Impact Factor ISSN: 1449-1907
Int J Med Sci 2026; 23(4):1519-1534. doi:10.7150/ijms.125059 This issue Cite
Research Paper
Magnesium Oxide Use and Clinical Outcomes in CKD Patients: Evidence from a Nationwide Population-Based Cohort Study in Taiwan
1. Division of Nephrology, Department of Internal Medicine, Tri-Service General Hospital, National Defense Medical University, Taipei, Taiwan.
2. Division of Nephrology, Department of Internal Medicine, Taoyuan Armed Forces General Hospital, Taoyuan, Taiwan.
3. Department of Life Sciences, National Central University, Taoyuan, Taiwan.
4. Division of Biochemistry, Department of Chemistry, Hofstra University, Hempstead, New York, USA.
5. Division of Cardiovascular Medicine, Taoyuan Armed Forces General Hospital, Taoyuan, Taiwan.
6. Cardiovascular Division, Tri-service General Hospital, National Defense Medical University, Taipei, Taiwan.
7. School of Medicine, National Tsing Hua University, Hsinchu, Taiwan.
8. Institute of Bioinformatics and Structural Biology, National Tsing Hua University, Hsinchu, Taiwan.
9. Division of Nephrology, Department of Internal Medicine, School of Medicine, College of Medicine, Taipei Medical University, Taipei, Taiwan.
10. Division of Nephrology, Department of Internal Medicine, Landseed International Hospital, Taoyuan City, Taiwan.
Received 2025-9-11; Accepted 2026-2-3; Published 2026-3-17
Abstract
Background: Magnesium homeostasis in chronic kidney disease (CKD) is complex, and serum magnesium concentrations reflect only approximately 1% of total body magnesium. Both magnesium deficiency (hypomagnesemia) and excess (hypermagnesemia) have been linked to adverse cardiovascular outcomes, a concern that is particularly relevant in patients with CKD. Magnesium oxide (MgO) is frequently prescribed for dyspepsia and constipation in clinical practice; however, its clinical impact in CKD patients remains uncertain and warrants further investigation.
Materials and methods: We included non-dialysis CKD patients identified from the Taipei Medical University Clinical Research Database (TMUCRD) between 1998 and 2021. Adherence to MgO was assessed using the medication possession ratio (MPR). The primary outcomes were acute kidney injury (AKI), acute kidney disease (AKD), hospitalization for AKI, end-stage renal disease (ESRD) requiring dialysis, congestive heart failure with pulmonary edema, cardiac arrhythmia, and acute myocardial infarction. Baseline comorbidities assessed prior to the index date included hypertension, diabetes, hyperlipidemia, ischemic heart disease (IHD), ischemic stroke, congestive heart failure, atrial fibrillation (AF), peripheral arterial disease (PAD), chronic obstructive pulmonary disease (COPD), chronic liver disease (CLD), and dementia. These variables, along with relevant medications, were included as covariates in multivariable models to adjust for potential confounders.
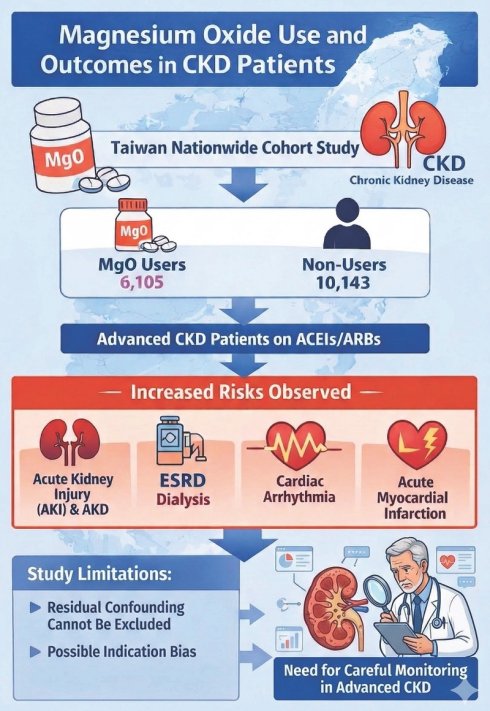
Results: Before matching, 6,105 MgO users and 10,143 non-users were identified; approximately 73% of MgO users had MPR <40%. In the ACE inhibitor (ACEI)/angiotensin receptor blocker (ARB) and pre-end-stage renal disease (pre-ESRD) program cohort, 207 MgO users and 1,401 non-users were included; after matching, 151 MgO users and 302 non-users remained. Dementia was more prevalent among MgO users, whereas diabetes was more common in non-users. MgO use was associated with higher risks of AKI, AKD, ESRD requiring dialysis, cardiac arrhythmia, and myocardial infarction in both unmatched and matched cohorts. In matched CKD patients, adjusted hazard ratios (aHRs) were 37.0 for AKI, 6.26 for AKD, 3.13 for ESRD, 2.06 for cardiac arrhythmia, and 1.86 for acute myocardial infarction. In the matched ACEI/ARB and pre-ESRD cohort, MgO users also demonstrated higher risks of AKI (aHR = 16.1) and AKD (aHR = 2.79). Cumulative incidence analyses consistently showed worse outcomes among MgO users. Among MgO users, advancing CKD stage was associated with progressively higher risks of adverse outcomes, particularly in stages 4-5. Both unmatched and matched analyses demonstrated a dose-response pattern, with the highest hazards observed for dialysis progression and cardiac arrhythmia.
Conclusion: In this large cohort study, MgO use in CKD patients was associated with increased risks of AKI, AKD, ESRD, arrhythmia, and myocardial infarction. The magnitude of risk appeared greater in advanced CKD stages. These findings highlight the importance of careful risk-benefit assessment and close clinical monitoring when prescribing MgO in this high-risk population.
Keywords: acute kidney injury, acute kidney disease, chronic kidney disease, end-stage renal disease, cardiac arrhythmia, myocardial infarction, magnesium oxide
Introduction
The kidneys play a central role in regulating magnesium balance by adjusting urinary excretion according to systemic magnesium status, ranging from 0.5% to 70% of filtered magnesium. Approximately 80-90% of magnesium reabsorption occurs through passive, paracellular pathways in the proximal tubule and thick ascending limb of Henle's loop, whereas only 5-10% is actively reabsorbed in the distal convoluted tubule. Magnesium homeostasis in chronic kidney disease (CKD) is complex. With progressive loss of renal function, as seen in advanced CKD and dialysis-dependent patients, there is an increased risk of magnesium accumulation and toxicity [1-3]. Conversely, conditions such as malnutrition, concurrent diuretic or proton pump inhibitor use, and dialysis with low-magnesium dialysate may predispose patients to hypomagnesemia [4-8]. Magnesium deficiency in CKD has been associated with higher parathyroid hormone levels, elevated systolic blood pressure, and increased cardiovascular risk. Kanbay M, et al. reported that CKD patients with serum magnesium levels below 2.05 mg/dL had significantly higher cardiovascular mortality [8]. Importantly, the clinical scenarios of hypomagnesemia may contribute to endothelial dysfunction, vascular stiffness, arteriosclerosis, and adverse cardiovascular events, which are the leading causes of mortality in the CKD population [9-14].
There is currently no definitive method for assessing magnesium status. In clinical practice, a combination of dietary history, serum magnesium, and urinary magnesium excretion is often used, although each approach has important limitations [15]. Given its diverse physiological functions, magnesium is essential in the prevention and management of numerous diseases. Hypomagnesemia has been linked to several chronic conditions, including Alzheimer's disease, insulin resistance and type 2 diabetes mellitus, hypertension, cardiovascular disease (such as stroke), migraine, and attention deficit hyperactivity disorder (ADHD) [16]. Dietary assessment, whether through food diaries or recall of magnesium-rich food intake, is inherently inconsistent due to its retrospective design, regional variability in food processing, and temporal changes in nutrient composition. Regular updates to food composition databases are therefore essential for accurate estimation of dietary magnesium intake [16].
Magnesium oxide (MgO) is widely prescribed in clinical practice for the management of constipation, dyspepsia, and occasionally hypophosphatemia due to its laxative and antacid properties. The use of MgO as a laxative is particularly prevalent in Taiwan and Japan, whereas in many Western and other international settings, osmotic agents such as polyethylene glycol or lactulose are more commonly employed as first-line therapies for constipation [17]. In patients with CKD, magnesium metabolism is often dysregulated, and both deficiency and excess can have important clinical implications. While MgO is commonly used in clinical practice, particularly for gastrointestinal indications and hypomagnesemia, its effects on magnesium balance and the broader impact on renal and cardiovascular outcomes in patients with advanced stages of CKD remain uncertain. Serum magnesium levels are influenced by multiple factors, including renal clearance, dietary intake, concomitant medications, and comorbidities, making interpretation in CKD particularly complex [13-16]. Although dysmagnesemia has been linked to increased risks of vascular calcification, cardiovascular events, and mortality, direct evidence regarding the impact of MgO use on clinical outcomes in CKD patients remains limited. Clarifying this relationship is crucial for optimizing treatment strategies and reducing potential complications in this high-risk population.
Materials and methods
Data source
We used the Taipei Medical University Clinical Research Database (TMUCRD) for this study. The TMUCRD contains electronic medical records of 4.1 million patients from three affiliated hospitals, which are Taipei Medical University Hospital, Wanfang Hospital, and Shuang Ho Hospital from 1998 to 2021. All basic information, cause of death, medical information, cancer registry, diagnosis, treatment procedure and laboratory test result of the participants were available for analysis.
Study design
In this study, the exposure group were those using magnesium oxide (MgO) and the control group were those never used MgO. The index date was defined as the index date of magnesium oxide use. We included the CKD patients not receiving dialysis in this study. A random date after the diagnosis of CKD was assigned to the control group as the index date. MgO users and non-users were matched according to sex, age, comorbidities, and medications by propensity score matching in 1:1 ratio. Patients who were younger than 30 years, and whom without a minimum of 2 years of data available following were excluded. We further investigated the cohort of CKD patients with receiving ACE inhibitors (ACEIs)/angiotensin receptor blockers (ARBs) and pre-end-stage renal disease (pre-ESRD) program. 1:2 propensity score matching was applied to obtain the matched cohort.
Patients were eligible for the program if they met the following criteria: the primary outcomes were AKI, AKD, ESRD with dialysis, cardiac arrhythmias, congestive heart failure with acute pulmonary edema, and acute myocardial infarction. Furthermore, in this study, the primary endpoints, which represented AKI-AKD-ESRD in the progression of dialysis, were those defined in the ADQI [18] and KDIGO [19] workshops. AKI was defined as an abrupt decrease in kidney function that occurred within 7 days or less after the index date and was divided into stages 0, 1, 2, and 3 multiplied by the serum creatinine level. AKD was described as acute or subacute damage and loss of kidney function for a duration of 7-90 days after exposure to an AKI episode and was divided into stages 0, 1, 2, and 3 multiplied by serum creatinine levels. All the serum creatinine levels included in the analysis were chosen on the basis of the respective highest values obtained within 0-7 and 7-90 days for AKI and AKD, respectively. ESRD with dialysis was defined as an order code by the National Health Insurance. In addition, multidisciplinary medical teams, including nephrologists, health education nurses, and nutritionists, provided comprehensive medical assessments, laboratory examinations, and patient education every three months. Those enrolled in the program were also cared for according to applicable clinical guidelines at different stages of CKD [20]. The date of initial enrolment in the program was treated as the index date.
Main outcome and covariates
The primary outcomes were acute kidney injury (AKI), acute kidney disease (AKD), hospitalization for AKI, ESRD requiring dialysis, congestive heart failure with pulmonary edema, cardiac arrhythmia, and acute myocardial infarction. Serum variables including creatinine, estimated glomerular filtration rate (eGFR), hemoglobin, and albumin were included as adjustment covariates. Baseline comorbidities assessed prior to the index date included hypertension, diabetes, hyperlipidemia, ischemic heart disease (IHD), ischemic stroke, congestive heart failure, atrial fibrillation (AF), peripheral arterial disease (PAD), chronic obstructive pulmonary disease (COPD), chronic lung disease and dementia. Concomitant medications considered were antiplatelets, warfarin, rivaroxaban, angiotensin-converting enzyme inhibitors/angiotensin receptor blockers (ACEI/ARB), β-blockers, calcium channel blockers (CCBs), statins, and antidiabetic agents, including metformin, thiazolidinediones, sulfonylureas, alpha-glucosidase inhibitors (AGIs), dipeptidyl peptidase-4 inhibitors (DPP4), and insulin. Patients were classified as MgO users if they had a pharmacy claim for MgO in combination with ACEIs/ARBs within three months of study initiation. Medication adherence was assessed using the medication possession ratio (MPR), calculated as the proportion of days covered by dispensed prescriptions during a specified interval. Participants who discontinued therapy for over 90 days during follow-up were censored and excluded from further analysis. MPR was categorized as <40% (poor adherence) and ≥80% (high adherence). An 80% threshold is commonly considered clinically meaningful, reflecting consistent medication availability with minimal treatment gaps [21, 22].
Statistical analysis
We summarized the number and percentage for categorical variables, and the mean and standard deviation for continuous variables. The chi-square test and Student's T-test were applied to assess the difference between two groups. The univariable and multivariable Cox proportional hazard model was used to obtain the risk of outcome. The cumulative incidence curves were plotted by the Kaplan-Meier method and tested by the log-rank test. All analyses were conducted using SAS software, version 9.4 (SAS Institute Inc., Cary, NC). A significant level was p-value less than 0.05. The incidence per 100 person-years of outcomes was calculated via Kaplan‒Meier estimation. A Cox proportional hazards regression model was used to evaluate the hazard ratio (HR) for risk outcomes associated with MgO, after controlling for demographic and clinical factors [23]. Patients who died, were lost to follow-up, or were discharged before the event of interest occurred during the follow-up period were censored.
Results
Baseline characteristics of CKD patients
The patient selection process is shown in detail in Figure 1. Table 1a shows the baseline characteristics of all CKD patients' cohort. Before the matching, 6,105 MgO users and 10,143 MgO non-users were included. More male and younger patients were in the MgO group. The value of creatinine, eGFR and albumin in MgO users were lower than that of the MgO non-user. The distribution of comorbidities and medication between MgO users and non-users were different. After the propensity score matching, 6,000 MgO users and non-user were enrolled. Fifty percent for men and women. Most patients were in 60-70 years old. There were difference of creatinine and eGFR between two groups. Besides COPD, the proportions of comorbidities among two group of patients were similar. MgO users were more likely to take rivaroxaban, β-blocker, CCB, statin, metformin, DPP4 and insulin. About 73 % of MgO group with medication possession ratio (MPR) lower than 40%. Table 1b is the baseline characteristics of CKD patients receiving ACEI/ARB and Pre-ESRD program cohort. In the total of 1,608 patients, 1,401 was MgO non-user and 207 was MgO user. Male patients present more in MgO user than that in MgO non-user. Creatinine value of MgO user was higher. More patients with history of diabetes and hyperlipidemia and medication of statin, metformin and sulfonylureas in the control group than in the MgO group. But, MgO group had more patients with dementia. The matching cohort was consisting of 151 MgO users and 302 non-users. The mean of creatinine and albumin was lower in MgO user than the non-user. There were more patients with hypertension and congestive heart failure, but less patients with ischemic stroke in the control group. The percentage of MgO user with <40%, 40-80% and 80% were 77.48%, 12.58% and 9.93%, respectively.
The study flow chart.
Baseline characteristics of CKD patients.
| Without matching | 1:1 PS matching | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Control | Magnesium oxide | Control | Magnesium oxide | |||||||
| N=10143 | N=6105 | N=6000 | N=6000 | |||||||
| Variables | n | % | n | % | p-value | n | % | n | % | p-value |
| Sex | 0.010 | 0.559 | ||||||||
| Female | 4809 | 47.41 | 3022 | 49.50 | 3005 | 50.08 | 2973 | 49.55 | ||
| Male | 5334 | 52.59 | 3083 | 50.50 | 2995 | 49.92 | 3027 | 50.45 | ||
| Age, year | <0.001 | 0.006 | ||||||||
| 30-70 | 5954 | 58.70 | 3108 | 50.91 | 3213 | 53.55 | 3062 | 51.03 | ||
| >70 | 4189 | 41.30 | 2997 | 49.09 | 2787 | 46.45 | 2938 | 48.97 | ||
| Mean (SD) | 66.74 | (14.22) | 68.85 | (13.29) | <0.001 | 68.74 | (13.77) | 68.8 | (13.27) | 0.796 |
| Serum data, mean (SD) | ||||||||||
| Mg (mg/Dl) | 2.21 | (0.42) | 2.08 | (0.43) | 0.005 | 2.23 | (0.39) | 2.08 | (0.44) | 0.004 |
| Creatinine (mg/dL) | 1.47 | (1.92) | 1.31 | (1.43) | <0.001 | 1.18 | (1.27) | 1.11 | (1.04) | 0.018 |
| eGFR (mL/min/1.73m2) | 77.52 | (43.74) | 74.14 | (48.97) | 0.011 | 69.88 | (34.31) | 64.16 | (32.36) | <0.001 |
| Hemoglobin (g/dL) | 10.81 | (1.83) | 10.87 | (1.94) | 0.874 | 10.99 | (1.84) | 10.91 | (1.94) | 0.852 |
| Albumin (g/dL) | 3.71 | (0.62) | 3.52 | (0.60) | 0.016 | 3.68 | (0.67) | 3.53 | (0.61) | 0.160 |
| Comorbidities | ||||||||||
| Hypertension | 4590 | 45.25 | 2907 | 47.62 | 0.003 | 2847 | 47.45 | 2854 | 47.57 | 0.898 |
| Diabetes | 5141 | 50.69 | 2949 | 48.30 | 0.003 | 2865 | 47.75 | 2897 | 48.28 | 0.559 |
| Hyperlipidemia | 2894 | 28.53 | 1895 | 31.04 | 0.001 | 1874 | 31.23 | 1865 | 31.08 | 0.859 |
| IHD | 3470 | 34.21 | 1713 | 28.06 | <0.001 | 1652 | 27.53 | 1685 | 28.08 | 0.501 |
| Ischemic stroke | 125 | 1.23 | 153 | 2.51 | <0.001 | 112 | 1.87 | 143 | 2.38 | 0.050 |
| Congestive heart failure | 1049 | 10.34 | 509 | 8.34 | <0.001 | 505 | 8.42 | 502 | 8.37 | 0.921 |
| AF | 129 | 1.27 | 113 | 1.85 | 0.003 | 100 | 1.67 | 111 | 1.85 | 0.445 |
| PAD | 219 | 2.16 | 103 | 1.69 | 0.037 | 101 | 1.68 | 99 | 1.65 | 0.887 |
| COPD | 880 | 8.68 | 850 | 13.92 | <0.001 | 730 | 12.17 | 818 | 13.63 | 0.017 |
| CLD | 879 | 8.67 | 666 | 10.91 | <0.001 | 646 | 10.77 | 652 | 10.87 | 0.860 |
| Dementia | 381 | 3.76 | 350 | 5.73 | <0.001 | 307 | 5.12 | 337 | 5.62 | 0.224 |
| Medication | ||||||||||
| Antiplatelet | 4859 | 47.91 | 2946 | 48.26 | 0.665 | 2836 | 47.27 | 2888 | 48.13 | 0.342 |
| Warfarin | 379 | 3.74 | 215 | 3.52 | 0.480 | 216 | 3.60 | 214 | 3.57 | 0.922 |
| Rivaroxaban | 33 | 0.33 | 51 | 0.84 | <0.001 | 28 | 0.47 | 50 | 0.83 | 0.012 |
| ACEI/ARB | 4159 | 41.00 | 2660 | 43.57 | 0.001 | 2525 | 42.08 | 2609 | 43.48 | 0.121 |
| Beta-2 blocker | 3902 | 38.47 | 2524 | 41.34 | <0.001 | 2306 | 38.43 | 2474 | 41.23 | 0.002 |
| CCB | 3857 | 38.03 | 2801 | 45.88 | <0.001 | 2371 | 39.52 | 2731 | 45.52 | <0.001 |
| Statin | 3208 | 31.63 | 2109 | 34.55 | <0.001 | 1936 | 32.27 | 2074 | 34.57 | 0.008 |
| Metformin | 3253 | 32.07 | 2268 | 37.15 | <0.001 | 1929 | 32.15 | 2225 | 37.08 | 0.000 |
| Thiazolidinedione | 648 | 6.39 | 371 | 6.08 | 0.427 | 378 | 6.30 | 367 | 6.12 | 0.677 |
| Sulfonylureas | 2788 | 27.49 | 1766 | 28.93 | 0.048 | 1667 | 27.78 | 1729 | 28.82 | 0.209 |
| AGIs | 1074 | 10.59 | 681 | 11.15 | 0.260 | 637 | 10.62 | 669 | 11.15 | 0.348 |
| DPP4 | 1315 | 12.96 | 1090 | 17.85 | <0.001 | 811 | 13.52 | 1066 | 17.77 | <0.001 |
| Insulin | 2356 | 23.23 | 2201 | 36.05 | <0.001 | 1448 | 24.13 | 2146 | 35.77 | <0.001 |
| PPI | 1474 | 14.53 | 1393 | 22.82 | <0.001 | 951 | 15.85 | 1393 | 22.82 | <0.001 |
| Thiazide | 875 | 8.63 | 640 | 10.48 | <0.001 | 544 | 9.07 | 640 | 10.48 | 0.0137 |
| Loop diuretic | 1730 | 17.06 | 1339 | 21.93 | <0.001 | 1070 | 17.83 | 1339 | 21.93 | <0.001 |
| MPR | ||||||||||
| <40% | 4426 | 72.50 | 4354 | 72.57 | ||||||
| 40-80% | 827 | 13.55 | 809 | 13.48 | ||||||
| >80% | 852 | 13.96 | 837 | 13.95 | ||||||
| CKD stage | <0.001 | <0.001 | ||||||||
| 1 | 521 | 5.14 | 549 | 8.99 | 324 | 5.40 | 527 | 8.78 | ||
| 2 | 587 | 5.79 | 934 | 15.3 | 403 | 6.72 | 912 | 15.2 | ||
| 3 | 374 | 3.69 | 992 | 16.25 | 260 | 4.33 | 965 | 16.08 | ||
| 4 | 128 | 1.26 | 243 | 3.98 | 91 | 1.52 | 240 | 4.00 | ||
| 5 | 8533 | 84.13 | 3387 | 55.48 | 4922 | 82.03 | 3356 | 55.93 | ||
IHD: ischemic heart disease; AF: Atrial fibrillation; PAD: peripheral artery disease; COPD: chronic obstructive pulmonary disease; CLD: chronic liver disease; ACEI: angiotensin-converting enzyme inhibitor; ARB: angiotensin II receptor blocker; CCB: calcium channel blockers; AGIs: alpha-glucosidase inhibitors; DPP4: dipeptidyl peptidase 4; PPI: proton-pump inhibitor; MPR: medication possession ratio.
Baseline characteristics of study for CKD patients with receiving ACEI/ARB and Pre-ESRD program.
| Without matching | 1:1 PS matching | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Control | Magnesium oxide | Control | Magnesium oxide | ||||||||
| N=1401 | N=207 | N=302 | N=151 | ||||||||
| Variables | n | % | n | % | p-value | n | % | n | % | p-value | |
| Sex | 0.007 | 0.842 | |||||||||
| Female | 742 | 52.96 | 89 | 43.00 | 161 | 53.31 | 79 | 52.32 | |||
| Male | 659 | 47.04 | 118 | 57.01 | 141 | 46.69 | 72 | 47.68 | |||
| Age | 0.521 | 0.466 | |||||||||
| 30-80 | 985 | 70.31 | 141 | 68.12 | 265 | 87.75 | 136 | 90.07 | |||
| >80 | 416 | 29.69 | 66 | 31.88 | 37 | 12.25 | 15 | 9.93 | |||
| Mean (SD) | 72.5 | (11.83) | 73.05 | (11.97) | 0.532 | 66.37 | (11.45) | 64.97 | (11.81) | 0.224 | |
| Comorbidities | |||||||||||
| Hypertension | 1162 | 82.94 | 170 | 82.13 | 0.772 | 246 | 81.46 | 109 | 72.19 | 0.024 | |
| Diabetes | 1089 | 77.73 | 135 | 65.22 | <0.001 | 237 | 78.48 | 118 | 78.15 | 0.936 | |
| Hyperlipidemia | 694 | 49.54 | 75 | 36.23 | <0.001 | 148 | 49.01 | 60 | 39.74 | 0.062 | |
| IHD | 744 | 53.11 | 98 | 47.34 | 0.121 | 148 | 49.01 | 70 | 46.36 | 0.595 | |
| Ischemic stroke | 54 | 3.85 | 6 | 2.90 | 0.498 | 6 | 1.99 | 14 | 9.27 | <0.001 | |
| Congestive heart failure | 256 | 18.27 | 42 | 20.29 | 0.486 | 63 | 20.86 | 15 | 9.93 | 0.004 | |
| AF | 68 | 4.85 | 6 | 2.90 | 0.210 | 8 | 2.65 | 2 | 1.33 | 0.366 | |
| PAD | 51 | 3.64 | 6 | 2.90 | 0.590 | 7 | 2.318 | 5 | 3.311 | 0.535 | |
| COPD | 240 | 17.13 | 41 | 19.81 | 0.344 | 44 | 14.57 | 27 | 17.88 | 0.361 | |
| CLD | 153 | 10.92 | 29 | 14.01 | 0.190 | 45 | 14.90 | 14 | 9.27 | 0.093 | |
| Dementia | 124 | 8.85 | 31 | 14.98 | 0.005 | 10 | 3.31 | 12 | 7.95 | 0.030 | |
| Medication | |||||||||||
| Antiplatelet | 1140 | 81.37 | 158 | 76.33 | 0.086 | 233 | 77.15 | 106 | 70.20 | 0.108 | |
| Warfarin | 1140 | 81.37 | 158 | 76.33 | 0.086 | 233 | 77.15 | 106 | 70.20 | 0.108 | |
| Rivaroxaban | 43 | 3.07 | 4 | 1.93 | 0.365 | 8 | 2.65 | 6 | 3.97 | 0.443 | |
| ACEI/ARB | 1387 | 100.00 | 191 | 100.00 | 300 | 100.00 | 142 | 100.00 | |||
| Beta-2 blocker | 1032 | 73.66 | 140 | 67.63 | 0.069 | 228 | 75.50 | 107 | 70.86 | 0.289 | |
| CCB | 1136 | 81.09 | 176 | 85.02 | 0.172 | 234 | 77.48 | 116 | 76.82 | 0.874 | |
| Statin | 940 | 67.10 | 115 | 55.56 | 0.001 | 194 | 64.24 | 89 | 58.94 | 0.272 | |
| Metformin | 874 | 62.38 | 86 | 41.55 | <0.001 | 189 | 62.58 | 104 | 68.87 | 0.187 | |
| Thiazolidinedione | 180 | 12.85 | 19 | 9.18 | 0.135 | 38 | 12.58 | 14 | 9.27 | 0.297 | |
| Sulfonylureas | 743 | 53.03 | 86 | 41.55 | 0.002 | 153 | 50.66 | 77 | 50.99 | 0.947 | |
| AGIs | 327 | 23.34 | 55 | 26.57 | 0.308 | 56 | 18.543 | 26 | 17.22 | 0.730 | |
| DPP4 | 327 | 23.34 | 55 | 26.57 | 0.308 | 56 | 18.543 | 26 | 17.22 | 0.730 | |
| Insulin | 327 | 23.34 | 55 | 26.57 | 0.308 | 56 | 18.543 | 26 | 17.22 | 0.730 | |
| PPI | 440 | 31.41 | 90 | 43.48 | 0.001 | 99 | 32.78 | 61 | 40.40 | 0.110 | |
| Thiazide | 362 | 25.84 | 62 | 29.95 | 0.210 | 66 | 21.85 | 35 | 23.18 | 0.750 | |
| Loop diuretic | 616 | 43.97 | 137 | 66.18 | <0.001 | 137 | 45.36 | 68 | 45.03 | 0.947 | |
| MPR | |||||||||||
| <40% | 149 | 71.98 | 117 | 77.48 | |||||||
| 40-80% | 25 | 12.08 | 19 | 12.58 | |||||||
| >80% | 33 | 15.94 | 15 | 9.93 | |||||||
| CKD stage | <0.001 | <0.001 | |||||||||
| 1 | 14 | 1.00 | 0 | 0.00 | 6 | 1.99 | 1 | 0.66 | |||
| 2 | 115 | 8.21 | 4 | 1.93 | 38 | 12.58 | 12 | 7.95 | |||
| 3 | 731 | 52.18 | 85 | 41.06 | 128 | 42.38 | 103 | 68.21 | |||
| 4 | 129 | 9.21 | 52 | 25.12 | 27 | 8.94 | 17 | 11.26 | |||
| 5 | 412 | 29.41 | 66 | 31.88 | 103 | 34.11 | 18 | 11.92 | |||
IHD: ischemic heart disease; AF: Atrial fibrillation; PAD: peripheral artery disease; COPD: chronic obstructive pulmonary disease; CLD: chronic liver disease; ACEI: angiotensin-converting enzyme inhibitor; ARB: angiotensin II receptor blocker; CCB: calcium channel blockers; AGIs: alpha-glucosidase inhibitors; DPP4: dipeptidyl peptidase 4; PPI: proton-pump inhibitor; MPR: medication possession ratio.
Clinical outcomes for CKD patients
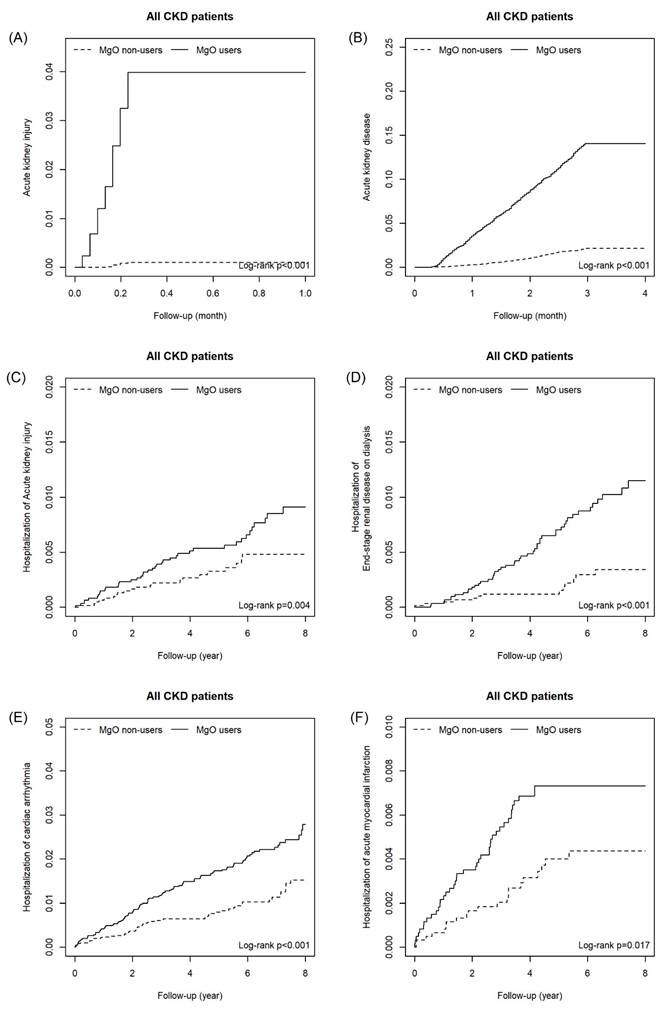
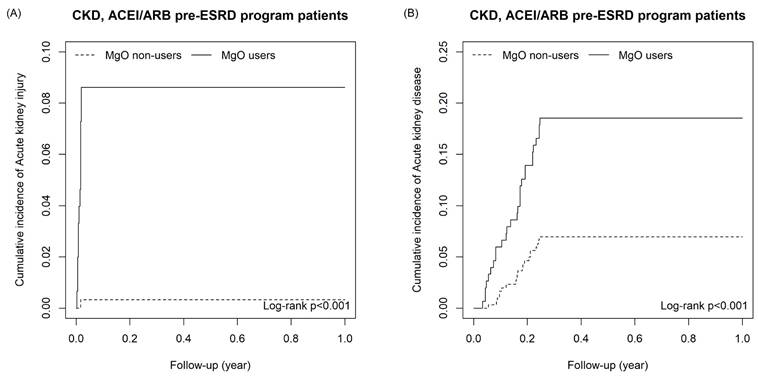
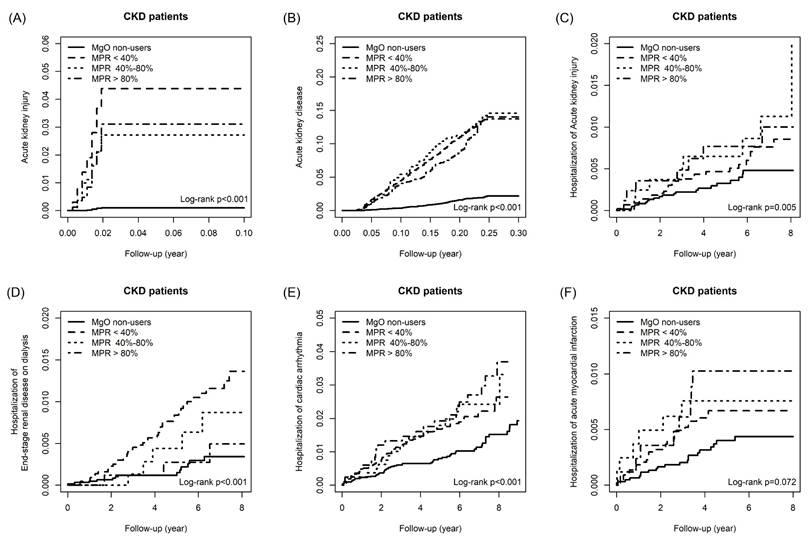
As shown as Table 2a, the risk of outcomes was higher in MgO user relative to the control group in all CKD cohort with and without matching. For all CKD patients, the adjusted HR of AKI, AKD, hospitalization of acute kidney injury, end-stage renal disease on dialysis, congestive heart failure with acute pulmonary edema, cardiac arrhythmia and acute myocardial infarction were 45.40 (22.4, 92.07), 6.43(5.53, 7.47), 1.87(1.16, 2.99), 2.59(1.58, 4.22), 2.79(1.07,7.27), 2.18(1.66, 2.86) and 1.75(1.12, 2.73), respectively. For the matching cohort, AKI (aHR=37.00, 95%CI=16.46, 83.41), AKD (aHR=6.26, 95%CI=5.20, 7.53), hospitalization of acute kidney injury (aHR=1.97, 95%CI=1.15, 3.37), end-stage renal disease on dialysis (aHR=2.06, 95%CI=1.49, 2.84) and cardiac arrhythmia (aHR=1.86, 95%CI=1.10, 3.15) remained significantly higher. Figure 2 presents the cumulative incidence of six outcomes between MgO user and non-user in all CKD patients. Table 2b presents that in the un-matched cohort of CKD patients with receiving ACEI/ARB and Pre-ESRD program, the risk of all outcomes were higher in the MgO user than that in the MgO non-user. For the matched cohort, patients used MgO have a higher risk of AKI (aHR=16.1; 95%CI=1.90, 136.96) and AKD (aHR=2.79; 95%CI=1.50, 5.20). The cumulative incidence curves of AKI and AKD for CKD patients with receiving ACEI/ARB and Pre-ESRD program presented in Figure 3.
Risk outcomes for CKD patients.
| Control | Magnesium oxide | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Outcome | n | PY | IR | n | PY | IR | cHR | (95% CI) | aHRꝉ | (95% CI) |
| Without matching | ||||||||||
| Acute kidney injury | 8 | 62316 | 0.13 | 250 | 33107 | 7.55 | 52.8 | (26.13, 106.81)*** | 45.4 | (22.4, 92.07)*** |
| Acute kidney disease | 221 | 61140 | 3.61 | 892 | 29694 | 30.04 | 7.20 | (6.21, 8.34)*** | 6.43 | (5.53, 7.47)*** |
| Hospitalization of acute kidney injury | 37 | 62252 | 0.59 | 45 | 34354 | 1.31 | 2.41 | (1.52, 3.80)*** | 1.87 | (1.16, 2.99)** |
| Hospitalization of end-stage renal disease on dialysis | 33 | 62314 | 0.53 | 54 | 34354 | 1.57 | 3.12 | (1.94, 5.02)*** | 2.59 | (1.58, 4.22)*** |
| Hospitalization of congestive heart failure with acute pulmonary edema | 8 | 62345 | 0.13 | 13 | 34436 | 0.38 | 3.37 | (1.33, 8.53)* | 2.79 | (1.07, 7.27)* |
| Hospitalization of cardiac arrhythmia | 192 | 61411 | 3.13 | 197 | 33639 | 5.86 | 2.41 | (1.86, 3.14)*** | 2.18 | (1.66, 2.86)*** |
| Hospitalization of acute myocardial infarction | 40 | 62188 | 0.64 | 41 | 34289 | 1.20 | 1.71 | (1.10, 2.64)* | 1.75 | (1.12, 2.73)* |
| Death | 471 | 72022 | 6.54 | 579 | 39934 | 14.5 | 2.11 | (1.87, 2.39)*** | 1.78 | (1.56, 2.02)*** |
| 1:1 PS matching | ||||||||||
| Acute kidney injury | 6 | 33713 | 0.18 | 239 | 32600 | 7.33 | 40.4 | (18.01, 91.02)*** | 37.0 | (16.46, 83.41)*** |
| Acute kidney disease | 132 | 33038 | 4.00 | 844 | 29366 | 28.74 | 6.82 | (5.68, 8.20)*** | 6.26 | (5.20, 7.53)*** |
| Hospitalization of acute kidney injury | 23 | 33676 | 0.68 | 44 | 33787 | 1.3 | 2.14 | (1.26, 3.64)** | 1.97 | (1.15, 3.37)* |
| Hospitalization of end-stage renal disease on dialysis | 16 | 33713 | 0.47 | 53 | 33784 | 1.57 | 3.70 | (1.97, 6.96)*** | 3.13 | (1.65, 5.94)*** |
| Hospitalization of congestive heart failure with acute pulmonary edema | 5 | 33739 | 0.15 | 12 | 33865 | 0.35 | 2.31 | (0.81, 6.59) | 1.94 | (0.67, 5.63) |
| Hospitalization of cardiac arrhythmia | 127 | 33081 | 3.84 | 180 | 33121 | 5.43 | 2.08 | (1.52, 2.86)*** | 2.06 | (1.49, 2.84)*** |
| Hospitalization of acute myocardial infarction | 21 | 33662 | 0.62 | 41 | 33719 | 1.22 | 1.87 | (1.11, 3.17)* | 1.86 | (1.10, 3.15)* |
| Death | 252 | 39488 | 6.38 | 563 | 39277 | 14.33 | 2.15 | (1.85, 2.49)*** | 2.10 | (1.80, 2.44)*** |
N: number of event; PY: person-year; IR: incidence rate per 1000 person-years; cHR: crude hazard ratio; aHR: adjusted hazard ratio; ꝉ: adjusted for sex, age, comorbidities and medication, *: p-value<0.05; **: p-value<0.01; ***: p-value<0.001.
The cumulative incidence of Acute kidney injury (A), Acute kidney disease (B), Hospitalization of acute kidney injury (C), Hospitalization of end-stage renal disease on dialysis (D), Hospitalization of cardiac arrhythmia (E) and Hospitalization of acute myocardial infarction (F) in CKD patients.
Risk outcomes for CKD patients with receiving ACEI/ARB.
| Control | Magnesium oxide | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Outcome | n | PY | IR | n | PY | IR | cHR | (95% CI) | aHR | (95% CI) |
| Without matching | ||||||||||
| Acute kidney injury | 3 | 5318 | 0.56 | 13 | 805 | 16.15 | 29.9 | (8.54, 105.21)*** | 47.0 | (10.7, 206.68)*** |
| Acute kidney disease | 120 | 4934 | 24.32 | 62 | 635 | 97.56 | 4.02 | (2.96, 5.47)*** | 3.74 | (2.71, 5.17)*** |
| Hospitalization of acute kidney injury | 12 | 5315 | 2.26 | 8 | 840 | 9.52 | 4.17 | (1.7, 10.23)** | 6.34 | (2.34, 17.19)*** |
| Hospitalization of end-stage renal disease on dialysis | 18 | 5305 | 3.39 | 14 | 833 | 16.81 | 4.15 | (1.83, 9.38)*** | 4.08 | (1.61, 10.35)** |
| Hospitalization of congestive heart failure with acute pulmonary edema | 2 | 5329 | 0.38 | 1 | 852 | 1.17 | 3.19 | (0.29, 35.17) | NA | - |
| Hospitalization of cardiac arrhythmia | 23 | 5237 | 4.39 | 8 | 829 | 9.65 | 2.70 | (0.95, 7.66) | 3.59 | (1.14, 11.31)* |
| Hospitalization of acute myocardial infarction | 1 | 5326 | 0.19 | 3 | 847 | 3.54 | 20.3 | (2.12, 195.8)** | 26.9 | (2.39, 304.42)** |
| Death | 284 | 6390 | 44.44 | 93 | 957 | 97.15 | 2.23 | (1.77, 2.83)*** | 2.04 | (1.58, 2.65)*** |
| 1:2 PS matching | ||||||||||
| Acute kidney injury | 1 | 909 | 1.1 | 13 | 426 | 30.55 | 26.9 | (3.53, 206.16)** | 16.1 | (1.90, 136.96)* |
| Acute kidney disease | 21 | 847 | 24.79 | 28 | 385 | 72.66 | 2.86 | (1.62, 5.03)*** | 2.79 | (1.50, 5.20)** |
| Hospitalization of acute kidney injury | 1 | 911 | 1.1 | 1 | 471 | 2.12 | 1.94 | (0.12, 31.08) | NA | - |
| Hospitalization of end-stage renal disease on dialysis | 9 | 899 | 10.02 | 3 | 467 | 6.42 | 0.61 | (0.16, 2.24) | 1.15 | (0.19, 6.92) |
| Hospitalization of congestive heart failure with acute pulmonary edema | 0 | 912 | 0 | 0 | 471 | 0 | NA | - | NA | - |
| Hospitalization of cardiac arrhythmia | 1 | 912 | 1.1 | 3 | 464 | 6.47 | 5.65 | (0.59, 54.35) | NA | - |
| Hospitalization of acute myocardial infarction | 1 | 912 | 1.1 | 1 | 468 | 2.14 | NA | - | NA | - |
| Death | 18 | 1189 | 15.14 | 6 | 614 | 9.78 | 0.64 | (0.25, 1.62) | 0.39 | (0.13, 1.23) |
N: number of events; PY: person-year; IR: incidence rate per 1000 person-years; cHR: crude hazard ratio; aHR: adjusted hazard ratio; ꝉ: adjusted for sex, age, comorbidities and medication, *: p-value<0.05; **: p-value<0.01; ***: p-value<0.001.
The cumulative incidence of Acute kidney injury (A) and Acute kidney disease (B) in CKD, ACEI/ARB pre-ESRD program patients.
Considering the association between medication possession ratio (MPR) and outcomes in all CKD patients, Table 3 illustrated that patients with MPR <40%, 40%-80% and >80% increase the risk of AKI by 41.10 folds (95%CI=18.23, 92.97), 25.50 folds (95%CI=10.35, 63.13) and 28.40 folds (95%CI =11.71, 69.30), respectively, compared to the MgO non-user. For the outcome of AKD, the aHR of MPR<40%, 40-80% and >80% were 6.21 (95%CI=5.13, 7.50), 6.83 (95%CI=1.50, 6.87) and 6.02 (95%CI=4.86, 7.74). Only patients with MPR<40% (aHR= 1.86; 95%CI=1.03, 3.34) and 40-80% (aHR= 3.21; 95%CI=1.50, 6.87) had a higher risk of hospitalization of acute kidney injury relative to the control group. After controlling the confounder, MPR <40% user increase the risk of hospitalization of end-stage renal disease on dialysis by 3.46 times (95%CI=1.80, 6.64). With MgO non-user as the reference group, the aHR of hospitalization of cardiac arrhythmia for MPR<40% patients were 1.96 (95%CI=1.39, 2.77), that for MPR 40-80% patients were 2.21 (95%CI=1.30, 3.78) and that for MPR>80% patients were 2.37 (95%CI=1.39, 2.77). Only patients with MPR >80% (aHR=2.66; 95%CI=1.17, 6.04) increase the risk of hospitalization of acute myocardial infarction. Figure 4 is the cumulative incidence curves of all outcomes among different MPR patients.
The association of MPR and outcomes in the matched cohort of CKD patients.
| Outcome | n | PY | IR | cHR | (95% CI) | aHR | (95% CI) |
|---|---|---|---|---|---|---|---|
| Acute kidney injury | |||||||
| Control | 6 | 33713 | 0.18 | 1.00 | (reference) | 1.00 | (reference) |
| MPR < 40% | 191 | 22905 | 8.34 | 44.70 | (19.84, 100.75)*** | 41.10 | (18.23, 92.97)*** |
| MPR 40%-80% | 22 | 4652 | 4.73 | 27.40 | (11.14, 67.77)*** | 25.50 | (10.35, 63.13)*** |
| MPR > 80% | 26 | 5043 | 5.16 | 31.30 | (12.91, 76.2)*** | 28.40 | (11.71, 69.30)*** |
| Acute kidney disease | |||||||
| Control | 132 | 33038 | 4.00 | 1.00 | (reference) | 1.00 | (reference) |
| MPR < 40% | 611 | 20709 | 29.50 | 6.81 | (5.65, 8.22)*** | 6.21 | (5.13, 7.50)*** |
| MPR 40%-80% | 118 | 4117 | 28.66 | 7.12 | (5.55, 9.13)*** | 6.83 | (5.32, 8.76)*** |
| MPR > 80% | 115 | 4540 | 25.33 | 6.59 | (5.13, 8.46)*** | 6.02 | (4.68, 7.74)*** |
| Hospitalization of acute kidney injury | |||||||
| Control | 23 | 33676 | 0.68 | 1.00 | (reference) | 1.00 | (reference) |
| MPR < 40% | 26 | 23850 | 1.09 | 1.80 | (1.00, 3.23)* | 1.86 | (1.03, 3.34)* |
| MPR 40%-80% | 10 | 4758 | 2.10 | 3.43 | (1.60, 7.34)** | 3.21 | (1.50, 6.87)** |
| MPR > 80% | 8 | 5180 | 1.54 | 2.48 | (1.09, 5.65)* | 2.14 | (0.94, 4.89) |
| Hospitalization of end-stage renal disease on dialysis | |||||||
| Control | 16 | 33713 | 0.47 | 1.00 | (reference) | 1.00 | (reference) |
| MPR < 40% | 45 | 23823 | 1.89 | 4.39 | (2.31, 8.36)*** | 3.46 | (1.80, 6.64)*** |
| MPR 40%-80% | 5 | 4763 | 1.05 | 2.67 | (0.94, 7.58) | 2.78 | (0.97, 7.98) |
| MPR > 80% | 3 | 5198 | 0.58 | 1.47 | (0.42, 5.22) | 1.54 | (0.43, 5.52) |
| Hospitalization of congestive heart failure with acute pulmonary edema | |||||||
| Control | 5 | 33739 | 0.15 | 1.00 | (reference) | 1.00 | (reference) |
| MPR < 40% | 9 | 23888 | 0.38 | 2.53 | (0.84, 7.62) | 2.30 | (0.75, 7.09) |
| MPR 40%-80% | 2 | 4771 | 0.42 | 2.66 | (0.52, 13.74) | 1.71 | (0.31, 9.42) |
| MPR > 80% | 1 | 5206 | 0.19 | 1.15 | (0.13, 9.88) | 0.91 | (0.10, 7.96) |
| Hospitalization of cardiac arrhythmia | |||||||
| Control | 127 | 33081 | 3.84 | 1.00 | (reference) | 1.00 | (reference) |
| MPR < 40% | 124 | 23404 | 5.30 | 1.98 | (1.41, 2.78)*** | 1.96 | (1.39, 2.77)*** |
| MPR 40%-80% | 27 | 4656 | 5.80 | 2.19 | (1.29, 3.72)** | 2.21 | (1.30, 3.78)** |
| MPR > 80% | 29 | 5061 | 5.73 | 2.46 | (1.50, 4.04)*** | 2.37 | (1.43, 3.92)*** |
| Hospitalization of acute myocardial infarction | |||||||
| Control | 21 | 33662 | 0.62 | 1.00 | (reference) | 1.00 | (reference) |
| MPR < 40% | 27 | 23797 | 1.13 | 1.71 | (0.97, 3.03) | 1.69 | (0.96, 3.00) |
| MPR 40%-80% | 6 | 4745 | 1.26 | 2.01 | (0.81, 4.99) | 2.07 | (0.84, 5.15) |
| MPR > 80% | 8 | 5176 | 1.55 | 2.56 | (1.13, 5.78)* | 2.66 | (1.17, 6.04)* |
N: number of event; PY: person-year; IR: incidence rate per 1000 person-years; cHR: crude hazard ratio; aHR: adjusted hazard ratio; MPR: Medication Possession Ratio; ꝉ: adjusted for sex, age, comorbidities and medication, *: p-value<0.05; **: p-value<0.01; ***: p-value<0.001.
The cumulative incidence of Acute kidney injury (A), Acute kidney disease (B), Hospitalization of acute kidney injury (C), Hospitalization of end-stage renal disease on dialysis (D), Hospitalization of cardiac arrhythmia (E) and Hospitalization of acute myocardial infarction (F) in CKD patients among difference medication possession ratios.
Association of different CKD stages with outcomes
Table 4a and b present the associations between different CKD stages and clinical outcomes among MgO users, both in unmatched and propensity score-matched cohorts. For AKI, the risk rose progressively with advancing CKD stage. Compared with stage 1, patients in stage 3-5 exhibited significantly higher risks, with aHR of 2.16 for stage 3, 3.97 for stage 4, and 1.60-2.38 for stage 5 in unmatched and matched cohorts. For AKD, risk was strongly stage-dependent. Patients with stage 3 had a 1.70-fold higher risk, stage 4 a 2.60-fold risk, and stage 5 a 3.70-fold risk in the unmatched cohort, with similar magnitudes after matching. Notably, stage 5 patients demonstrated the highest incidence rates (197-229 per 1000 person-years). Regarding hospitalization for AKI, CKD stage 3-5 patients carried substantially elevated risks. Stage 4 was associated with aHR of 6.06-4.41, while stage 5 patients had 3.73-fold higher risks than stage 1 after matching. For progression to ESRD requiring dialysis, the risks increased steeply by CKD stage. Compared with stage 1, adjusted risks were 6.72-fold higher in stage 4 and 27.9-fold higher in stage 5 in the unmatched cohort; after matching, stage 5 remained markedly elevated (aHR 13.5, 95% CI 10.2-17.9). For cardiac arrhythmia, stage 3 and stage 4 patients consistently had higher risks, with aHR of ~2.0 and 3.2-4.2, respectively, while stage 5 patients also showed increased but more variable risks. For acute myocardial infarction, the associations were weaker; only stage 5 showed a modest increase in the unmatched analysis (aHR 3.24, 95% CI 0.96-10.93), though confidence intervals were wide. Overall, both analyses confirmed a clear dose-response relationship: advancing CKD stage in MgO users was associated with progressively higher risks of AKI, AKD, dialysis initiation, and arrhythmia, with the strongest effects observed in stage 4-5 disease.
Association of different CKD stages with outcomes in the unmatched Cohort of MgO users.
| Without matching | 1:1 PS matching | |||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Outcome | n | PY | IR | cHR | (95% CI) | aHR | (95% CI) | n | PY | IR | cHR | (95% CI) | aHR | (95% CI) |
| Acute kidney injury | ||||||||||||||
| CKD stage 1 | 113 | 23066 | 4.90 | 1.00 | (reference) | 1.00 | (reference) | 118 | 22853 | 5.16 | 1.00 | (reference) | 1.00 | (reference) |
| CKD stage 2 | 33 | 4695 | 7.03 | 1.16 | (0.78, 1.70) | 1.12 | (0.76, 1.66) | 24 | 3914 | 6.13 | 0.87 | (0.56, 1.36) | 0.90 | (0.58, 1.40) |
| CKD stage 3 | 66 | 4048 | 16.31 | 2.79 | (2.06, 3.78)*** | 2.16 | (1.57, 2.98)*** | 54 | 4262 | 12.67 | 1.96 | (1.42, 2.71)*** | 1.50 | (1.07, 2.10)* |
| CKD stage 4 | 27 | 722 | 37.39 | 5.82 | (3.82, 8.85)*** | 3.97 | (2.55, 6.16)*** | 22 | 920 | 23.92 | 3.33 | (2.11, 5.25)*** | 1.89 | (1.17, 3.05)** |
| CKD stage 5 | 11 | 577 | 19.05 | 2.99 | (1.61, 5.55)*** | 1.60 | (0.84, 3.06) | 21 | 651 | 32.27 | 4.41 | (2.77, 7.01)*** | 2.38 | (1.44, 3.93)*** |
| Acute kidney disease | ||||||||||||||
| CKD stage 1 | 449 | 21118 | 21.26 | 1.00 | (reference) | 1.00 | (reference) | 449 | 20862 | 21.52 | 1.00 | (reference) | 1.00 | (reference) |
| CKD stage 2 | 138 | 4179 | 33.02 | 1.23 | (1.01, 1.48)* | 1.21 | (1.00, 1.47)* | 111 | 3548 | 31.28 | 1.06 | (0.86, 1.31) | 1.08 | (0.87, 1.33) |
| CKD stage 3 | 178 | 3505 | 50.79 | 1.95 | (1.64, 2.33)*** | 1.70 | (1.41, 2.04)*** | 150 | 3770 | 39.79 | 1.45 | (1.21, 1.75)*** | 1.20 | (0.99, 1.46) |
| CKD stage 4 | 57 | 585 | 97.40 | 3.27 | (2.48, 4.31)*** | 2.60 | (1.96, 3.46)*** | 55 | 785 | 70.04 | 2.27 | (1.72, 3.00)*** | 1.60 | (1.19, 2.14)** |
| CKD stage 5 | 70 | 306 | 228.51 | 5.71 | (4.44, 7.34)*** | 3.70 | (2.82, 4.87)*** | 79 | 401 | 197.18 | 5.10 | (4.02, 6.48)*** | 3.11 | (2.39, 4.05)*** |
| Hospitalization of acute kidney injury | ||||||||||||||
| CKD stage 1 | 16 | 23682 | 0.68 | 1.00 | (reference) | 1.00 | (reference) | 23 | 23523 | 0.98 | 1.00 | (reference) | 1.00 | (reference) |
| CKD stage 2 | 5 | 4846 | 1.03 | 1.55 | (0.56, 4.28) | 1.46 | (0.52, 4.07) | 3 | 4021 | 0.75 | 0.75 | (0.22, 2.59) | 0.79 | (0.23, 2.72) |
| CKD stage 3 | 18 | 4349 | 4.14 | 5.62 | (2.82, 11.23)*** | 4.23 | (2.00, 8.93)*** | 8 | 4471 | 1.79 | 2.05 | (0.90, 4.64) | 1.51 | (0.64, 3.54) |
| CKD stage 4 | 5 | 846 | 5.91 | 9.14 | (3.33, 25.09)*** | 6.06 | (2.06, 17.84)** | 6 | 1027 | 5.84 | 6.68 | (2.68, 16.62)*** | 4.41 | (1.63, 11.93)** |
| CKD stage 5 | 1 | 632 | 1.58 | 2.45 | (0.32, 18.54) | 1.47 | (0.18, 11.73) | 4 | 745 | 5.37 | 6.20 | (2.12, 18.13)*** | 3.73 | (1.16, 12.04)* |
| Hospitalization of end-stage renal disease on dialysis | ||||||||||||||
| CKD stage 1 | 196 | 23012 | 8.52 | 1.00 | (reference) | 1.00 | (reference) | 211 | 22801 | 9.25 | 1.00 | (reference) | 1.00 | (reference) |
| CKD stage 2 | 24 | 4796 | 5.00 | 0.57 | (0.37, 0.87)** | 0.58 | (0.38, 0.88)* | 21 | 3986 | 5.27 | 0.53 | (0.34, 0.84)** | 0.56 | (0.36, 0.88)* |
| CKD stage 3 | 61 | 4256 | 14.33 | 1.64 | (1.23, 2.19)*** | 1.40 | (1.03, 1.89)* | 43 | 4414 | 9.74 | 1.01 | (0.73, 1.40) | 0.84 | (0.59, 1.17) |
| CKD stage 4 | 62 | 729 | 85.06 | 9.40 | (7.05, 12.53)*** | 6.72 | (4.94, 9.13)*** | 56 | 913 | 61.34 | 6.07 | (4.51, 8.17)*** | 3.88 | (2.81, 5.34)*** |
| CKD stage 5 | 106 | 207 | 510.97 | 44.50 | (34.84, 56.86)*** | 27.90 | (21.09, 37.12)*** | 100 | 356 | 281.04 | 24.10 | (18.90, 30.74)*** | 13.50 | (10.17, 17.92)*** |
| Hospitalization of congestive heart failure with acute pulmonary edema | ||||||||||||||
| CKD stage 1 | 9 | 23695 | 0.38 | 1.00 | (reference) | 1.00 | (reference) | 8 | 23538 | 0.34 | 1.00 | (reference) | 1.00 | (reference) |
| CKD stage 2 | 1 | 4851 | 0.21 | 0.72 | (0.09, 5.72) | 0.59 | (0.07, 4.82) | 1 | 4023 | 0.25 | 1.02 | (0.13, 8.30) | 0.91 | (0.11, 7.79) |
| CKD stage 3 | 3 | 4389 | 0.68 | 2.06 | (0.56, 7.63) | 1.72 | (0.42, 7.03) | 1 | 4497 | 0.22 | 0.81 | (0.10, 6.50) | 0.51 | (0.06, 4.76) |
| CKD stage 4 | 0 | 865 | 0.00 | 2 | 1043 | 1.92 | 7.42 | (1.56, 35.41)* | 6.48 | (1.12, 37.53)* | ||||
| CKD stage 5 | 0 | 637 | 0.00 | 0 | 763 | 0.00 | NA | - | NA | - | ||||
| Hospitalization of cardiac arrhythmia | ||||||||||||||
| CKD stage 1 | 105 | 23302 | 4.51 | 1.00 | (reference) | 1.00 | (reference) | 94 | 23139 | 4.06 | 1.00 | (reference) | 1.00 | (reference) |
| CKD stage 2 | 27 | 4759 | 5.67 | 1.42 | (0.87, 2.31) | 1.42 | (0.87, 2.33) | 23 | 3955 | 5.82 | 1.44 | (0.84, 2.46) | 1.50 | (0.87, 2.57) |
| CKD stage 3 | 45 | 4168 | 10.80 | 2.23 | (1.44, 3.44)*** | 2.01 | (1.26, 3.20)** | 41 | 4324 | 9.48 | 1.79 | (1.11, 2.88)* | 1.71 | (1.03, 2.83)* |
| CKD stage 4 | 13 | 796 | 16.34 | 3.99 | (2.05, 7.75)*** | 4.16 | (2.08, 8.29)*** | 15 | 968 | 15.49 | 3.40 | (1.74, 6.62)*** | 3.23 | (1.59, 6.53)** |
| CKD stage 5 | 7 | 614 | 11.40 | 2.61 | (1.05, 6.46)* | 2.39 | (0.93, 6.14) | 7 | 735 | 9.53 | 2.27 | (0.91, 5.64) | 2.40 | (0.93, 6.22) |
| Hospitalization of acute myocardial infarction | ||||||||||||||
| CKD stage 1 | 27 | 23600 | 1.14 | 1.00 | (reference) | 1.00 | (reference) | 30 | 23432 | 1.28 | 1.00 | (reference) | 1.00 | (reference) |
| CKD stage 2 | 6 | 4828 | 1.24 | 0.94 | (0.39, 2.27) | 0.84 | (0.35, 2.05) | 3 | 4012 | 0.75 | 0.47 | (0.14, 1.55) | 0.45 | (0.14, 1.46) |
| CKD stage 3 | 5 | 4372 | 1.14 | 0.90 | (0.35, 2.34) | 0.83 | (0.31, 2.21) | 2 | 4492 | 0.45 | 0.30 | (0.07, 1.24) | 0.28 | (0.07, 1.18) |
| CKD stage 4 | 0 | 865 | 0.00 | NA | - | NA | - | 4 | 1027 | 3.89 | 2.51 | (0.88, 7.13) | 2.63 | (0.90, 7.67) |
| CKD stage 5 | 3 | 624 | 4.81 | 3.60 | (1.09, 11.88)* | 3.24 | (0.96, 10.93) | 2 | 756 | 2.64 | 1.73 | (0.41, 7.23) | 1.61 | (0.38, 6.86) |
N: number of event; PY: person-year; IR: incidence rate per 1000 person-years; cHR: crude hazard ratio; aHR: adjusted hazard ratio; ꝉ: adjusted for sex, age, comorbidites and medication, *: p-value<0.05; **: p-value<0.01; ***: p-value<0.001.
Discussion
To our knowledge, this is the first study to explore the associations between MgO use and the risk of cardiovascular events and dialysis in patients with advanced stage of CKD treated with ACEIs/ARBs. In this large cohort study, we examined the association between MgO use and clinical outcomes, with a focus on AKI, AKD, ESRD, and cardiovascular complications. These findings suggest that CKD patients, particularly those in more advanced stages of renal impairment, may be highly vulnerable to the adverse renal and cardiovascular outcomes associated with MgO use. Given the widespread use of MgO as a common therapeutic agent for constipation, dyspepsia, and suspected magnesium deficiency in clinical practice, these observations are clinically important. Physicians must carefully evaluate the balance between potential therapeutic benefits and the risks of harm when considering MgO therapy in this population. Close monitoring of renal function, cardiovascular status, and serum magnesium levels may be warranted to mitigate the likelihood of adverse outcomes. MgO use was associated with markedly increased risks; however, the magnitude of these associations suggests potential residual confounding or indication bias that cannot be fully excluded. Ultimately, further prospective studies are needed to clarify the causal relationship and to establish safer strategies for magnesium supplementation in CKD patients.
Magnesium homeostasis in humans
Laboratory indices, including serum total magnesium and ionized Mg²⁺, are also imperfect markers. Their values are influenced by circadian rhythm, systemic inflammation, hypoalbuminemia, renal function, and concurrent medication use. Moreover, serum concentrations reflect only ~1% of total body magnesium, as the majority (~90%) is stored intracellularly within bone and muscle tissue [24-26]. Mobilization from these stores can maintain serum magnesium within the normal range despite significant depletion. Consequently, normal serum magnesium levels may not accurately represent total body stores, and subclinical magnesium deficiency may remain undetected. Such hidden deficits can exert long-term physiological effects, increasing susceptibility to cardiovascular disease, metabolic disorders, and other chronic conditions [25-27].
Clinical outcomes and MgO use
Our results revealed that MgO users had significantly higher adjusted risks of AKI, AKD, and progression to ESRD, both in unmatched and propensity score-matched cohorts. These findings appear paradoxical, as prior observational studies have suggested that higher serum magnesium is protective against vascular calcification, cardiovascular mortality, and sudden cardiac death in CKD. One possible explanation is that MgO users in our cohort may have had worse baseline clinical profiles, including higher comorbidity burdens, gastrointestinal symptoms, or medication exposures, leading clinicians to prescribe MgO. Indeed, Table 1a and b demonstrated that MgO users had lower eGFR, lower albumin, and greater prevalence of comorbidities such as COPD and dementia. Although propensity matching minimized these imbalances, residual confounding cannot be excluded. Furthermore, poor adherence emerged as a critical determinant of outcomes. Patients with MPR <40% had a 41-fold increased risk of AKI and six-fold increased risk of AKD compared with non-users, and their risk of dialysis initiation and arrhythmia was significantly higher. These findings suggest that inconsistent MgO use may exacerbate fluctuations in magnesium balance, exposing patients to both hypomagnesemia and treatment gaps. In contrast, CKD patients with high adherence (MPR ≥80%) failed to show clear protective benefits, suggesting that MgO supplementation may be inadequate to offset the numerous risk factors in CKD and could potentially contribute to adverse outcomes.
Association of different CKD stages with outcomes in the matched Cohort of MgO users.
| Without matching | 1:1 PS matching | |||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Outcome | n | PY | IR | cHR | (95% CI) | aHR | (95% CI) | n | PY | IR | cHR | (95% CI) | aHR | (95% CI) |
| Acute kidney injury | ||||||||||||||
| CKD stage 1 | 113 | 23066 | 4.90 | 1.00 | (reference) | 1.00 | (reference) | 118 | 22853 | 5.16 | 1.00 | (reference) | 1.00 | (reference) |
| CKD stage 2 | 33 | 4695 | 7.03 | 1.16 | (0.78, 1.70) | 1.12 | (0.76, 1.66) | 24 | 3914 | 6.13 | 0.87 | (0.56, 1.36) | 0.90 | (0.58, 1.40) |
| CKD stage 3 | 66 | 4048 | 16.3 | 2.79 | (2.06, 3.78)*** | 2.16 | (1.57, 2.98)*** | 54 | 4262 | 12.67 | 1.96 | (1.42, 2.71)*** | 1.50 | (1.07, 2.10)* |
| CKD stage 4 | 27 | 722 | 37.4 | 5.82 | (3.82, 8.85)*** | 3.97 | (2.55, 6.16)*** | 22 | 920 | 23.92 | 3.33 | (2.11, 5.25)*** | 1.89 | (1.17, 3.05)** |
| CKD stage 5 | 11 | 577 | 19.1 | 2.99 | (1.61, 5.55)*** | 1.60 | (0.84, 3.06) | 21 | 651 | 32.27 | 4.41 | (2.77, 7.01)*** | 2.38 | (1.44, 3.93)*** |
| Acute kidney disease | ||||||||||||||
| CKD stage 1 | 449 | 21118 | 21.3 | 1.00 | (reference) | 1.00 | (reference) | 449 | 20862 | 21.52 | 1.00 | (reference) | 1.00 | (reference) |
| CKD stage 2 | 138 | 4179 | 33.0 | 1.23 | (1.01, 1.48)* | 1.21 | (1.00, 1.47)* | 111 | 3548 | 31.28 | 1.06 | (0.86, 1.31) | 1.08 | (0.87, 1.33) |
| CKD stage 3 | 178 | 3505 | 50.8 | 1.95 | (1.64, 2.33)*** | 1.70 | (1.41, 2.04)*** | 150 | 3770 | 39.79 | 1.45 | (1.21, 1.75)*** | 1.20 | (0.99, 1.46) |
| CKD stage 4 | 57 | 585 | 97.4 | 3.27 | (2.48, 4.31)*** | 2.60 | (1.96, 3.46)*** | 55 | 785 | 70.04 | 2.27 | (1.72, 3.00)*** | 1.60 | (1.19, 2.14)** |
| CKD stage 5 | 70 | 306 | 228.5 | 5.71 | (4.44, 7.34)*** | 3.70 | (2.82, 4.87)*** | 79 | 401 | 197.18 | 5.10 | (4.02, 6.48)*** | 3.11 | (2.39, 4.05)*** |
| Hospitalization of acute kidney injury | ||||||||||||||
| CKD stage 1 | 16 | 23682 | 0.68 | 1.00 | (reference) | 1.00 | (reference) | 23 | 23523 | 0.98 | 1.00 | (reference) | 1.00 | (reference) |
| CKD stage 2 | 5 | 4846 | 1.03 | 1.55 | (0.56, 4.28) | 1.46 | (0.52, 4.07) | 3 | 4021 | 0.75 | 0.75 | (0.22, 2.59) | 0.79 | (0.23, 2.72) |
| CKD stage 3 | 18 | 4349 | 4.14 | 5.62 | (2.82, 11.23)*** | 4.23 | (2.00, 8.93)*** | 8 | 4471 | 1.79 | 2.05 | (0.90, 4.64) | 1.51 | (0.64, 3.54) |
| CKD stage 4 | 5 | 846 | 5.91 | 9.14 | (3.33, 25.09)*** | 6.06 | (2.06, 17.84)** | 6 | 1027 | 5.84 | 6.68 | (2.68, 16.62)*** | 4.41 | (1.63, 11.93)** |
| CKD stage 5 | 1 | 632 | 1.58 | 2.45 | (0.32, 18.54) | 1.47 | (0.18, 11.73) | 4 | 745 | 5.37 | 6.20 | (2.12, 18.13)*** | 3.73 | (1.16, 12.04)* |
| Hospitalization of end-stage renal disease on dialysis | ||||||||||||||
| CKD stage 1 | 196 | 23012 | 8.52 | 1.00 | (reference) | 1.00 | (reference) | 211 | 22801 | 9.25 | 1.00 | (reference) | 1.00 | (reference) |
| CKD stage 2 | 24 | 4796 | 5.00 | 0.57 | (0.37, 0.87)** | 0.58 | (0.38, 0.88)* | 21 | 3986 | 5.27 | 0.53 | (0.34, 0.84)** | 0.56 | (0.36, 0.88)* |
| CKD stage 3 | 61 | 4256 | 14.33 | 1.64 | (1.23, 2.19)*** | 1.40 | (1.03, 1.89)* | 43 | 4414 | 9.74 | 1.01 | (0.73, 1.40) | 0.84 | (0.59, 1.17) |
| CKD stage 4 | 62 | 729 | 85.06 | 9.40 | (7.05, 12.53)*** | 6.72 | (4.94, 9.13)*** | 56 | 913 | 61.34 | 6.07 | (4.51, 8.17)*** | 3.88 | (2.81, 5.34)*** |
| CKD stage 5 | 106 | 207 | 510.97 | 44.50 | (34.84, 56.86)*** | 27.90 | (21.09, 37.12)*** | 100 | 356 | 281.04 | 24.10 | (18.90, 30.74)*** | 13.50 | (10.17, 17.92)*** |
| Hospitalization of congestive heart failure with acute pulmonary edema | ||||||||||||||
| CKD stage 1 | 9 | 23695 | 0.38 | 1.00 | (reference) | 1.00 | (reference) | 8 | 23538 | 0.34 | 1.00 | (reference) | 1.00 | (reference) |
| CKD stage 2 | 1 | 4851 | 0.21 | 0.72 | (0.09, 5.72) | 0.59 | (0.07, 4.82) | 1 | 4023 | 0.25 | 1.02 | (0.13, 8.30) | 0.91 | (0.11, 7.79) |
| CKD stage 3 | 3 | 4389 | 0.68 | 2.06 | (0.56, 7.63) | 1.72 | (0.42, 7.03) | 1 | 4497 | 0.22 | 0.81 | (0.10, 6.50) | 0.51 | (0.06, 4.76) |
| CKD stage 4 | 0 | 865 | 0.00 | 2 | 1043 | 1.92 | 7.42 | (1.56, 35.41)* | 6.48 | (1.12, 37.53)* | ||||
| CKD stage 5 | 0 | 637 | 0.00 | 0 | 763 | 0.00 | ||||||||
| Hospitalization of cardiac arrhythmia | ||||||||||||||
| CKD stage 1 | 105 | 23302 | 4.51 | 1.00 | (reference) | 1.00 | (reference) | 94 | 23139 | 4.06 | 1.00 | (reference) | 1.00 | (reference) |
| CKD stage 2 | 27 | 4759 | 5.67 | 1.42 | (0.87, 2.31) | 1.42 | (0.87, 2.33) | 23 | 3955 | 5.82 | 1.44 | (0.84, 2.46) | 1.50 | (0.87, 2.57) |
| CKD stage 3 | 45 | 4168 | 10.80 | 2.23 | (1.44, 3.44)*** | 2.01 | (1.26, 3.20)** | 41 | 4324 | 9.48 | 1.79 | (1.11, 2.88)* | 1.71 | (1.03, 2.83)* |
| CKD stage 4 | 13 | 796 | 16.34 | 3.99 | (2.05, 7.75)*** | 4.16 | (2.08, 8.29)*** | 15 | 968 | 15.49 | 3.40 | (1.74, 6.62)*** | 3.23 | (1.59, 6.53)** |
| CKD stage 5 | 7 | 614 | 11.40 | 2.61 | (1.05, 6.46)* | 2.39 | (0.93, 6.14) | 7 | 735 | 9.53 | 2.27 | (0.91, 5.64) | 2.40 | (0.93, 6.22) |
| Hospitalization of acute myocardial infarction | ||||||||||||||
| CKD stage 1 | 27 | 23600 | 1.14 | 1.00 | (reference) | 1.00 | (reference) | 30 | 23432 | 1.28 | 1.00 | (reference) | 1.00 | (reference) |
| CKD stage 2 | 6 | 4828 | 1.24 | 0.94 | (0.39, 2.27) | 0.84 | (0.35, 2.05) | 3 | 4012 | 0.75 | 0.47 | (0.14, 1.55) | 0.45 | (0.14, 1.46) |
| CKD stage 3 | 5 | 4372 | 1.14 | 0.90 | (0.35, 2.34) | 0.83 | (0.31, 2.21) | 2 | 4492 | 0.45 | 0.30 | (0.07, 1.24) | 0.28 | (0.07, 1.18) |
| CKD stage 4 | 0 | 865 | 0.00 | 4 | 1027 | 3.89 | 2.51 | (0.88, 7.13) | 2.63 | (0.90, 7.67) | ||||
| CKD stage 5 | 3 | 624 | 4.81 | 3.60 | (1.09, 11.88)* | 3.24 | (0.96, 10.93) | 2 | 756 | 2.64 | 1.73 | (0.41, 7.23) | 1.61 | (0.38, 6.86) |
N: number of event; PY: person-year; IR: incidence rate per 1000 person-years; cHR: crude hazard ratio; aHR: adjusted hazard ratio; ꝉ: adjusted for sex, age, comorbidites and medication, *: p-value<0.05; **: p-value<0.01; ***: p-value<0.001.
Dysmagnesemia and cardiovascular complications in CKD
The association between dysmagnesemia and cardiovascular disease has been reported. Low magnesium levels are linked to hypertension, insulin resistance, left ventricular hypertrophy, and arrhythmogenesis. Our study adds to this evidence by showing that MgO users, particularly those with low adherence, faced higher risks of cardiac arrhythmia and myocardial infarction. Interestingly, only patients with MPR ≥80% demonstrated a significantly increased risk of myocardial infarction, suggesting that both magnesium status and potential adverse effects of MgO therapy warrant further investigation. One possible hypothesis is that MgO-induced gastrointestinal side effects or other unknown cardiovascular harmfulness, such as diarrhea, may worsen volume status and precipitate cardiovascular stress in CKD patients. Previous research results regarding magnesium supplementation in CKD were controversial. Higher serum magnesium was inversely associated with cardiovascular mortality in hemodialysis patients [8]. Other epidemiological studies emphasized the protective role of magnesium against vascular calcification [9, 14]. However, several studies highlighted the unanswered questions regarding the safety of magnesium supplementation in patients with advanced stages of CKD, particularly in the context of limited renal excretion [28-33].
Alternative strategies to optimize magnesium status—such as dietary counseling, tailored dialysate magnesium concentration, or use of other magnesium formulations with better bioavailability—should be considered in CKD care. Clinicians should remain alert to the potential adverse effects of MgO, such as gastrointestinal intolerance and the risk of hypermagnesemia in patients with advanced CKD, and adjust therapy as appropriate. Several studies also highlighted important safety considerations regarding the widespread use of MgO for constipation, particularly in populations with impaired renal function. Terashima et al. reported that MgO administration was associated with an increased risk of hospital readmission among patients with both heart failure and constipation [34]. This finding suggests that even standard-dose MgO may contribute to clinical instability in vulnerable cardiovascular populations, potentially through fluid-electrolyte disturbances, altered neurohormonal responses, or subclinical accumulation of magnesium in the setting of reduced renal perfusion. Ishii et al. further demonstrated that low creatinine clearance and higher daily magnesium intake were significant and independent predictors of hypermagnesemia in older adults [35]. Because creatinine clearance declines physiologically with age—and much more profoundly in patients with CKD—these results underscore the heightened susceptibility of CKD patients to magnesium accumulation even when MgO is prescribed at conventional doses. Hypermagnesemia in advanced CKD can develop insidiously and may remain asymptomatic until serum magnesium concentrations exceed the compensatory threshold.
Mori et al. also presented the clinical manifestations of MgO-induced hypermagnesemia in constipated patients [36]. Reported symptoms ranged from nonspecific findings such as nausea, lethargy, and hypotonia to more serious consequences including bradyarrhythmia, hypotension, and depressed consciousness. Notably, many affected individuals were elderly and had unrecognized renal impairment, supporting the concept that routine monitoring is essential when MgO is used in populations at risk. Collectively, these studies indicate that CKD patients—particularly those with stage 3-5 disease, heart failure comorbidity, or older age—are at substantial risk for magnesium accumulation and adverse events [36-38]. Given the impaired renal excretion of magnesium and the narrow safety margin in CKD, clinicians should consider alternative constipation therapies, closely monitor serum magnesium levels, and individualize dosing strategies when MgO is deemed necessary.
In a large CKD cohort study, serum magnesium levels <1.9 mg/dL or >2.1 mg/dL were linked to increased all-cause mortality. Low magnesium was reported to be associated with new-onset atrial fibrillation but not with composite cardiovascular events [39]. Further research is warranted to define the optimal serum magnesium range in CKD for preventing adverse outcomes. Our study findings extend these observations by demonstrating that real-world MgO prescriptions do not necessarily translate into improved outcomes. Additionally, an evidence-based serum magnesium reference interval that reflects optimal health, current dietary patterns, and population characteristics is urgently needed. In patients with CKD, where magnesium homeostasis is frequently disrupted, such a reference range would be particularly valuable for guiding clinical decision-making and preventing both deficiency- and excess-related complications. Patients with CKD commonly exhibit cardiovascular-kidney-metabolic (CKM) syndrome. A magnesium-depleted state, defined as a magnesium depletion score (MgDS) ≥ 2, represents a significant risk factor for progression to advanced CKM stages. This finding shows that early identification of magnesium deficiency and optimization of magnesium nutritional status may help reduce the risk of advancing CKM [40].
Strengths and limitations
The strengths of this study include its large sample size, long follow-up, and detailed adjustment for comorbidities and medications. The use of MPR provided an objective measure of adherence, which revealed strong associations with outcomes. This study has several implications for clinical practice. First, routine monitoring of serum magnesium levels is essential, as normal values may mask subclinical magnesium deficiency due to mobilization from bone and muscle stores. Second, adherence to MgO therapy should be carefully assessed, as poor compliance markedly increased the risks of both renal and cardiovascular events. Several limitations must be acknowledged. In this cohort study, serum magnesium levels were not available for all patients and may not accurately reflect total body magnesium stores. Residual confounding by indication is possible, as patients prescribed MgO may have had unmeasured differences in nutritional status or gastrointestinal conditions. Several outcomes, particularly AKI and AKD, demonstrate high adjusted hazard ratios. While statistically significant, these estimates may raise concerns regarding residual confounding or outcome misclassification. Baseline renal vulnerability, surveillance bias, or medication indication bias may have impacts on the clinical outcomes. Although the propensity score matching was rigorously performed, the potential for residual confounding cannot be completely excluded. Patients receiving MgO therapy may have unmeasured characteristics—such as underlying gastrointestinal disorders, frailty, or nutritional deficiencies—that are not fully captured in administrative data and could independently influence renal or cardiovascular outcomes. In fact, serum magnesium reflects only a small fraction of total body magnesium and may not accurately represent intracellular or tissue magnesium stores. This clarification strengthens the biological interpretation of our findings and prevents misinterpretation of MgO as a direct causal agent. Additionally, the observational design precludes causal inference, and further interventional trials are warranted to clarify the role of magnesium supplementation in CKD patients.
Conclusion
In summary, while MgO remains a commonly used laxative in East Asia, its use in CKD requires a risk-stratified approach. Patients with mild CKD and without cardiovascular comorbidities or frailty may still use MgO cautiously at low doses with periodic monitoring of serum magnesium. However, individuals with moderate-to-severe CKD, older adults with reduced creatinine clearance, and those with heart failure or other high-risk features face a substantially higher likelihood of magnesium accumulation and related renal or cardiovascular complications. Integrating the current results with existing evidence underscores the importance of prioritizing safer alternatives, such as polyethylene glycol or lactulose, in these vulnerable groups. Clinicians should remain alert for early manifestations of hypermagnesemia and tailor constipation therapy to each patient's renal function and comorbidity profile to optimize safety in CKD management.
Acknowledgements
Funding
This study was supported by grants from the Research Fund of the Taoyuan Armed Forces General Hospital (TYAFGH-D-115024 and TYAFGH-E-115051) and Ministry of National Defense-Medical Affairs Bureau (MND-MAB-E-115253).
Data availability
The data that support the findings of this study are available from the corresponding author upon reasonable request.
Institutional review board statement
This study was approved by the Taipei Medical University Joint Institutional Review Board (TMU-JIRB No. N202410006).
Author contributions
PJH and CLC conceived and designed the study. Material preparation was performed by PJH, LLT, LYH, RLW, YHK, and CLC. Data collection was performed by PJH, LYH, and CLC. Data analysis was conducted by PJH, YHK, and CLC. Validation was performed by CCY, JSC, KLW, YHK, and PJH. PJH and CLC drafted the manuscript. All authors critically reviewed and revised the manuscript, approved the final version, and agreed to be accountable for all aspects of the work.
Competing Interests
The authors have declared that no competing interest exists.
References
1. van de Wal-Visscher ER, Kooman JP, van der Sande FM. Magnesium in Chronic Kidney Disease: Should We Care? Blood Purif. 2018;45:173-8
2. Felsenfeld AJ, Levine BS, Rodriguez M. Pathophysiology of Calcium, Phosphorus, and Magnesium Dysregulation in Chronic Kidney Disease. Semin Dial. 2015;28:564-77
3. Jahnen-Dechent W, Ketteler M. Magnesium basics. Clin Kidney J. 2012;5:i3-i14
4. Blaine J, Chonchol M, Levi M. Renal control of calcium, phosphate, and magnesium homeostasis. Clin J Am Soc Nephrol. 2015;10:1257-72
5. Spiegel DM. Magnesium in chronic kidney disease: unanswered questions. Blood Purif. 2011;31:172-6
6. Kumar SR, Kumar KGS, Gayathri R. Hypomagnesemia in Patients with Type 2 Diabetes Mellitus. J Assoc Physicians India. 2024;72:25-8
7. Schmaderer C, Braunisch MC, Suttmann Y, Lorenz G, Pham D, Haller B. et al. Reduced Mortality in Maintenance Haemodialysis Patients on High versus Low Dialysate Magnesium: A Pilot Study. Nutrients. 2017;9:926
8. Kanbay M, Yilmaz MI, Apetrii M, Saglam M, Yaman H, Unal HU. et al. Relationship between serum magnesium levels and cardiovascular events in chronic kidney disease patients. Am J Nephrol. 2012;36:228-37
9. Gommers LMM, Hoenderop JGJ, de Baaij JHF. Mechanisms of proton pump inhibitor-induced hypomagnesemia. Acta Physiol (Oxf). 2022;235:e13846
10. Costello RB, Elin RJ, Rosanoff A. et al. Perspective: The Case for an Evidence-Based Reference Interval for Serum Magnesium: The Time Has Come. Adv Nutr. 2016;7:977-93
11. DiNicolantonio JJ, O'Keefe JH, Wilson W. Subclinical magnesium deficiency: a principal driver of cardiovascular disease and a public health crisis. Open Heart. 2018;5:e000668
12. Kanbay M, Goldsmith D, Uyar ME, Turgut F, Covic A. Magnesium in chronic kidney disease: challenges and opportunities. Blood Purif. 2010;29:280-92
13. Chrysant SG. Proton pump inhibitor-induced hypomagnesemia complicated with serious cardiac arrhythmias. Expert Rev Cardiovasc Ther. 2019;17:345-51
14. Massy ZA, Drüeke TB. Magnesium and outcomes in patients with chronic kidney disease: focus on vascular calcification, atherosclerosis and survival. Clin Kidney J. 2012;5:i52-i61
15. Costello RB, Nielsen F. Interpreting magnesium status to enhance clinical care: key indicators. Curr Opin Clin Nutr Metab Care. 2017;20:504-11
16. Gröber U, Schmidt J, Kisters K. Magnesium in Prevention and Therapy. Nutrients. 2015;7:8199-226
17. Mori H, Tack J, Suzuki H. Magnesium Oxide in Constipation. Nutrients. 2021;13:421
18. Chawla LS, Bellomo R, Bihorac A, Goldstein SL, Siew ED, Bagshaw SM. et al. Acute kidney disease and renal recovery: consensus report of the Acute Disease Quality Initiative (ADQI) 16 Workgroup. Nat Rev Nephrol. 2017;13:241-57
19. Lameire NH, Levin A, Kellum JA, Cheung M, Jadoul M, Winkelmayer WC. et al. Harmonizing acute and chronic kidney disease definition and classification: report of a Kidney Disease: Improving Global Outcomes (KDIGO) Consensus Conference. Kidney Int. 2021;100:516-26
20. Goldstein M, Yassa T, Dacouris N, McFarlane P. Multidisciplinary predialysis care and morbidity and mortality of patients on dialysis. Am J Kidney Dis. 2004;44:706-14
21. Sikka R, Xia F, Aubert RE. Estimating medication persistency using administrative claims data. Am J Manag Care. 2005;11:449-57
22. Chien LN, Hsiao PJ, Chiu CC, Chen WT, Cheng CJ, Tsou LL. et al. Low-dose Spironolactone Combined with ACEIs/ARBs May Reduce Cardiovascular Events in Patients with CKD Stages 3b-5: A Nationwide Population-Based Cohort Study in Taiwan. Int J Med Sci. 2025;22:1404-14
23. Moolgavkar SH, Chang ET, Watson HN, Lau EC. An Assessment of the Cox Proportional Hazards Regression Model for Epidemiologic Studies. Risk Anal. 2018;38:777-94
24. Elin RJ. Assessment of magnesium status for diagnosis and therapy. Magnes Res. 2010;23:S194-8
25. Barbagallo M, Dominguez LJ. Magnesium and aging. Curr Pharm Des. 2010;16:832-9
26. Rosique-Esteban N, Guasch-Ferré M, Hernández-Alonso P, Salas-Salvadó J. Dietary Magnesium and Cardiovascular Disease: A Review with Emphasis in Epidemiological Studies. Nutrients. 2018;10:168
27. Tang H, Zhang X, Zhang J, Li Y, Del Gobbo LC, Zhai S. et al. Elevated serum magnesium associated with SGLT2 inhibitor use in type 2 diabetes patients: a meta-analysis of randomised controlled trials. Diabetologia. 2016;59:2546-51
28. Žeravica R, Ilinčić B, Čabarkapa V, Radosavkić I, Samac J, Nikoletić K. et al. Fractional excretion of magnesium and kidney function parameters in nondiabetic chronic kidney disease. Magnes Res. 2018;31:49-57
29. Uslu Gökceoğlu A, Comak E, Dogan CS, Koyun M, Akbas H, Akman S. Magnesium excretion and hypomagnesemia in pediatric renal transplant recipients. Ren Fail. 2014;36:1056-9
30. Deekajorndech T. A biomarker for detecting early tubulointerstitial disease and ischemia in glomerulonephropathy. Ren Fail. 2007;29:1013-17
31. Webster AC, Nagler E V, Morton RL, Masson P. Chronic Kidney Disease. Lancet. 2017;389:1238-52
32. Gheissari A, Andalib A, Labibzadeh N, Modarresi M, Azhir A, Merrikhi A. Fractional excretion of magnesium (FEMg), a marker for tubular dysfunction in children with clinically recovered ischemic acute tubular necrosis. Saudi J Kidney Dis Transpl. 2011;22:476-81
33. Hsiao PJ, Liao CY, Kao YH, Chan JS, Lin YF, Chuu CP. et al. Comparison of fractional excretion of electrolytes in patients at different stages of chronic kidney disease: A cross-sectional study. Medicine (Baltimore). 2020;99:e18709
34. Terashima J, Kambara T, Hori E, Koketsu R, Sakaguchi T, Osanai H. et al. Association between magnesium oxide use and readmission risk in patients with heart failure and constipation. J Pharm Health Care Sci. 2025;11:68
35. Ishii H, Sawada R, Shiomi M, Shibuya K. A case-control study showing low creatinine clearance and high magnesium intake as risk factors for hypermagnesemia in older individuals. Magnes Res. 2023;36:23-30
36. Mori H, Suzuki H, Hirai Y, Okuzawa A, Kayashima A, Kubosawa Y. et al. Clinical features of hypermagnesemia in patients with functional constipation taking daily magnesium oxide. J Clin Biochem Nutr. 2019;65:76-81
37. Sakaguchi Y, Fujii N, Shoji T, Hayashi T, Rakugi H, Iseki K, et al; Committee of Renal Data Registry of the Japanese Society for Dialysis Therapy. Magnesium modifies the cardiovascular mortality risk associated with hyperphosphatemia in patients undergoing hemodialysis: a cohort study. PLoS One. 2014;9:e116273
38. Sakaguchi Y, Hamano T, Isaka Y. Effects of Magnesium on the Phosphate Toxicity in Chronic Kidney Disease: Time for Intervention Studies. Nutrients. 2017;9:112
39. Negrea L, DeLozier SJ, Janes JL, Rahman M, Dobre M. Serum Magnesium and Cardiovascular Outcomes and Mortality in CKD: The Chronic Renal Insufficiency Cohort (CRIC). Kidney Med. 2021;3:183-92.e1
40. Tian J, Ding X, Ren X, Wang G, Wang W, Liu J. Association between Magnesium Depletion Score and Advanced Cardiovascular-Kidney-Metabolic Syndrome Stages in US Adults. Int J Med Sci. 2025;22:3101-11
Author contact
Corresponding author: Chu-Lin Chou, E-mail: chulin.choucom.

 Global reach, higher impact
Global reach, higher impact