: The baseline characteristics assessed in this study encompassed a diverse range of demographic, clinical, and laboratory parameters. Demographic factors included BMI, age at index, sex distribution (male), and racial/ethnic composition (White, African American, Asian, and other races). Furthermore, comorbidity data were collected for hypertensive diseases, dyslipidemia, ischemic heart diseases, disorders of thyroid gland, other anxiety disorders, nicotine dependence, hepatic failure, cerebrovascular diseases, fatty liver, gout, cardiomyopathy, pulmonary heart disease, atherosclerosis of native arteries of the extremities, liver cirrhosis, and chronic rheumatic heart diseases. In addition, medication use was documented, including insulin, biguanides, sulfonylureas, sodium-glucose co-transporter 2 inhibitors (SGLT2i), thiazolidinediones, statins, fibrates, renin-angiotensin system blockade (RASB), beta-blockers, calcium channel blockers, antithrombotic agents, nonsteroidal anti-inflammatory drugs (NSAIDs), immunosuppressants, and immunostimulants. Moreover, comprehensive laboratory measurements were obtained, encompassing estimated glomerular filtration rate (eGFR), urea nitrogen, sodium, potassium, calcium, phosphate, albumin, hemoglobin, hemoglobin A1c, alanine aminotransferase (ALT), aspartate aminotransferase (AST), alkaline phosphatase, low-density lipoprotein (LDL), high-density lipoprotein (HDL), triglycerides, C-reactive protein, and urine albumin-to-creatinine ratio (UACR). eGFR was calculated using the 2021 Chronic Kidney Disease Epidemiology Collaboration (CKD-EPI) equation without inclusion of a race coefficient [21]. The codes for the baseline covariates are shown in Supplementary Method S1.
Study outcomes
The study focused on several key clinical outcomes that occurred within a time window of three to four years after the index event, with follow-up data collected until August 17, 2025. The primary outcome assessed was MAKE, which included acute kidney injury, end-stage kidney disease, initiation of dialysis, and death. AKI was identified using diagnosis codes for acute kidney failure (ICD-10-CM N17). ESKD on dialysis was defined using diagnosis codes for end-stage renal disease (ICD-10-CM N18.6) and dependence on renal dialysis (ICD-10-CM Z99.2), as well as dialysis-related procedure codes. In addition to the composite primary outcome, several secondary outcomes were evaluated to capture broader clinical implications. These included individual kidney-related outcomes such as entering dialysis and acute kidney injury, as well as major cardiovascular and health outcomes such as all-cause mortality, acute myocardial infarction (AMI), heart failure, stroke, hospitalization for any cause, and sepsis. Together, these outcomes provided a comprehensive view of renal, cardiovascular, and overall health trajectories among patients over the follow-up period. The outcome codes are listed in Supplementary Method S1.
Sensitivity analysis
To ensure our findings were robust, we conducted four sensitivity analyses to address potential limitations and confirm the consistency of our results. First, we applied different propensity score matching (PSM) models, each adjusted for a range of confounding factors, to minimize bias and isolate the effects under investigation. Second, we assessed the risk across multiple time intervals to address possible violations of the proportional hazard assumption, ensuring the validity of our conclusions over time. Third, we performed analyses using 3-month lags after the index date to test the stability of our results with respect to different latency periods. Finally, the E-value was calculated for each result as a sensitivity analysis to quantify the minimal strength of association that an unmeasured confounder would need to have with both the exposure and the outcome to fully explain away the observed association, after accounting for the measured covariates.[22] These approaches collectively strengthened the credibility and generalizability of our findings.
Statistical analysis
Categorical variables were presented as counts and percentages, while numerical variables were reported as means with standard deviations. Group differences were assessed using standardized mean differences (SMD) [23], considering values below 0.1 as an indication of well-balanced groups. To minimize baseline confounding and improve comparability, PSM was implemented, resulting in two matched cohorts [24]. This approach allowed for more accurate evaluation of treatment effects by ensuring groups were similar across baseline demographic, clinical, and laboratory characteristics. Propensity scores were derived utilizing logistic regression and subsequently matched through a greedy nearest-neighbor approach with a 0.1 caliper, aimed at achieving equilibrium among treatment groups for the purpose of analysis. In the process of PSM, the age at the index date was regarded as a continuous variable, whereas sex, race, lifestyle attributes, comorbid conditions, and pharmacological interventions were classified as categorical (present/absent). Laboratory metrics were deemed present if they were obtainable, or absent if not recorded within the designated timeframe.
Cox proportional hazards regression was used to calculate hazard ratios (HRs) for comparing clinical outcome risks between groups. The proportional hazards assumption was evaluated using the generalized Schoenfeld method within TriNetX. Event-free probabilities were estimated with Kaplan-Meier curves, providing a visual comparison of outcome incidence over time. In the present investigation, we examined 9 hypotheses while anticipating a false discovery rate of 0.05. The Benjamini-Hochberg approach was employed to adjust the P values accordingly.
Subgroup analyses were performed, with all subgroups being re-matched through propensity score matching (PSM) based on initial characteristics to guarantee well-balanced and comparable pairs. Because interaction testing is not directly supported in the TriNetX interface, subgroup heterogeneity was assessed using Cochran's Q test and I2 statistics in a random-effects model [25]. The p-value for heterogeneity was calculated to determine whether the influence of SGLT2 inhibitors on clinical outcomes demonstrated significant variability across different subgroups.
All statistical analysis was conducted in the TriNetX platform. A two-sided P-value of < 0.05 was considered significant. R software version 4.4.2 (Free Software Foundation Inc.), with the Forestploter and ggplot2 packages, was used to create the figures in this study.
Results
Baseline characteristics of patients
Figure 1 summarizes cohort selection. Among 67,259 adults with type 2 diabetes and BMI ≤ 30 kg/m², exclusions included prior GLP-1 RA or DPP4i exposure, kidney failure, transplantation, malignancy, recent cardiovascular events, and concurrent comparator use, yielding 23,103 GLP-1 RA initiators and 44,156 DPP4i initiators before matching. After 1:1 propensity score matching, two balanced cohorts of 20,928 patients each were formed. Baseline characteristics were well aligned, with all standardized mean differences < 0.1 (Table 1). Mean age was approximately 61 years, and 52% were male. Comorbidities, laboratory values, and concomitant medications were highly comparable between groups. The study structure, exposure definition, and follow-up scheme followed a prespecified target trial framework (Table S1), and the overall design—including eligibility windows, assignment, and index date construction—is shown in Figure S1.
Baseline characteristics in the population with lower body mass index (<30 Kg/m2)
| Before matching | After matching | |||||
|---|---|---|---|---|---|---|
| Parameter | GLP-1 RA (n = 23,103) | DPP4i (n = 44,456) | SMD | GLP-1 RA (n = 20,928) | DPP4i (n = 20,928) | SMD |
| BMI (kg/m2), mean ± SD | 26.6 ± 2.7 | 25.7 ± 3.3 | - | 26.6 ± 2.8 | 26 ± 3.1 | - |
| Age at Index (years), mean ± SD | 60.6 ± 11.8 | 65.1 ± 11.7 | 0.383 | 61.4 ± 11.4 | 61.3 ± 12.2 | 0.008 |
| Male, n (%) | 11,895 (51.5) | 24,730 (55.6) | 0.083 | 10,943 (52.3) | 11,059 (52.8) | 0.011 |
| Race, n (%) | ||||||
| White | 13,822 (59.8) | 22,537 (50.7) | 0.184 | 12,296 (58.8) | 12,265 (58.6) | 0.003 |
| African American | 3,313 (14.3) | 6,644 (14.9) | 0.017 | 3,029 (14.5) | 3,063 (14.6) | 0.005 |
| Asian | 1,679 (7.3) | 6,194 (13.9) | 0.218 | 1,648 (7.9) | 1,589 (7.6) | 0.011 |
| Other race | 1,539 (6.7) | 3,058 (6.9) | 0.009 | 1,394 (6.7) | 1,393 (6.7) | < 0.001 |
| Comorbidity, n (%) | ||||||
| Hypertensive diseases | 13,132 (56.8) | 28,399 (63.9) | 0.144 | 12,141 (58) | 11,999 (57.3) | 0.014 |
| Dyslipidemia | 13,467 (58.3) | 24,927 (56.1) | 0.045 | 12,079 (57.7) | 11,997 (57.3) | 0.008 |
| Ischemic heart diseases | 3,077 (13.3) | 7,860 (17.7) | 0.121 | 2,912 (13.9) | 2,877 (13.7) | 0.005 |
| Disorders of thyroid gland | 3,199 (13.8) | 5,595 (12.6) | 0.037 | 2,836 (13.6) | 2,765 (13.2) | 0.010 |
| Other anxiety disorders | 2,057 (8.9) | 3,188 (7.2) | 0.064 | 1,796 (8.6) | 1,768 (8.4) | 0.005 |
| Nicotine dependence | 1,549 (6.7) | 3,413 (7.7) | 0.038 | 1,444 (6.9) | 1,482 (7.1) | 0.007 |
| Hepatic failure | 1,076 (4.7) | 3,466 (7.8) | 0.130 | 1,048 (5) | 1,007 (4.8) | 0.009 |
| Cerebrovascular diseases | 863 (3.7) | 2,586 (5.8) | 0.098 | 830 (4) | 811 (3.9) | 0.005 |
| Fatty liver | 603 (2.6) | 880 (2) | 0.042 | 515 (2.5) | 520 (2.5) | 0.002 |
| Gout | 391 (1.7) | 1,089 (2.5) | 0.053 | 369 (1.8) | 369 (1.8) | 0.000 |
| Cardiomyopathy | 329 (1.4) | 931 (2.1) | 0.051 | 313 (1.5) | 282 (1.3) | 0.013 |
| Pulmonary heart disease | 289 (1.3) | 997 (2.2) | 0.076 | 284 (1.4) | 268 (1.3) | 0.007 |
| Atherosclerosis of native arteries of the extremities | 288 (1.2) | 846 (1.9) | 0.053 | 273 (1.3) | 256 (1.2) | 0.007 |
| Liver cirrhosis | 276 (1.2) | 618 (1.4) | 0.017 | 255 (1.2) | 267 (1.3) | 0.005 |
| Chronic rheumatic heart diseases | 223 (1) | 787 (1.8) | 0.069 | 220 (1.1) | 207 (1) | 0.006 |
| Medication, n (%) | ||||||
| Insulin | 10,382 (44.9) | 16,669 (37.5) | 0.152 | 8,819 (42.1) | 8,724 (41.7) | 0.009 |
| Biguanides | 12919 (58.8) | 26,643 (64.9) | 0.126 | 1,1596 (60.7) | 11,692 (61.2) | 0.010 |
| Sulfonylureas | 5,191 (22.5) | 13,695 (30.8) | 0.189 | 4,975 (23.8) | 4,931 (23.6) | 0.005 |
| SGLT2i | 6,114 (26.5) | 6,485 (14.6) | 0.297 | 4,867 (23.3) | 4,813 (23) | 0.006 |
| Thiazolidinediones | 1,392 (6) | 2,490 (5.6) | 0.018 | 1,219 (5.8) | 1,223 (5.8) | 0.001 |
| Statin | 14,091 (61) | 28,915 (65) | 0.084 | 12,818 (61.2) | 12,786 (61.1) | 0.003 |
| Fibrates | 1,312 (5.7) | 2,468 (5.6) | 0.006 | 1,193 (5.7) | 1,161 (5.5) | 0.007 |
| RASB | 11,810 (51.1) | 24,922 (56.1) | 0.099 | 10,837 (51.8) | 10,749 (51.4) | 0.008 |
| Beta-blocker | 6,280 (27.2) | 16,576 (37.3) | 0.217 | 6,009 (28.7) | 5,878 (28.1) | 0.014 |
| Calcium channel blockers | 4,374 (18.9) | 12,005 (27) | 0.193 | 4,207 (20.1) | 4,110 (19.6) | 0.012 |
| Antithrombotic agents | 7,920 (34.3) | 20,963 (47.2) | 0.264 | 7,574 (36.2) | 7,477 (35.7) | 0.010 |
| NSAID | 4,908 (21.2) | 9,468 (21.3) | 0.001 | 4,413 (21.1) | 4,412 (21.1) | < 0.001 |
| Immunosuppressants | 577 (2.5) | 1,051 (2.4) | 0.009 | 504 (2.4) | 499 (2.4) | 0.002 |
| Immunostimulants | 15 (0.1) | 51 (0.1) | 0.017 | 15 (0.1) | 14 (0.1) | 0.002 |
| Laboratory, mean ± SD | ||||||
| eGFR (mL/min/1.73m2) | 78.4 ± 29.8 | 73.4 ± 32.5 | 0.161 | 77.5 ± 29.8 | 79.4 ± 31.8 | 0.060 |
| Urea nitrogen (mg/dL) | 18.4 ± 9.9 | 20.4 ± 12.8 | 0.176 | 18.6 ± 10.1 | 18.3 ± 10.7 | 0.025 |
| Sodium (mmol/L) | 138 ± 3.3 | 137.9 ± 3.7 | 0.015 | 138 ± 3.3 | 137.9 ± 3.5 | 0.017 |
| Potassium (mmol/L) | 4.3 ± 0.5 | 4.3 ± 0.5 | 0.010 | 4.3 ± 0.5 | 4.3 ± 0.5 | 0.005 |
| Calcium (mg/dL) | 9.4 ± 0.6 | 9.3 ± 0.7 | 0.203 | 9.4 ± 0.6 | 9.4 ± 0.6 | 0.034 |
| Phosphate (mg/dL) | 3.4 ± 0.9 | 3.5 ± 1 | 0.009 | 3.4 ± 0.9 | 3.4 ± 1 | 0.009 |
| Albumin (g/dL) | 4.1 ± 0.6 | 4 ± 0.6 | 0.248 | 4.1 ± 0.6 | 4.1 ± 0.6 | 0.014 |
| Hemoglobin (g/dL) | 13.5 ± 2.1 | 12.7 ± 2.3 | 0.348 | 13.4 ± 2.1 | 13.4 ± 2.1 | 0.023 |
| Hemoglobin A1c (%) | 8.7 ± 2.3 | 8.3 ± 2.1 | 0.203 | 8.6 ± 2.3 | 8.6 ± 2.2 | 0.010 |
| ALT (U/L) | 28.6 ± 27.8 | 29.1 ± 59.8 | 0.010 | 28.6 ± 28.2 | 29.1 ± 50.7 | 0.013 |
| AST (U/L) | 24.9 ± 24.8 | 27.2 ± 69 | 0.043 | 25 ± 25.4 | 25.7 ± 52.6 | 0.017 |
| Alkaline phosphatase (U/L) | 87.9 ± 45 | 86.7 ± 51.2 | 0.024 | 88 ± 45.9 | 85.8 ± 48.5 | 0.045 |
| LDL (mg/dL) | 94.6 ± 42.3 | 91.3 ± 40.2 | 0.078 | 94.1 ± 42.4 | 94.3 ± 40.8 | 0.004 |
| HDL (mg/dL) | 45.5 ± 15.7 | 44.7 ± 16.6 | 0.052 | 45.6 ± 15.8 | 44.4 ± 16.4 | 0.076 |
| Triglyceride (mg/dL) | 188.2 ± 249.5 | 170.6 ± 173.9 | 0.082 | 183.9 ± 221.5 | 181.8 ± 198.3 | 0.010 |
| C reactive protein (mg/L) | 41.5 ± 71.9 | 46.7 ± 69.2 | 0.074 | 43.7 ± 73 | 41.3 ± 67.1 | 0.034 |
| UACR (mg/g) | 639.4 ± 13881.5 | 1187.4 ± 36154 | 0.020 | 702.3 ± 14741.3 | 310 ± 3253.7 | 0.037 |
Abbreviation: BMI, body mass index; GLP-1 RA, glucagon-like peptide-1 receptor agonists; DPP4i, dipeptidyl peptidase-4 inhibitor; SGLT2i, sodium-glucose co-transporter 2 Inhibitors; RASB, renin-angiotensin system blockade; NSAID, nonsteroidal anti-inflammatory drug.; eGFR, estimated glomerular filtration rate; ALT, alanine aminotransferase; AST, aspartate aminotransferase; LDL, low density lipoprotein; HDL, high density lipoprotein; UACR, urine albumin / creatinine ratio;
Laboratory data were incompletely available for a substantial proportion of patients. In particular, urine albumin-to-creatinine ratio (uACR) measurements were missing in most individuals, with missing rates of 95.1% in the GLP-1 RA group and 96.5% in the DPP-4i group after matching (Supplementary Method S1). Accordingly, uACR was not incorporated into the primary propensity score model and was interpreted cautiously in subgroup analyses.
Major adverse kidney outcomes
Kaplan-Meier curves demonstrated significantly higher event-free survival among GLP-1 RA users for MAKE (p = 0.005) and ESKD requiring dialysis (p < 0.001) (Figure S2A-B). No differences were observed in all-cause mortality (p = 0.818) or acute kidney injury (AKI; p = 0.222) (Figure S2C-D). GLP-1 RA use was also associated with a lower risk of sepsis (p = 0.001) (Figure S2E). As shown in Table 2, MAKE occurred in 14.8% of GLP-1 RA users versus 16.8% of DPP4i users (HR 0.93, 95% CI 0.89-0.98). The risk of ESKD on dialysis was significantly lower with GLP-1 RA (HR 0.78, 95% CI 0.70-0.87), whereas risks of AKI, acute myocardial infarction, and mortality were similar between groups. Associations for MAKE and dialysis remained significant after false discovery rate correction, and E-value analysis suggested that the observed associations for MAKE (1.36) and dialysis (1.88) would require a confounder with at least moderate strength of association with both treatment and outcome to be fully explained away. Subsequent to the implementation of statistical adjustments for multiple comparisons, those levels of significance were preserved.
Clinical outcomes in type 2 diabetes patients with low BMI (<30 kg/m2)
| Clinical Outcomes | GLP-1 RA user (n = 20,928) | DPP4i user (n = 20,928) | GLP-1 RA vs. DPP4i | |||||
|---|---|---|---|---|---|---|---|---|
| Events (n) | % | Events (n) | % | HR (95%CI) | P value | FDR-corrected p value | E-value | |
| Primary outcome | ||||||||
| #MAKE | 3,097 | 14.8 | 3,507 | 16.8 | 0.93 (0.89-0.98) | 0.005† | 0.011 | 1.36 |
| AKI | 2,218 | 10.6 | 2,446 | 11.7 | 0.97 (0.91-1.02) | 0.222† | 0.285 | 1.21 |
| ESKD on dialysis | 573 | 2.7 | 765 | 3.7 | 0.78 (0.7-0.87) | < 0.001† | 0.009 | 1.88 |
| Mortality | 987 | 4.7 | 1,069 | 5.1 | 1.01 (0.93-1.1) | 0.818 | 0.888 | 1.11 |
| Secondary outcome | ||||||||
| AMI | 862 | 4.1 | 863 | 4.1 | 1.08 (0.98-1.18) | 0.121 | 0.182 | 1.37 |
| Heart failure | 2,125 | 10.2 | 2,252 | 10.8 | 0.99 (0.94-1.05) | 0.789 | 0.888 | 1.11 |
| Stroke | 950 | 4.5 | 944 | 4.5 | 1.08 (0.99-1.18) | 0.086 | 0.155 | 1.37 |
| Hospitalization | 4,358 | 20.8 | 5,347 | 25.5 | 0.84 (0.8-0.87) | < 0.001† | 0.009 | 1.67 |
| Sepsis | 1,149 | 5.5 | 1,401 | 6.7 | 0.88 (0.81-0.95) | 0.001 | 0.009 | 1.53 |
#MAKE includes acute kidney injury, end stage of kidney disease, entering dialysis, and death. †This indicate the proportional hazard assumption is violated.
Abbreviation: HR, hazard ratio; CI, confidence interval; MAKE, major adverse kidney events; AKI, acute kidney injury; ESKD, end stage of kidney disease; AMI, acute myocardial infarction; FDR, Benjamini-Hochberg false discovery rate.
Other clinical outcomes
In secondary analyses, GLP-1 RA users had significantly lower cumulative incidence of hospitalization (p < 0.001) and sepsis (p = 0.001), while event-free survival for acute myocardial infarction, heart failure, and stroke did not differ between groups (Figure S3). Hazard ratios from Table 2 were consistent with the KM findings: hospitalization (HR 0.84, p < 0.001) and sepsis (HR 0.88, p = 0.001) were significantly reduced, whereas cardiovascular outcomes and mortality were neutral (HRs ≈ 1.00). Similar robustness was observed for secondary outcomes, including hospitalization (E-value = 1.67) and sepsis (E-value = 1.53).
BMI-stratified analyses
Associations were generally consistent across BMI levels (Table 3). Among patients with BMI 25-30 kg/m², GLP-1 RA use was associated with reduced risks of entering dialysis (HR 0.87, 95% CI 0.74-1.01), AKI (HR 0.92, 95% CI 0.84-0.99), hospitalization (HR 0.84, 95% CI 0.79-0.86), and sepsis (HR 0.81, 95% CI 0.72-0.90). In those with BMI < 25 kg/m², GLP-1 RA therapy similarly lowered risks of entering dialysis (HR 0.72, 95% CI 0.54-0.95), hospitalization (HR 0.81, 95% CI 0.73-0.90), and sepsis (HR 0.80, 95% CI 0.65-0.98). Mortality and cardiovascular outcomes remained neutral across all BMI strata. Overall, renal and systemic benefits were preserved irrespective of BMI category.
Clinical outcomes analysis by different BMI level groups
| Clinical Outcomes | BMI 25-30 kg/m2 | BMI <25 kg/m2 | ||||
|---|---|---|---|---|---|---|
| GLP-1 RA vs. DPP4i | GLP-1 RA vs. DPP4i (n = 2,482 parings) | |||||
| HR (95%CI) | P value | E-value | HR (95%CI) | P value | E-value | |
| Primary outcome | ||||||
| #MAKE | 0.92 (0.86-0.99) | 0.022 | 1.39 | 0.91 (0.80-1.04) | 0.163 | 1.43 |
| AKI | 0.92 (0.84-0.99) | 0.030 | 1.39 | 0.96 (0.82-1.13) | 0.627 | 1.25 |
| ESKD on dialysis | 0.87 (0.74-1.01) | 0.074 | 1.56 | 0.72 (0.54-0.95) | 0.021 | 2.12 |
| Mortality | 0.99 (0.87-1.12) | 0.814 | 1.11 | 1.04 (0.82-1.33) | 0.742 | 1.24 |
| Secondary outcome | ||||||
| AMI | 0.98 (0.86-1.13) | 0.817 | 1.16 | 1.02 (0.76-1.36) | 0.917 | 1.16 |
| Heart failure | 1.01 (0.93-1.10) | 0.747 | 1.11 | 1.04 (0.88-1.24) | 0.649 | 1.24 |
| Stroke | 0.98 (0.86-1.12) | 0.806 | 1.16 | 1.13 (0.87-1.47) | 0.351 | 1.51 |
| Hospitalization | 0.84 (0.79-0.86) | <0.001 | 1.67 | 0.81 (0.73-0.90) | <0.001 | 1.77 |
| Sepsis | 0.81 (0.72-0.90) | <0.001 | 1.77 | 0.80 (0.65-0.98) | 0.027 | 1.81 |
#MAKE includes acute kidney injury, end stage of kidney disease, entering dialysis, and death. †This indicate the proportional hazard assumption is violated.
Abbreviation: HR, hazard ratio; CI, confidence interval; MAKE, major adverse kidney events; AKI, acute kidney injury; ESKD, end stage of kidney disease; AMI, acute myocardial infarction.
Subgroup analysis
Subgroup analyses in Figure 2 showed that the point estimates consistently favored GLP-1 RA over DPP-4i for both MAKE and dialysis across most demographic and clinical strata. Statistically significant reductions in MAKE were observed primarily among males, younger individuals (18-64 years), and those with BMI 25-30 kg/m2. Race-specific analyses demonstrated significant benefit in White and African American patients. As shown in Figure S4, although not all subgroups reached statistical significance, the hazard ratios were generally below 1.0, and no significant treatment-subgroup interactions were detected.
Subgroup analyses of MAKE and ESKD on dialysis comparing GLP-1 RA and DPP-4 inhibitor users. (A) Hazard ratios for MAKE and (B) ESKD requiring dialysis across key clinical subgroups. Points represent hazard ratios with 95% CIs; the dashed line denotes HR = 1.0. Values <1.0 favor GLP-1 RA over DPP-4i. Abbreviations: MAKE, major adverse kidney events; ESKD, end-stage kidney disease; eGFR, estimated glomerular filtration rate; HF, heart failure; GLP-1 RA, glucagon-like peptide-1 receptor agonist; DPP-4i, dipeptidyl peptidase-4 inhibitor.
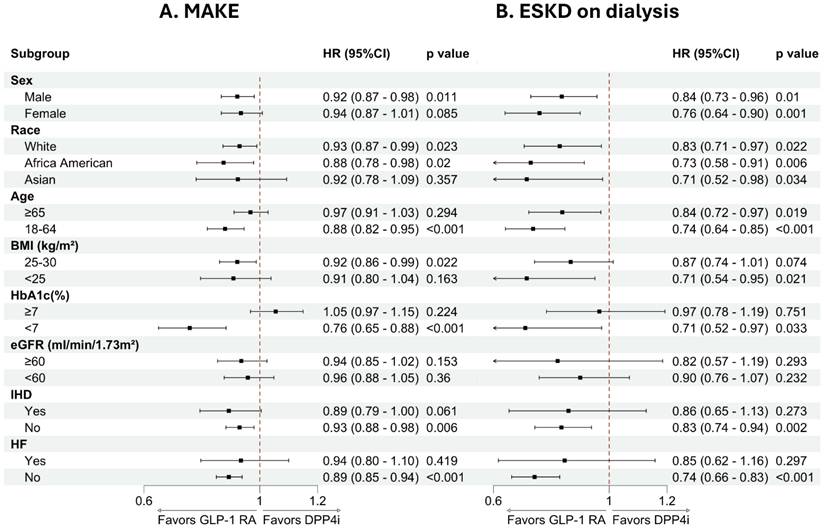
For the dialysis outcome (Figure S5), subgroup analyses demonstrated a similar overall pattern of risk reduction across strata. Statistically significant associations were observed in both sexes, across racial groups, and in both age categories. Reductions in dialysis risk were particularly evident among individuals with BMI <25 kg/m2 and those without heart failure. For other subgroups, the direction of effect consistently favored GLP-1 RA, although statistical significance was not uniformly observed.
Sensitivity analysis
Findings were robust across all sensitivity analyses. Results were consistent across multiple propensity score models (Table S2), across 1-, 2-, and 3-year follow-up windows (Table S3), and after applying a 3-month lag to minimize reverse causation (Table S4). The E-values for the significant outcomes (1.36-1.88) indicate the minimum strength of association that an unmeasured confounder would need to have with both treatment and outcome to fully account for the observed associations (Table 2). Although residual confounding cannot be excluded, the consistency of findings across multiple outcomes supports the robustness of the observed associations.
Discussion
In this large real-world cohort of adults with type 2 diabetes and BMI ≤30 kg/m², initiation of GLP-1 receptor agonists, compared with DPP-4 inhibitors, was associated with a modest but steady reduction in MAKE and a more pronounced reduction in progression to dialysis. These kidney benefits were accompanied by lower risks of hospitalization and sepsis, whereas cardiovascular outcomes and overall mortality did not differ between groups. The preservation of these benefits across BMI categories (<25 and 25-30 kg/m²) and across multiple clinical subgroups suggests that GLP-1 RA therapy may offer meaningful kidney and systemic protection in non-obese or mildly overweight individuals with type 2 diabetes, even in the absence of measurable cardiovascular advantages.
Our results parallel observations from a recent nationwide cohort in Taiwan using the Chang Gung Research Database, in which GLP-1 RA therapy was linked to substantial reductions in MACE, cardiovascular death, and heart failure hospitalization among patients with BMI ≥ 25 kg/m², but not in those with BMI < 25 kg/m² [16]. In that study, kidney outcomes—defined by major eGFR decline or dialysis—were comparable across BMI strata. A similar pattern emerged in our analysis: cardiovascular outcomes remained neutral within the BMI-restricted population, whereas kidney protection was evident. By applying a target trial emulation and excluding individuals with baseline kidney failure or early outcome events, the present study provides further evidence that the nephroprotective properties of GLP-1 RAs are maintained even when cardiovascular effects are less pronounced. This is consistent with several recent meta-analyses showing that reductions in MACE, cardiovascular death, and myocardial infarction are concentrated in patients with BMI ≥ 25 kg/m², with considerably weaker effects in those with lower BMI [17-19]. These findings support a biologically plausible mechanism in which obesity-driven inflammation and insulin resistance amplify cardiovascular responsiveness to GLP-1 RA therapy [26-30].
The kidney outcomes in our study align with findings from cardiovascular outcome trials such as LEADER, SUSTAIN-6, and REWIND, in which liraglutide, semaglutide, and dulaglutide lowered the risk of composite kidney outcomes by hazard ratios of roughly 0.64-0.85, driven mainly by reduced macroalbuminuria, with additional moderate slowing of eGFR decline and some indication of reduced kidney replacement therapy [31-34]. Pooled analyses show that these renal benefits are stable across BMI categories and other clinical subgroups [35-38]. Several biological processes may underlie the largely BMI-independent nature of kidney protection. Experimental studies demonstrate that GLP-1 signaling induces natriuresis through inhibition of the proximal tubular sodium-hydrogen exchanger NHE3 [14, 39], improves intraglomerular hemodynamics by reducing hyperfiltration and strengthening tubuloglomerular feedback [15, 40], and acts directly on GLP-1 receptors expressed in renal tubular and vascular cells [14]. Beyond these hemodynamic effects, GLP-1 RAs reduce renal inflammation, oxidative injury, and fibrosis via PKC-related pathways, PKA activation, and modulation of TGF-β/Smad signaling [41-43]. These mechanisms do not rely on weight loss or changes in BMI and fit well with the kidney protection observed in our non-obese cohort, including individuals with BMI < 25 kg/m².
The cardiovascular results contrasted with the renal findings. Rates of myocardial infarction, heart failure, stroke, and mortality were similar between groups, and survival curves did not diverge. Large cardiovascular outcome trials have shown MACE reduction with GLP-1 RAs in high-risk populations [10, 19, 33, 44-46], and the Taiwanese cohort reported clear benefits among users with BMI ≥ 25 kg/m² [16]. The lack of signal in our BMI-restricted cohort may reflect the influence of body composition on treatment response. Obesity accompanies marked insulin resistance, expanded visceral fat, and chronic low-grade inflammation—factors that contribute directly to endothelial dysfunction, atherosclerotic progression, and myocardial remodeling. GLP-1 RAs can counteract these abnormalities by improving insulin sensitivity in muscle and adipose tissue, reducing visceral fat, and lowering systemic inflammatory cytokines such as TNF-α and IL-6; these responses tend to be stronger in individuals with higher BMI [29, 47]. This biological context helps explain why cardiovascular effects were muted in our non-obese population despite clear kidney benefits [48, 49]. Evidence from SELECT and STEP-HFpEF further supports this interpretation, as semaglutide improved heart failure symptoms and reduced MACE in overweight and obese patients—including those without diabetes—highlighting the central role of adiposity in shaping therapeutic response [50-52]. In a cohort with limited adipose burden and relatively lower inflammatory activity, the treatment window for detectable cardiovascular benefit may simply be narrower.
In addition, the restriction of the study population to individuals with BMI ≤30 kg/m2 may have contributed to the absence of a detectable mortality benefit. Compared with populations enrolled in major cardiovascular outcome trials, our cohort likely had a lower baseline cardiovascular risk, which reduces statistical power to detect differences in hard endpoints such as death. This design choice was intentional, as our primary aim was to address the evidence gap in non-obese and mildly overweight patients with type 2 diabetes, who have been underrepresented in prior GLP-1 RA trials. Nevertheless, this focus necessarily limits the generalizability of our findings to patients with higher BMI, and our results should not be interpreted as excluding potential mortality benefits in more obese or higher-risk populations.
Beyond kidney and cardiovascular outcomes, our study identified meaningful reductions in hospitalization and sepsis among GLP-1 RA users, with divergence of curves early in follow-up. Similar reductions in infection-related hospitalizations were reported in the Chang Gung cohort across BMI categories [16]. Several biologic processes may contribute. Improved glycemic stability may reduce exposure to extreme hyperglycemia, which is known to compromise innate and adaptive immunity. Preclinical studies have shown that GLP-1 analogs blunt inflammatory signaling triggered by endotoxin exposure, reduce vascular oxidative stress, preserve myocardial performance, and improve survival in models of endotoxemia and sepsis [53, 54]. GLP-1 RAs also exert broader immune effects, including suppression of inflammatory cytokines, promotion of M2 macrophage activity, and downregulation of NF-κB signaling [55, 56]. Reduced macrophage infiltration in adipose tissue and lower systemic inflammatory markers have been documented as well [57]. Although our observational design does not allow direct attribution of the reduced sepsis risk to any specific pathway, the pattern is consistent with a combination of metabolic stabilization, kidney preservation, and systemic anti-inflammatory effects.
Subgroup analyses provided further insight into the consistency of treatment effects across clinical strata. The direction of association for both MAKE and dialysis generally favored GLP-1 RA therapy across age, sex, race, BMI, glycemic control, and comorbidity subgroups, with no signal of harm in any stratum. Statistically significant reductions in MAKE were mainly observed among males, younger individuals, and those with BMI 25-30 kg/m2, whereas reductions in dialysis risk were evident across both sexes and age categories and were particularly apparent among individuals without heart failure. These findings suggest that the renal benefits of GLP-1 RAs are broadly applicable in non-obese or mildly overweight patients with type 2 diabetes, rather than being confined to a narrowly defined clinical phenotype. The use of DPP-4 inhibitors as an active comparator further contextualizes the findings: EXAMINE, TECOS, and other major trials have shown neutral cardiovascular and kidney effects for this class [58, 59], making it a suitable reference rather than a competing cardiorenal therapy.
Several limitations warrant attention. Although the cohort was diverse, the applicability of our findings to Asian populations—where lean diabetes phenotypes are common—remains uncertain due to differences in diet, genetics, and clinical care. At the same time, the deliberate restriction to individuals with BMI ≤ 30 kg/m² represents a strength, as non-obese patients remain underrepresented in global GLP-1 RA trials. However, BMI ≤3 0 kg/m² in this study was defined based on a single recorded measurement rather than on longitudinal weight trajectories. Consequently, we were unable to distinguish individuals with stable long-term non-obese status from those who may have recently transitioned from higher BMI categories. Such preceding weight trends may be associated with underlying disease severity or treatment indications and could contribute to residual confounding.
As with all observational studies, residual confounding cannot be eliminated; factors such as socioeconomic status, physical activity, and genetic markers were not fully captured. A limitation is the requirement for ≥2 visits across the study period, which may introduce selection-related bias or potential immortal time bias by excluding patients with limited follow-up. However, because this criterion was applied uniformly to both cohorts before cohort assignment and propensity score matching, any potential bias would be expected to be nondifferential between groups. Outcome definitions relied on diagnostic coding and laboratory data, which introduces the possibility of misclassification, though the use of eGFR-based criteria mitigates some of these concerns. In particular, ESKD was identified using diagnosis and procedure codes rather than registry-based confirmation, and prior studies have shown that the specificity and positive predictive value of billing codes for ESKD may be limited. Misclassification of long-term dialysis status therefore cannot be fully excluded. In addition, urine albumin-to-creatinine ratio (uACR) was available for only a small subset of patients, which may introduce selection bias and limits the interpretability of subgroup analyses involving albuminuria. We also evaluated GLP-1 receptor agonists as a class; differences among individual agents—including pharmacokinetic properties, potency, and cardiovascular profiles—could not be examined. Lastly, medication adherence, dose escalation, and treatment duration were not fully assessed. In addition, our analyses were conducted under an intention-to-treat framework, and we did not perform an as-treated or per-protocol analysis. While our study shows a kidney-protective signal, the calculated E-values were modest (1.36 for MAKE and 1.88 for dialysis), indicating that an unmeasured confounder associated with both treatment selection and outcome by these magnitudes could potentially explain the observed associations. The relatively small E-value for MAKE suggests that the observed effect may be sensitive to residual confounding. Potential unmeasured factors, such as duration of diabetes, smoking status, hypertension severity, socioeconomic status, or medication adherence, may have influenced both treatment selection and outcomes.
The E-value analysis should be interpreted as a test of robustness rather than evidence of causality, and the observed associations may therefore be vulnerable to residual confounding. Similar ranges of E-values have been reported in other large real-world studies of GLP-1 receptor agonists. For example, Radwan et al. reported E-values of 1.21 and 2.73 for mortality outcomes in a nationwide observational cohort [60]. These comparisons indicate that the degree of unmeasured confounding required to negate the observed associations in the present study is comparable to that reported in prior pharmacoepidemiologic analyses. Accordingly, the modest E-values should be regarded as a limitation of the present study, and the results should be interpreted with appropriate caution. In addition, treatment discontinuation and changes in exposure over time in routine clinical practice may have attenuated the observed treatment effects, particularly under an intention-to-treat framework. These limitations underscore the need for mechanistic and prospective interventional trials to clarify the cardiometabolic and renal effects of GLP-1 RAs in non-obese populations.
Conclusion
In this BMI-restricted cohort of adults with type 2 diabetes, GLP-1 receptor agonists were associated with lower risks of major kidney events and dialysis initiation, with a generally consistent direction of benefit across clinical subgroups. Cardiovascular outcomes were neutral, but reductions in hospitalization and sepsis suggest additional systemic advantages. These findings indicate that GLP-1 RAs may provide kidney protection even in leaner patients. Randomized trials in low-BMI populations are warranted.
Supplementary Material
Supplementary methods, figures and tables.
Acknowledgements
This work was supported by the Shin Kong Wu Ho-Su Memorial Hospital sponsored this study (2025SKHADR010).
Data availability statement
The data utilized in this study were obtained from the TriNetX Global Health Research Network and are not publicly available due to licensing agreements and privacy regulations. TriNetX provides access to de-identified, aggregate-level data sourced from a global consortium of healthcare institutions. Researchers interested in accessing the data may submit a request via the TriNetX website (https://trinetx.com) or by contacting Privacy@TriNetX.com. Additionally, data may be made available from the corresponding author upon reasonable request.
Ethics statement
The study was conducted in accordance with the Declaration of Helsinki and approved by the Institutional Review Board of Shin Kong Wu Ho-Su Memorial Hospital (IRB number: 20250907R). The requirement for informed consent was waived due to the use of de-identified retrospective data.
Author contributions
Design: Chien-Lin Lu, Ming-Hsien Tsai; Conduct/Data Collection: Yu-Wei Fang, Yu-Han Tsai, Meng-Ting Chen, Kuo-Cheng Lu, Ming-Hsien Tsai; Analysis: Ming-Hsien Tsai, Chien-Lin Lu, Kuo-Cheng Lu; Writing Manuscript: Chien-Lin Lu, Ming-Hsien Tsai (original draft); Review and Editing: All authors.
Competing Interests
The authors have declared that no competing interest exists.
References
1. Jitraknatee J, Ruengorn C, Nochaiwong S. Prevalence and Risk Factors of Chronic Kidney Disease among Type 2 Diabetes Patients: A Cross-Sectional Study in Primary Care Practice. Scientific Reports. 2020 10
2. Fenta ET, Eshetu HB, Kebede N, Bogale EK, Zewdie A, Kassie TD. et al. Prevalence and predictors of chronic kidney disease among type 2 diabetic patients worldwide, systematic review and meta-analysis. Diabetology & Metabolic Syndrome. 2023 15
3. Shi L-X, Xue Y, Yu X, Wang Y, Hong T, Li X. et al. Prevalence and Risk Factors of Chronic Kidney Disease in Patients with Type 2 Diabetes in China: Cross-Sectional Study. JMIR Public Health and Surveillance. 2024 10
4. Luk A, So W, Kong A, Ozaki R, Ng V, Yu L. et al. Metabolic Syndrome Predicts New Onset of Chronic Kidney Disease in 5,829 Patients with Type 2 Diabetes. Diabetes Care. 2008;31:2357-61
5. Misra A, Sattar N, Ghosh A, Nassar M, Jayawardena R, Gupta R. Type 2 diabetes in South Asians. BMJ. 2025 390
6. Marso SP, Daniels GH, Brown-Frandsen K, Kristensen P, Mann JF, Nauck MA. et al. Liraglutide and Cardiovascular Outcomes in Type 2 Diabetes. N Engl J Med. 2016;375:311-22
7. Marso SP, Bain SC, Consoli A, Eliaschewitz FG, Jódar E, Leiter LA. et al. Semaglutide and Cardiovascular Outcomes in Patients with Type 2 Diabetes. N Engl J Med. 2016;375:1834-44
8. Gerstein HC, Colhoun HM, Dagenais GR, Diaz R, Lakshmanan M, Pais P. et al. Dulaglutide and cardiovascular outcomes in type 2 diabetes (REWIND): a double-blind, randomised placebo-controlled trial. The Lancet. 2019;394:121-30
9. Li X, Song Y, Guo T, Xiao G, Li Q. Effect of Glucagon-like Peptide 1 Receptor Agonists on the Renal Protection in Patients with Type 2 Diabetes: A Systematic Review and Meta-Analysis. Diabetes & metabolism. 2022: 101366.
10. Giugliano D, Scappaticcio L, Longo M, Caruso P, Maiorino M, Bellastella G. et al. GLP-1 receptor agonists and cardiorenal outcomes in type 2 diabetes: an updated meta-analysis of eight CVOTs. Cardiovascular Diabetology. 2021 20
11. Giugliano D, Maiorino M, Bellastella G, Longo M, Chiodini P, Esposito K. GLP-1 receptor agonists for prevention of cardiorenal outcomes in type 2 diabetes: An updated meta-analysis including the REWIND and PIONEER 6 trials. Diabetes. 2019;21:2576-80
12. Kawanami D, Takashi Y. GLP-1 Receptor Agonists in Diabetic Kidney Disease: From Clinical Outcomes to Mechanisms. Frontiers in Pharmacology. 2020 11
13. Toimil R, Ciudin A. GLP-1 Receptor Agonists in Diabetic Kidney Disease: From Physiology to Clinical Outcomes. Journal of Clinical Medicine. 2021 10
14. Yu J, Park S, Lee D, Kim N, Seo J. GLP-1 receptor agonists in diabetic kidney disease: current evidence and future directions. Kidney Research and Clinical Practice. 2022;41:136-49
15. Michos E, Bakris G, Rodbard H, Tuttle K. Glucagon-like peptide-1 receptor agonists in diabetic kidney disease: A review of their kidney and heart protection. American Journal of Preventive Cardiology. 2023 14
16. Chen TH, Hu EH, Chen DY, Lin Y, Chou TS, Lin MS. et al. GLP-1 RAs and Cardiovascular and Kidney Outcomes by Body Mass Index in Type 2 Diabetes. JAMA Netw Open. 2025;8:e2530952
17. Singh S, Garg A, Tantry U, Bliden K, Gurbel P, Gulati M. Safety and efficacy of glucagon-like peptide-1 receptor agonists on cardiovascular events in overweight or obese non-diabetic patients. Current problems in cardiology. 2024: 102403.
18. Kelkar R, Barve N, Kelkar R, Kharel S, Khanapurkar S, Yadav R. Comparison of glucagon-like peptide-1 receptor agonists vs. placebo on any cardiovascular events in overweight or obese non-diabetic patients: a systematic review and meta-analysis. Frontiers in Cardiovascular Medicine. 2024 11
19. Tariq S, Ali MA, Iftikhar HMH, Ali MF, Shah SQA, Perveen F. et al. Long-Term Cardiovascular Outcomes of Glucagon-Like Peptide-1 (GLP-1) Receptor Agonists in Type 2 Diabetes: A Systematic Review. Cureus. 2024 16
20. Zoccali C, Tripepi G. Clinical trial emulation in nephrology. J Nephrol. 2025;38:11-23
21. Inker LA, Eneanya ND, Coresh J, Tighiouart H, Wang D, Sang Y. et al. New Creatinine- and Cystatin C-Based Equations to Estimate GFR without Race. N Engl J Med. 2021;385:1737-49
22. VanderWeele TJ, Ding P. Sensitivity Analysis in Observational Research: Introducing the E-Value. Ann Intern Med. 2017;167:268-74
23. Cherubini JM, MacDonald MJ. Statistical Inferences Using Effect Sizes in Human Endothelial Function Research. Artery Res. 2021;27:176-85
24. Zhang Z, Kim HJ, Lonjon G, Zhu Y. Balance diagnostics after propensity score matching. Ann Transl Med. 2019;7:16
25. Cohen JF, Chalumeau M, Cohen R, Korevaar DA, Khoshnood B, Bossuyt PM. Cochran's Q test was useful to assess heterogeneity in likelihood ratios in studies of diagnostic accuracy. J Clin Epidemiol. 2015;68:299-306
26. Xiaoxuan Liu Z, Ilyas I Little P, Kamato D Sahebka A. et al. GLP-1 receptor agonists (GLP-1RAs): cardiovascular actions and therapeutic potential. International Journal of Biological Sciences. 2021;17:2050-68
27. Tirandi A, Montecucco F, Carbone F, Liberale L. Obesity, cardiovascular and cerebrovascular disease: the role of GLP-1 receptor agonists. Polish archives of internal medicine. 2024
28. Le R, Nguyen M, Allahwala M, Psaltis J, Marathe C, Marathe J. et al. Cardiovascular Protective Properties of GLP-1 Receptor Agonists: More than Just Diabetic and Weight Loss Drugs. Journal of Clinical Medicine. 2024 13
29. Szekeres Z, Nagy A, Jahner K, Szabados E. Impact of Selected Glucagon-like Peptide-1 Receptor Agonists on Serum Lipids, Adipose Tissue, and Muscle Metabolism—A Narrative Review. International Journal of Molecular Sciences. 2024 25
30. Qassab MA, Mneimneh M, Jradi A, Derbas B, Dabboussi D, Baini JK. et al. The Expanding Role of GLP-1 Receptor Agonists: Advancing Clinical Outcomes in Metabolic and Mental Health. Current Issues in Molecular Biology. 2025 47
31. Mann J, Buse J, Idorn T, Leiter L, Pratley R, Rasmussen S. et al. Potential kidney protection with liraglutide and semaglutide: Exploratory mediation analysis. Diabetes, Obesity & Metabolism. 2021;23:2058-66
32. Shaman A, Bain S, Bakris G, Buse J, Idorn T, Mahaffey K. et al. Effect of the Glucagon-Like Peptide-1 Receptor Agonists Semaglutide and Liraglutide on Kidney Outcomes in Patients with Type 2 Diabetes: Pooled Analysis of SUSTAIN 6 and LEADER. Circulation. 2021;145:575-85
33. Kristensen S, Rørth R, Jhund P, Docherty K, Sattar N, Preiss D. et al. Cardiovascular, mortality, and kidney outcomes with GLP-1 receptor agonists in patients with type 2 diabetes: a systematic review and meta-analysis of cardiovascular outcome trials. The lancet Diabetes & endocrinology. 2019
34. Gerstein H, Colhoun H, Dagenais G, Diaz R, Lakshmanan M, Pais P. et al. Dulaglutide and renal outcomes in type 2 diabetes: an exploratory analysis of the REWIND randomised, placebo-controlled trial. Lancet. 2019
35. Verma S, Bain S, Fries TM, Mazer C, Nauck M, Pratley R. et al. Duration of diabetes and cardiorenal efficacy of liraglutide and semaglutide: A post hoc analysis of the LEADER and SUSTAIN 6 clinical trials. Diabetes, Obesity & Metabolism. 2019;21:1745-51
36. Verma S, McGuire D, Bain S, Bhatt D, Leiter L, Mazer C. et al. Effects of glucagon-like peptide-1 receptor agonists liraglutide and semaglutide on cardiovascular and renal outcomes across body mass index categories in type 2 diabetes: Results of the LEADER and SUSTAIN 6 trials. Diabetes, Obesity & Metabolism. 2020;22:2487-92
37. Tuttle K, Bosch-Traberg H, Cherney D, Hadjadj S, Lawson J, Mosenzon O. et al. Post hoc analysis of SUSTAIN 6 and PIONEER 6 trials suggests that people with type 2 diabetes at high cardiovascular risk treated with semaglutide experience more stable kidney function compared to placebo. Kidney international. 2023
38. Apperloo E, Cherney D, Kuhlman A, Mann J, Rasmussen S, Rossing P. et al. Effect of semaglutide on kidney function across different levels of baseline HbA1c, blood pressure, body weight and albuminuria in SUSTAIN 6 and PIONEER 6. Nephrology Dialysis Transplantation. 2024;40:352-9
39. Crajoinas R, Oricchio F, Pessoa T, Pacheco B, Lessa L, Malnic G. et al. Mechanisms mediating the diuretic and natriuretic actions of the incretin hormone glucagon-like peptide-1. American journal of physiology Renal physiology. 2011 301 2
40. Greco E, Russo G, Giandalia A, Viazzi F, Pontremoli R, De Cosmo S. GLP-1 Receptor Agonists and Kidney Protection. Medicina. 2019 55
41. Vitale M, Haxhi J, Cirrito T, Pugliese G. Renal protection with glucagon-like peptide-1 receptor agonists. Current opinion in pharmacology. 2020;54:91-101
42. Sourris K, Ding Y, Maxwell S, Al-Sharea A, Kantharidis P, Mohan M. et al. Glucagon-like peptide-1 receptor signaling modifies the extent of diabetic kidney disease through dampening the receptor for advanced glycation end products-induced inflammation. Kidney international. 2023
43. Tian S, Zhou S, Wu W, Lin Y, Wang T, Sun H. et al. GLP-1 Receptor Agonists Alleviate Diabetic Kidney Injury via β-Klotho-Mediated Ferroptosis Inhibition. Advanced Science. 2024 12
44. Parab P, Chaudhary P, Mukhtar S, Moradi A, Kodali A, Okoye C. et al. Role of Glucagon-Like Peptide-1 (GLP-1) Receptor Agonists in Cardiovascular Risk Management in Patients with Type 2 Diabetes Mellitus: A Systematic Review. Cureus. 2023 15
45. Marx N, Husain M, Lehrke M, Verma S, Sattar N. GLP-1 Receptor Agonists for the Reduction of Atherosclerotic Cardiovascular Risk in Patients with Type 2 Diabetes. Circulation. 2022 146 24: 1882-94
46. Marsico F, Paolillo S, Gargiulo P, Bruzzese D, Dell'Aversana S, Esposito I. et al. Effects of glucagon-like peptide-1 receptor agonists on major cardiovascular events in patients with Type 2 diabetes mellitus with or without established cardiovascular disease: a meta-analysis of randomized controlled trials. European heart journal. 2020
47. Bednarz K, Kowalczyk K, Cwynar M, Czapla D, Czarkowski W, Kmita D. et al. The Role of Glp-1 Receptor Agonists in Insulin Resistance with Concomitant Obesity Treatment in Polycystic Ovary Syndrome. International Journal of Molecular Sciences. 2022 23
48. Helmstädter J, Keppeler K, Küster L, Münzel T, Daiber A, Steven S. Glucagon-like peptide-1 (GLP-1) receptor agonists and their cardiovascular benefits—The role of the GLP-1 receptor. British Journal of Pharmacology. 2021;179:659-76
49. Zhang X, Cao C, Zheng F, Liu C, Tian X. Therapeutic Potential of GLP-1 Receptor Agonists in Diabetes and Cardiovascular Disease: Mechanisms and Clinical Implications. Cardiovascular drugs and therapy. 2025
50. Lincoff A, Brown-Frandsen K, Colhoun H, Deanfield J, Emerson S, Esbjerg S. et al. Semaglutide and Cardiovascular Outcomes in Obesity without Diabetes. The New England journal of medicine. 2023
51. Deanfield J, Verma S, Scirica B, Kahn S, Emerson S, Ryan D. et al. Semaglutide and cardiovascular outcomes in patients with obesity and prevalent heart failure: a prespecified analysis of the SELECT trial. The Lancet. 2024;404:773-86
52. Kosiborod M, Petrie M, Borlaug B, Butler J, Davies M, Hovingh G. et al. Semaglutide in Patients with Obesity-Related Heart Failure and Type 2 Diabetes. The New England journal of medicine. 2024
53. Alharbi S. Anti-inflammatory role of glucagon-like peptide 1 receptor agonists and its clinical implications. Therapeutic Advances in Endocrinology and Metabolism. 2024 15
54. Mehdi S, Pusapati S, Anwar MS, Lohana D, Kumar P, Nandula S. et al. Glucagon-like peptide-1: a multi-faceted anti-inflammatory agent. Frontiers in Immunology. 2023 14
55. Sun H, Hao Y, Liu H, Gao F. The immunomodulatory effects of GLP-1 receptor agonists in neurogenerative diseases and ischemic stroke treatment. Frontiers in Immunology. 2025 16
56. Migliorisi G, Gabbiadini R, Buono D, Ferraris M, Privitera G, Petronio L. et al. GLP-1 receptor agonists in IBD: exploring the crossroads of metabolism and inflammation. Frontiers in Immunology. 2025 16
57. Ren Y, Chen Y, Zheng W, Kong W, Liao Y, Zhang J. et al. The effect of GLP-1 receptor agonists on circulating inflammatory markers in type 2 diabetes patients: A systematic review and meta-analysis. Diabetes. 2025;27:3607-26
58. Zannad F, Cannon C, Cushman W, Bakris G, Menon V, Perez A. et al. Heart failure and mortality outcomes in patients with type 2 diabetes taking alogliptin versus placebo in EXAMINE: a multicentre, randomised, double-blind trial. The Lancet. 2015;385:2067-76
59. McGuire D, Van De Werf F, Armstrong P, Standl E, Koglin J, Green J. et al. Association Between Sitagliptin Use and Heart Failure Hospitalization and Related Outcomes in Type 2 Diabetes Mellitus: Secondary Analysis of a Randomized Clinical Trial. JAMA cardiology. 2016 1 2: 126-35
60. Radwan RM, Lu Y, Dai H, George TJ, Guo Y, Guo J. et al. GLP-1 RA Use and Survival Among Older Adults with Cancer and Type 2 Diabetes. JAMA Netw Open. 2025;8:e2521887
Author contact
Corresponding author: Chien-Lin Lu, MD, PhD, Fu Jen Catholic University, New Taipei City, Taiwan, 510 Zhongzheng Rd., Xinzhuang Dist., New Taipei City 24205, Taiwan. Email: 096195fju.edu.tw.

 Global reach, higher impact
Global reach, higher impact