Impact Factor ISSN: 1449-1907
Int J Med Sci 2026; 23(4):1431-1443. doi:10.7150/ijms.126651 This issue Cite
Review
Atropine Eye Drops in Childhood Myopia Control: A Review
1. Department of Pediatric Ophthalmology, Affiliated Hospital of Yunnan University, Kunming 650021, China.
2. Department of Ophthalmology, Affiliated Hospital of Yunnan University, Kunming 650021, China.
3. Department of Refractive Surgery Center, Affiliated Hospital of Yunnan University, Kunming 650021, China.
4. BioTissue (Tissue Tech Inc), Miami, Florida 33126, USA.
*The first 3 authors contribute equally to this manuscript.
Received 2025-9-21; Accepted 2026-2-5; Published 2026-3-9
Abstract
Myopia is a pandemic problem in this world. Per 2019 report of the World Health Organization in their first-ever report on vision, ~27% world's population are suffering from myopia. This trend is expected to increase to ~50% by 2050. Unfortunately, no cure method is available so far due to its complications of the mechanistic actions. Therefore, control of myopia is the focus clinically. Currently, it is suggested that 0.05% atropine is optimal, balanced in efficacy and rebound in controlling myopia initiation and progression. In such a control process, measurement of reduction in spherical equivalent refraction (SER) progression and axial length (AL) elongation are critical for efficacy and rebound of atropine eye drops. For example, in Low-concentration Atropine for Myopia Progression (LAMP) study over one year, 0.05%, 0.025%, and 0.01% atropine can reduce SER progression by 67%, 43%, and 27%, decrease AL elongation by 51%, 29%, and 12%, respectively, while 0.5%, 0.1%, and 0.01% atropine may increase rebound by 68.4%, 58.9%, and 24.3%, respectively. Mechanistically, atropine, a muscarinic receptor antagonist, is used as the primary medication for controlling myopia due to its effectiveness and affordability.
In this review, we provide a concurrent view of myopia epidemiology, atropine use to control myopia, its tentative mechanism, balance of its effects as well as side effects, and its clinical application methods in hope for clinical ophthalmologists to effectively control this problematic disease worldwide.
Keywords: atropine, myopia, axial length, spherical equivalent, rebound
1. Introduction: The Global Myopia Epidemic
Myopia (nearsightedness) is characterized by increased axial length (AL) of the eye, and this causes the refractive error known as “nearsightedness” [1] due to increased anterior-posterior diameter of the eye relative to refracting power of cornea and lens, distorted image focal point and blurred image in front of retina [2] (also reviewed in [3-8]). Technically, myopia is defined as the spherical equivalent of refractive error < -0.50 diopter (D) but > -5.00 D, high myopia as a spherical equivalent of refractive error ≤ -6.00 D in one eye [9].
Childhood myopia has evolved into a global public health crisis of unprecedented scale. Epidemiologically, ~50% people in the world will suffer myopia by 2050, with ~10% of high myopia (≤ -6.00 diopters) [10]. In some East Asian regions, nearly 90% of secondary-school children have myopia, with ~20% of high myopia [11], substantially increasing their risk of sight-threatening complications such as retinal detachment, myopic maculopathy, glaucoma, and cataracts [12].
Although there are significant differences of myopia occurrence among different regions, with East Asia having the highest rates (80-90% in teens), myopia is a global epidemic with rising rates in Europe, North America (around 42%), and developing nations, though lower than in Asia. South America, Africa, and parts of South Asia show lower, but still increasing, prevalence, demonstrating a global rise driven by lifestyle changes like increased near work and less outdoor time, making it a major public health concern worldwide [13].
The economic burden is staggering, encompassing direct costs of optical correction, medical management of complications, and immeasurable impacts on quality of life and productivity. In high-prevalence regions of East Asia, myopia onset now commonly occurs between 6-8 years of age, with rapid progression during primary school years [14]. A clinical investigation shows 0.01%-0.05% atropine satisfactorily controls myopia progression in East Asia regions [15]. This early onset is particularly concerning given the strong association between younger age at onset and eventual progression to high myopia. Consequently, effective interventions to slow down myopia progression in childhood have become one of the fastest-growing areas of ophthalmic research and clinical innovation, with low-dose atropine emerging as a cornerstone of pharmacological management.
Myopia's economic impact is massive, spanning billions in direct correction costs (glasses, contacts, surgery) and huge indirect losses from lost productivity, with projections hitting $229 billion by 2050 due to rising prevalence, especially high myopia linked to serious complications like glaucoma. Controlling progression saves money by reducing lifelong care and preventing costly, vision-threatening conditions, proving management is cheaper than treatment, even with upfront costs for interventions like specialized lenses or atropine, highlighting the societal benefit of prevention [16].
Myopia stems from a mix of genetics (runs in families) and environment, with key risk factors being lots of near work (screens, reading) and not enough outdoor time, which exposes eyes to bright light that slows eye growth. Factors like ethnicity (higher in Asians), early hyperopia, and certain binocular vision issues also increase risk, while spending over 90 mins outdoors daily significantly lowers onset risk, though its effect on progression after onset is less clear [17].
Atropine eyedrops are now being used for prevention of the onset of myopia in pre-myopic children with significant effect. For example, a graduated-concentration atropine protocol (0.025%, 0.05%, 0.125%) achieved satisfactory outcomes, defined as prevention of myopia onset and maintenance of axial elongation below the predefined progression threshold, in 64% of premyopic children at 2-year follow-up [18], a concentration of 0.025% atropine resulted that 21% of children in atropine group suffered myopia onset compared to 54% in control group in one year, indicating effectiveness in preventing myopia onset [19]. A case-control study demonstrated that 0.01% atropine group progressed -0.31D / 0.12mm compared to -0.76D / 0.21mm in the control group in one year and the atropine group progressed -0.60D / 0.21mm compared to -1.75D / 0.48mm in the control group in two years [20]. A crossover trial in China demonstrated that in 0.01% atropine group, myopia progressed -0.15D and 0.17mm compared to -0.34D and 0.28mm in placebo group in a 1-year study [21]. In LAMP2 Clinical Trial, nightly use of 0.05% atropine eye drops significantly reduced the 2-year cumulative incidence of myopia to 28.4%, compared to 53.0% respectively in the placebo group. This study demonstrated that 0.05% atropine is effective in preventing early myopia onset [22].
2. Pharmacological Profile and Putative Mechanisms of Action
2.1 Basic Pharmacology
Atropine, a non-selective muscarinic antagonist derived from the deadly nightshade plant Atropa belladonna, has a complex pharmacological profile that extends beyond its classical effects on pupil dilation (mydriasis) and accommodation paralysis (cycloplegia). When administered in low concentrations (typically 0.01% to 0.05%), it exerts biological effects at concentrations several orders of magnitude below those traditionally used for cycloplegic refraction or uveitis treatment. The molecular basis of its action involves competitive inhibition of acetylcholine at muscarinic receptors (M1-M5), though its affinity varies across receptor subtypes and tissue distributions [23].
Muscarinic acetylcholine receptors (M1-M5) are widely distributed throughout the central nervous system (CNS) and peripheral nervous system (PNS), particularly in organs innervated by parasympathetic neurons. Their specific distribution dictates their physiological function [24]. Importantly, atropine's ocular pharmacokinetics differ significantly according to its concentration. Low-concentration formulations may achieve minimal systemic absorption due to the minute quantities delivered (approximately 5-50 µg per drop), resulting in negligible serum concentrations and thus an excellent systemic safety profile. However, local ocular effects, particularly on retinal and scleral signaling, remain profound even at these ultra-low doses.
2.2 Evolving Understanding of Mechanisms
The precise mechanisms through which atropine slows myopia progression have been extensively investigated yet remain incompletely characterized (reviewed in [7, 25]). Earlier hypotheses centered on accommodation blockade, positing that by paralyzing the ciliary muscle, atropine might eliminate accommodative lag and its associated hyperopic defocus on the peripheral retina - a putative stimulus for axial elongation. However, animal studies have fundamentally challenged this view.
Non-accommodative Pathways
Intravitreal injection of atropine in animal models effectively inhibits axial elongation without affecting accommodative amplitude or pupillary constriction, demonstrating that its anti-myopic effects occur independently of accommodation [26]. The non-accommodative pathways include:
Muscarinic Receptors: Gene expression studies in myopic animal models reveal that atropine administration significantly upregulates muscarinic M1, M3, and M4 receptors in the sclera while leaving M2 and M5 largely unchanged. This receptor-specific pattern suggests targeted effects on scleral remodeling pathways [27].
The muscarinic receptors belong to G-protein coupled receptors (GPCRs), which might be activated by neurotransmitter acetylcholine [28, 29]. GPCRs activation proceeds with ligand binding, activating phospholipase C, inducing second messengers or diacylglycerol (DAG) and inositol triphosphate (IP3) [28, 29]. DAG and IP3 might then promote protein kinase C activation and calcium release intracellularly to induce signaling cascade [28, 29]. Alternatively, induction of inhibitory Gi-coupled receptors might lead to reduction of adenylyl cyclase, and thus protein kinase A, leading to cellular cyclic adenosine monophosphate inhibition and thus inhibitory cellular responses [28, 29]. Among muscarinic receptors, M1, M3, and M5 are stimulatory while M2 and M4 are inhibitory [29, 30].
Scleral Remodeling: Atropine appears to modulate the extracellular matrix composition of the sclera. In progressive myopia, scleral fibroblasts demonstrate reduced proliferation and downregulation of type I collagen and glycosaminoglycans (GAGs), leading to biomechanical weakening and ocular elongation. Mechanistically, atropine reduces myopia progression through increasing collagen synthesis and thus scleral thickness in the stromal layer [31].
During axial length elongation, structural changes occurred in myopia onset and progression [32]. In this process, sclera thinning arose primarily at the posterior pole [33]. Scleral remodeling relies on the changes of the scleral extracellular matrix (ECM), which plays a critical role in thinning of the sclera [34, 35]. During myopia progression, scleral collagen is steadily decreasing, leading to its progressive breakdown [36]. In addition, sclera proteoglycan is also steadily decreasing [37]. As a result, scleral fibril assembly is disrupted, and the tissue biomechanics deteriorate [38]. In summary, the results indicate that scleral structural changes contributes to myopia progression by scleral ECM remodeling (reviewed in [39]).
Scleral remodeling in myopia involves the following molecular pathways that mediate the synthesis as well as degradation of scleral extracellular matrix (ECM), particularly collagen. Key factors and signaling pathways and factors include the following: 1. Hypoxia-Inducible Factor-1α (HIF-1α) Signaling: Scleral tissue might become hypoxic (oxygen-deficient) in case of myopia because of decreased choroidal blood flow, which induces HIF-1α to promote trans-differentiation from fibroblasts to myofibroblasts and decreases the synthesis of type I collagen. This process leads to scleral thinning and weakening. 2. Matrix Metalloproteinases (MMPs) and Tissue Inhibitors of Metalloproteinases (TIMPs) Involved: Scleral remodeling is mediated by the balance of ECM-degrading enzymes (MMPs, such as MMP-2 and MMP-9) and their inhibitors (TIMPs, such as TIMP-2). In myopic sclera, MMP activity increases while TIMP activity decreases, causing excessive collagen degradation and decreased collagen fiber content. 3. Transforming Growth Factor-β (TGF-β) Signaling: The TGF-β family plays a significant role. For example, decreased levels of TGF-β subtypes, such as TGF-β1, are associated with reduced collagen synthesis, whereas increased levels of other types, such as TGF-β2, may contribute to scleral thinning by promoting degradation. This signaling interacts with endoplasmic reticulum (ER) stress and the Wnt pathways. 4. Wnt/β-catenin Signaling: Activation of Wnt signaling is associated with myopia progression by promoting scleral fibroblasts to myofibroblasts and decrease of COL-1 synthesis. 5. cAMP Pathway: cAMP signaling is linked to mediating eye growth. Increased cAMP is linked to inhibition of eye elongation through decreasing MMP-2 and increasing collagen synthesis. 6. YAP Signaling: Mechanical stiffness from ECM plays a significant role. Decrease of scleral stiffness in myopia is associated with decreased expression and nuclear translocation of YES-associated protein (YAP), which normally enhances COL1A1 expression. 6. Inflammatory and Other Factors: Pathways involving inflammatory cytokines such as IL-6, TNF-α, Sonic Hedgehog (Shh) pathway, Retinoic Acid (RA), as well as microRNAs also influence the complex regulation of scleral remodeling. In summary, these pathways ultimately cause scleral thinning, disorganizing collagen fibrils, and decreasing biomechanical strength in myopic eyes, and facilitating axial elongation (reviewed in [39]).
Retinal and Choroidal Signaling: Emerging evidence indicates that atropine increases choroidal thickness and choroidal blood perfusion pressure, potentially improving oxygenation of retinal tissues and counteracting hypoxia-driven ocular growth [40]. However, this conclusion is controversial because a recent report suggested that low dose atropine did affect choroidal thickness [41]. Additionally, atropine influences dopaminergic signaling in the retina, enhances GABAergic inhibition of ocular growth, and modulates inflammatory cytokine profiles in the retina and retinal pigment epithelium [42], probably through binding to mAchR on the cells to promote release of dopamine, thus inhibiting myopia progression [43, 44].
The current consensus suggests that atropine's anti-myopic effects result from a multifactorial orchestration of receptor-mediated events across multiple ocular tissues rather than a single unified mechanism. This complexity may explain why individual treatment responses vary and why combination therapies targeting multiple pathways simultaneously show promise.
The most well-studied pathway in illustrating atropine's mechanism of action is the competitive antagonist of muscarinic acetylcholine receptors. This core mechanism is the central mechanism to its primary clinical applications. Atropine binds to and inhibits muscarinic acetylcholine receptors, competitively blocking the effects of acetylcholine and other choline esters [7, 45, 46]. Such receptors include M1, M2, M3, M4, and M5 receptor subtypes [47]. Atropine inhibits effects of acetylcholine on postganglionic cholinergic nerves in tissue such as central nervous system, cardiac smooth muscle, and exocrine glands. It also acts in less innervated smooth muscle that responds to endogenous acetylcholine [45, 48]. The actions of atropine can be overcome by increasing the concentration of acetylcholine at receptor sites (for instance, the use of anticholinesterase agents that inhibit the hydrolysis of acetylcholine) [7]. Other mechanisms include, for example: 1. Atropine can act through cycloplegic action on smooth ciliary muscles to reduce accommodative eye reactions; 2. Atropine might promote choroidal thickening by ocular growth mechanistically (reviewed in [7]).
Summary of Key Mechanisms Implicated in Atropine's Anti-Myopic Effects
| Biological Target | Proposed Mechanism | Experimental Evidence |
|---|---|---|
| Muscarinic Receptors (M1, M3, M4) | Regulation of scleral fibroblast activity and extracellular matrix remodeling | Upregulation of receptor mRNA in sclera; increased collagen synthesis [49] |
| Choroidal Vasculature | Increased blood flow and thickness; improved retinal oxygenation | Enhanced choroidal perfusion in clinical studies; association with reduced axial elongation [50] |
| Retinal Dopamine Pathways | Modulation of dopamine release and receptor signaling | Restoration of dopamine circadian rhythms in form-deprived models; synergy with L-DOPA [51] |
| GABA Receptors | Activation of inhibitory GABAergic pathways | GABA agonists mimic atropine effects; GABA antagonists block atropine in animal models [42] |
| Inflammatory Cytokines | Downregulation of pro-growth cytokines | Reduced TNF-α, IL-1β, and other cytokines in retinal/RPE tissue after atropine [52] |
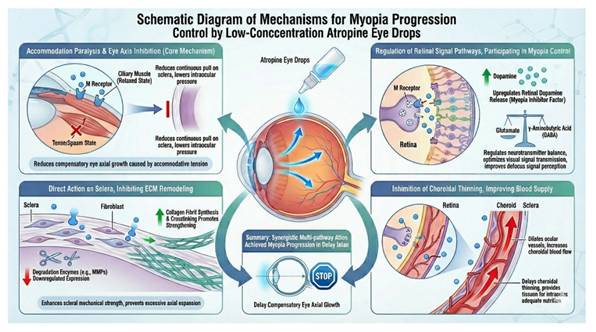
In summary, the major myopia control mechanism is summarized as follows.
Summary of Current Myopia Control Mechanism.
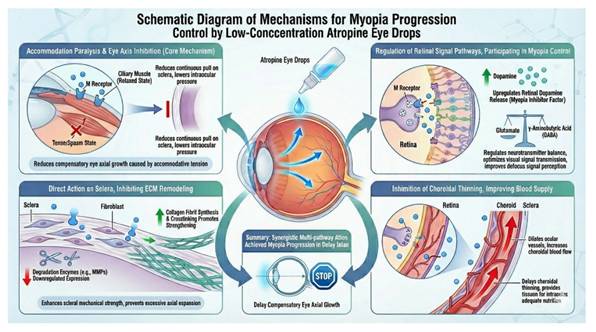
3. Clinical Efficacy Across Concentrations
3.1 Landmark Clinical Trials
The effect of atropine concentrations on myopia progression has been extensively investigated in several randomized controlled assessments, which sheds light on our clinical practice (reviewed in [53]):
ATOM Studies (Singapore): The pioneering ATOM1 Trial demonstrated that 1% atropine produced a remarkable 80% reduction in myopia progression compared to placebo over two years. Nevertheless, the dose at 1% concentration results in significant side effects, such as near vision blur and photophobia, closely related to a profound rebound side effect (~ -1.2 D) when discontinued [54]. In ATOM 1 Trial, axial length increased by 0.02 ± 0.35 mm in 1% atropine group after two years [55]. By contrast, axial length increased by 0.38 ± 0.38 mm in the control group [55]. After three years, axial length increased by 0.29 ± 0.37 mm in 1% atropine group in contrast to 0.52 ± 0.45 mm from the control group [56]. The subsequent ATOM2 Trial compared lower concentrations (0.5%, 0.1%, and 0.01%), finding that 0.01% atropine offered the optimal balance of efficacy (approximately 50-60% reduction in progression), minimal side effects, and negligible rebound after cessation [57]. In subsequent ATOM 2 Trial, the researchers found that the effectiveness of atropine was concentration dependent, based on axial length increase (by 0.27 ± 0.25 mm, by 0.28 ± 0.28 mm and by 0.41 ± 0.32 mm in 0.5%, 0.1% and 0.01% atropine groups respectively [58, 59]. High concentration, for example, 1% atropine, was profoundly more effective than low concentration atropine, for instance, 0.01% atropine, to inhibit axial length elongation by as much as 70% and 94% in a few clinical trials [54, 55, 59-63]. Currently, the ATOM3 Study, a randomized clinical trial enrolled children aged 5 to 9 years old, with one or two myopic parents and spherical equivalent refraction of +1.00 to -1.50, is still underway using 0.01% atropine eyedrops to prevent the onset or delay the progression of myopia in school children.
Long-term (10-20 years) follow-up data from ATOM participants, especially in Atropine Treatment Long-term Assessment Study (ATLAS), have confirmed the excellent safety profile of low-concentration atropine, with no increased incidence of cataracts, glaucoma, or other visually significant complications such as macular degeneration [64]. The drawback is that while no rebound effect after withdrawal of atropine treatment, myopic progression still occurs as adults, without overall changes of final refractive errors [64].
LAMP Study (Hong Kong): This rigorous RCT compared 0.05%, 0.025%, and 0.01% atropine over two years in children aged 4-12 years. Results demonstrated a clear concentration-dependent effect: 0.05% atropine reduced progression by 67% (spherical equivalent) and 51% (axial length), significantly outperforming both lower concentrations [65]. Notably, the 0.01% formulation showed only modest efficacy (27% reduction in SE progression), challenging the ATOM2 findings. The LAMP2 Extension further demonstrated that delayed initiation of 0.05% atropine in previously untreated children still produced substantial benefits, whereas switching from 0.01% to placebo resulted in accelerated "catch-up" progression [22]. In the subsequent five-year LAMP Phase 4 Study, the authors showed that treatment with 0.05% atropine effectively controlled myopia, and that re-treatment with 0.05% atropine remained effective after a period of treatment discontinuation [66].
In summary, the LAMP Study provides strong evidence that 0.05% atropine is the optimal concentration for myopia control among the tested doses, demonstrating a significant reduction in myopia progression and superior performance compared to lower concentrations like 0.01%.
3.2 Comparative Efficacy Analysis
More recent studies have refined our understanding of concentration-response relationships:
0.05% Concentration: The MOSAIC Trial (Europe) confirmed that 0.05% atropine significantly reduced progression compared to placebo over 24 months, even in older children (mean age 13.9 years) [67]. After three years, children who switched from placebo to 0.05% atropine experienced less progression and less axial elongation than those who switched from 0.01% atropine to placebo, highlighting its robust efficacy even when initiated late [67]. Similarly, a 2024 prospective study of 0.03% atropine demonstrated significant reductions in both spherical equivalent progression and axial elongation [68].
0.01% Concentration: Efficacy findings for this concentration have been somewhat inconsistent across trials. While ATOM2 reported a significant reduction in progression in two years, the LAMP Study found only a less reduction, and the LAMP2 Prevention Trial showed no significant difference from placebo in delaying myopia onset [22, 69, 70]. A 2023 U.S. Trial (n=489) provided further clarification: 0.01% atropine produced consistent and statistically significant slowing of both refractive shift and axial elongation over three years, though the magnitude of effect was more modest than higher concentrations [71]. This concentration appears most suitable for younger children with mild progression or as maintenance therapy after initial control with stronger concentrations.
The efficacy of different atropine concentrations may vary across populations and study regions. In particular, the difference in concentration-dependent efficacy between Eastern and Western populations are especially evident in the following: Higher Efficacy in Asian Populations: Many studies, including ATOM and LAMP Trials in Hong Kong and Singapore, have indicated greater efficacy of atropine with various concentrations for East Asian children, compared to white or South Asian populations. For example, meta-analyses consistently demonstrate a greater pooled treatment effect of atropine in East Asian populations compared with White populations [72]. Optimal Concentration Difference: For East Asian children, concentrations of 0.05% atropine are indicated as optimal. Higher concentrations (for example, 0.5% or 1%) of atropine show maximal efficacy, which are associated with more pronounced adverse effects and rapid myopia rebound after atropine cessation [73]. For white populations, the efficacy of 0.01% atropine was modest or not significant in several studies (for example, the PEDIG study in the US), while some other studies in Spain and USA have concluded its effectiveness with minimal side effects. More pronounced side effects, such as photophobia and reading difficulty, have been reported in concentrations like 0.05% in Caucasian populations compared with Asian children [68, 74]. Factors Affecting Mechanisms: Baseline Myopia Progression: Asian populations exhibit faster baseline myopia progression as well as greater axial elongation than European populations, which might cause greater treatment effect [75]. Genetic Factors: Genetic predispositions might affect individuals respond for atropine treatment [76]. Environmental Factors: Environmental factors, such as elevated educational pressure and less outdoors activities in some Asian countries, also affect the progression rates and treatment response [77]. Physiological Differences: East Asian populations have greater photopic pupil dilation on atropine treatment, compared to white Europeans related to iris pigmentation differences [77]. Overall Efficacy: Atropine retards myopia progression more effectively in Asian populations, especially in East Asian populations than in South Asian populations or white people [77]. Optimal Concentration: For East Asian children, 0.01% atropine is considered as effective while 0.05% atropine considered as optimal concentration [70], while in white European children, 0.01% atropine often does not show much effectiveness. The optimal concentration for Western children remains unclear [78, 79]. Side Effects: Side effects, such photophobia and reading difficulties, are more pronounced in Caucasian populations than Asian children [68]. Pupil Dilation: East Asian populations exhibit greater photopic pupil dilation by atropine compared to white European populations. This difference might be due to differences of iris pigmentation and genetic factors [77].
Comparative Efficacy of Different Atropine Concentrations in Major Clinical Trials
| Atropine Concentration (%) | Trial, Duration and Location | Key Characteristics | |
|---|---|---|---|
| 0.01% | LAMP (1 Year), Hong Kong [65] | n=438, After 1 year, the mean SE change was -0.27±0.61 D, with an increase in AL of 0.20±0.25 mm. The accommodation amplitude was reduced by 1.98±2.82 D. The pupil sizes under photopic and mesopic conditions were increased by 0.23±0.46 mm. All p<0.001. | |
| 0.01%-0.02% | U.S. Trial (3 Years) [71] | n=576, At month 36, 0.02% atropine slowed mean axial elongation (LSM difference, -0.08 mm; 95% CI, -0.13 mm to -0.02 mm; p = 0.005) while 0.01% atropine increased the responder proportion (OR, 4.54; 95% CI, 1.15-17.97; p = 0.03), slowed mean SER progression (LSM difference, 0.24 D; 95% CI, 0.11 D-0.37 D; p < 0.001), and slowed axial elongation (LSM difference, -0.13 mm; 95% CI, -0.19 mm to -0.07 mm; P < 0.001). Intermediate efficacy, Consistent effect over longer duration | |
| 0.025% | LAMP (1 Year) Hong Kong [65] | n=438, After 1 year, the mean SE change was -0.46±0.45 D, with a respective mean increase in AL of 0.29±0.20 mm. The accommodation amplitude was reduced by 1.61±2.61 D. The pupil sizes under photopic and mesopic conditions were increased respectively by 0.76±0.90 mm and 0.43±0.61 mm. All p<0.001. | |
| 0.03% | Simonaviciute et al. (1 Year) Lithuania [68] | n=72, After 1 year, a mean change by atropine in SE was -0.34 (0.44) D/year, lower than the -0.60 (0.50) D/year in the control group (p = 0.024). The change in AL was 0.19 (0.17) mm, compared to 0.31 (0.20) mm in the control group (p = 0.015). The atropine group had a significantly greater increase in ACD (p = 0.015). In total, 64.5% of patients in the atropine group showed progression <0.5 D/year, in contrast to 39.0% in the control group (p = 0.032). | |
| 0.01%, 0.05% | MOSAIC (3 Years) Ireland, Australia [67] | n=199, Compared with the group taking placebo then 0.05% atropine, the combined atropine then placebo groups had more spherical equivalent progression (adjusted difference, -0.13 diopters (D); 95% CI, -0.22 to -0.04 D; p = 0.01) and axial elongation (adjusted difference, 0.06 mm; 95% CI, 0.02-0.09 mm; p = 0.008), and the group taking 0.01% atropine then tapering 0.01% atropine had more axial elongation (adjusted difference, 0.04 mm; 95% CI, 0.009-0.07 mm; p = 0.04). | |
| 1.0% | ATOM1 (2 Years) Singapore [55] | n=346, After 2 years, the mean progression of myopia and of axial elongation in the placebo group was -1.20+/-0.69 D and 0.38+/-0.38 mm, respectively. In the atropine group, myopia progression was -0.28+/-0.92 D, whereas the axial length remained unchanged compared with baseline (-0.02+/-0.35 mm). The differences in myopia progression and axial elongation between the 2 groups were -0.92 D (95% confidence interval, -1.10 to -0.77 D; p<0.001) and 0.40 mm (95% confidence interval, 0.35-0.45 mm; p<0.001). | |
*Compared to switch from 0.01% to placebo; SE = Spherical Equivalent; AL = Axial Length
Notes: Eligibility Criteria for Above Trials/Investigation:
1. Eligibility criteria for LAMP (1 Year), Hong Kong [65] (atropine 0.01%, 0.025%) were children aged 4-12 years with myopic refraction > 1.0 D in both eyes, astigmatism of < 2.5 D, and documented myopic progression of > 0.5 D in the past one year were enrolled. Excluded were those also with ocular diseases (e.g., cataract, congenital retinal diseases, amblyopia, and strabismus), previous use of atropine, pirenzepine, orthokeratology lens, or other optical methods for myopia control, allergy to atropine, or systemic diseases (e.g., endocrine, cardiac, and respiratory diseases). 2. Eligibility criteria for U.S. Trial (3 Years) [71] were enrolled participants with -0.50 diopter (D) to -6.00 D spherical equivalent refractive error (SER) and no worse than -1.50 D astigmatism. 3. Eligibility criteria for Lithuania [68] were Caucasian children aged 6-12 years with a cycloplegic SE (sphere plus half of the cylinder power) refraction ranging between -1.0 D and -5.0 D and astigmatism and anisometropia <1.5 D. Exclusion criteria included prior use of atropine eye drops or other myopia progression control, congenital or chronic ocular diseases (e.g., cataract, congenital retinal diseases, amblyopia, strabismus), anisometropia or astigmatism exceeding 1.5 D, a history of ocular surgeries, and systemic conditions (e.g., cardiac, endocrinological and respiratory diseases, or chromosomal abnormalities). 4. Eligibility Criteria for MOSAIC (3 Years) Ireland, Australia [67] were eligibility criteria for MOSAIC Trial were age 6-16 years with spherical equivalent less than -0.50 diopters (D), no prior myopia therapy, no significant comorbidities, and no contraindications to atropine. 5. Eligibility criteria for ATOM1 (2 Years) Singapore [55] were recruitment of participants was from children aged 6-12 years, refractive error of spherical equivalent between 1.00 D to 6.00 D in each eye as measured by cycloplegic autorefraction Anisometropia of spherical equivalent less than or equal to 1.50 D as measured by cycloplegic autorefraction Astigmatism of < 1.50 D or better in both eyes with normal intraocular pressure of 21 mmHg with no history of cardiac or significant respiratory diseases, no allergy to atropine, cyclopentolate, proparacaine, and benzalkonium chloride, no previous or current use of contact lenses, bifocals, progressive addition lenses, or other forms of treatment including atropine, no amblyopia or manifest strabismus, including intermittent tropia willing and able to tolerate monocular cycloplegia and mydriasis.
4. Safety and Tolerability Profile
4.1 Ocular Adverse Effects
The safety profile of low-concentration atropine is generally favorable, particularly when compared to higher (0.5-1.0%) concentrations. However, several important considerations warrant attention:
Visual Symptoms: Pupillary dilation (typically 1-2 mm with 0.01%, increasing to 2-3 mm with 0.05%) can cause photophobia (light sensitivity) and impaired near vision due to reduced accommodation amplitude. In the MOSAIC trial, 15% of 0.05% atropine users reported near vision blur, and 8% reported photophobia, whereas these symptoms were uncommon with the 0.01% dose (3% and 0%, respectively) [67]. These effects are generally well-tolerated in children, particularly with photochromic lenses are used for outdoor activities and reading glasses are provided as needed for near work.
Intraocular Pressure (IOP) Concerns: Theoretical concerns exist regarding atropine's potential to elevate IOP, particularly through mechanisms involving trabecular meshwork obstruction by iris pigment released during pupillary movement or reduced trabecular tension secondary to ciliary muscle relaxation. However, a comprehensive 2024 review concluded that most studies show no clinically significant IOP changes in healthy children using low-concentration atropine [80]. Nevertheless, caution is warranted in children with ocular hypertension or strong family history of glaucoma, as isolated cases of IOP elevation have been reported, including a documented case of a 7-year-old Chinese child who developed elevated IOP after unsupervised use of low-concentration atropine [81].
Ocular Surface Effects: Allergic reactions (e.g., conjunctival hyperemia, itching, and lid swelling) occur in approximately 3-5% of users, more commonly with higher concentrations and preservatives like benzalkonium chloride. Preservative-free formulations may reduce this risk. In the 0.03% atropine study, 35% of participants reported adverse events (mostly mild allergic reactions), with 16% discontinuing treatment due to tolerability issues despite no serious adverse events [68].
4.2 Systemic Safety and Long-Term Considerations
The systemic absorption of low-concentration atropine is minimal, with serum concentrations typically below detection limits. Consequently, systemic anticholinergic effects (e.g., facial flushing, dry mouth, urinary retention, constipation, and tachycardia) are exceptionally rare at concentrations ≤ 0.05%. Long-term follow-up data extending 10-20 years from the ATOM cohort provide robust reassurance regarding long-term safety: no increased incidence of cataracts, glaucoma, retinal detachment, or other visually significant complications were observed in former atropine users compared to matched controls [64]. Similarly, European and Taiwanese studies that followed children using 0.01% atropine long-term found no serious adverse events attributable to treatment [82, 83].
Safety and Tolerability Profile of Low-Concentration Atropine
| Adverse Effect | 0.01% Atropine | 0.05% Atropine | Management Strategies |
|---|---|---|---|
| Photophobia (Light Sensitivity) | Uncommon (<5%) | Moderate (8-15%) | Photochromic lenses; sunglasses |
| Near Vision Blur / Accommodative Insufficiency | Mild (5-10%) | Moderate (15-25%) | Reading glasses for close work; monitor accommodative amplitude |
| Pupillary Dilation | Minimal (0.5-1.5 mm) | Moderate (2-3 mm) | Usually well-tolerated; cosmetic concerns rare |
| Allergic Conjunctivitis | 3-5% | 5-10% | Preservative-free formulations; switch to alternative concentration |
| Intraocular Pressure Elevation | Rare; no consistent pattern | Rare; isolated case reports | Baseline and periodic IOP checks; avoid glaucoma suspects |
| Systemic Effects | Extremely rare | Extremely rare | Nasolacrimal occlusion after instillation; wipe excess |
4.3 Safety Management Recommendations
To manage the common side effects of atropine eye drops for myopia control, practical strategies focus on timing the dosage and using optical aids like photochromic or bifocal lenses (reviewed in [84, 85]), including but not limiting: 1. Managing Photophobia (Light Sensitivity): Photophobia is a common side effect caused by pupil dilation, usually mild, especially in the case of low concentrations of atropine (for example, 0.01%). 2. Wear Sunglasses: They provide a simple way to reduce light sensitivity outdoors. 3. Use of Photochromic Lenses Ware: Spectacle lenses darken in bright daylight, helping protection of children in indoor and outdoor activities. 4. Tinted Glasses: For those sensitive to light, tinted glasses might be an option.
To manage near vision blur, use low-dose atropine to reduce amplitude of accommodation (reviewed in [84, 85]). 1. Alter Administration Time: Instilling the eye drops at bedtime is common for myopia control, which minimizes daytime blur as well as light sensitivity. 2. Near Addition Glasses: When near blur is an issue, prescribe glasses with a reading addition, such as bifocal or progressive lenses, to support near work. 3. Recommendation of Adequate Reading Distance: Encorage children to maintain an adequate reading distance and good lighting for near work. 4. Combine with Optical Treatments: Atropine can be used in combination with multifocal contact lenses or orthokeratology, which can address both distance and near vision needs.
For administration of atropine (reviewed in [85]), the methods include: 1. Proper Instillation: Use punctal occlusion technique (applying light pressure to inside corner of an eye for 2 minutes after instillation) to reduce systemic absorption as well as potential side effects. 2. Hand Hygiene: Wash hands before and after applying eye drops to prevent contamination. 3. Monitor and Adjust Follow-up Visits: For example, 3-6 months intervals are necessary to monitor efficacy, assess side effects, and adjust the atropine concentration or other management methods as needed. 4. Parental Help: Make sure that children are monitored when atropine drops are administered to ensure correct atropine eye drop application as well as safe storage.
5. Clinical Management Considerations
5.1 Patient Selection and Initiation Criteria
Appropriate patient selection is crucial for optimizing outcomes with atropine therapy. Ideal candidates typically include children aged 4-12 years with progressive myopia, defined as ≥ -0.50 D increase per year [86], particularly those with younger age of onset, family history of high myopia, or minimal time spent outdoors [87]. The 2024 Expert Consensus on low-concentration atropine recommends initiating treatment when progression exceeds 0.50 D/year, though some specialists advocate earlier intervention in rapidly progressing cases or children approaching high myopia thresholds [84]. Importantly, the landmark LAMP2 Trial demonstrated that 0.05% atropine significantly reduced myopia incidence (28.4% vs. 53.0% in placebo) when used preventatively in pre-myopic children with cycloplegic spherical equivalent between 0 and +1.00 D, suggesting potential for expanding indications to high-risk pre-myopes [22].
5.2 Practical Management Protocols
• Dosing Regimens: Once-daily evening instillation (one drop per eye) is standard, maximizing ocular contact time while minimizing light-related side effects. The MOSAIC Trial demonstrated excellent adherence (81%) with this regimen [67].
• Concentration Selection: Risk-stratified approaches are emerging: 0.05% may be preferred for rapid progressors (>1 D/year) or older children with less time for intervention [88, 89]; 0.03% offers intermediate efficacy without significant effects [68]; 0.01% provides maintenance therapy for young children without significant progression [90-92], such a concept is not all agreed [93]. Ethnic considerations may apply, with some evidence suggesting higher concentrations are more effective in Asian populations.
• Monitoring Protocol: Comprehensive baseline assessment should include cycloplegic refraction, axial length measurement, visual acuity, accommodative amplitude, pupil size, IOP measurement, and anterior and posterior segment examination. Follow-up evaluations every 3 months should assess efficacy (refraction, axial length), side effects (accommodative amplitude, pupil size), IOP, and adherence [94]. Optical coherence tomography (OCT) monitoring of retinal nerve fiber layer and macular thickness may be considered, though studies show no consistent atropine-related changes [95].
• Duration and Discontinuation: Treatment should ideally continue until mid-late adolescence (14-17 years), when myopia progression typically stabilizes. The ATOM follow-up demonstrated that early discontinuation (before age 14) is associated with greater rebound progression [57]. Gradual tapering strategies (e.g., reducing concentration or frequency) over 6-12 months may mitigate rebound effects, particularly after higher concentrations [74].
The management of myopia is summarized in Table 4:
Myopia Management Protocols
| Step | Myopia Management Stage | Key Actions and Protocols | |
|---|---|---|---|
| 1 | Patient Identification and Risk Assessment | Perform comprehensive eye exams, such as cycloplegic refraction, keratometry as well as axial length measurement to determine true axial myopia. | |
| Identify and record various risk factors such as age onset, parental history, ethnicity, and outdoor activities. | |||
| Educate parents and patients of risks of progressive myopia and goals of myopia management to slow down axial elongation. | |||
| 2 | Lifestyle and Environmental Adaptation | Encourage daily outdoor activities. | |
| Educate children of guidelines on near work, looking at distant objects from time to time, and increasing outdoors activities daily. | |||
| Educate pupils to use appropriate reading distances (>33 cm) as well as adequate lighting. | |||
| 3 | Treatment Plan | Choose a practical treatment strategy based on progression rate, children age, practical lifestyle, and binocular vision status. | |
| Treatment: daily low-concentration atropine eye drops, spectacle lenses, multifocal soft contact lenses, orthokeratology, and their combinations. | |||
| 4 | Monitor and Follow-up | Schedule follow-up visits (for example, every 6 months for progressing myopes) to monitor efficacy and compliance. | |
| Monitor treatment efficacy by axial length measurements as well as cycloplegic refraction. | |||
| Adjust the treatment when progression is off-target or when pronounced side effects occur. | |||
| 5 | Cessation and Tapering | Continue atropine treatment until myopia stabilizes, depending on individual variability in myopia stabilization. | |
| Tapering should be considered, and stabilization varies based on individual. | |||
5.3 Combination and Alternative Therapies
• Synergistic Approaches: Atropine demonstrates additive or synergistic effects when combined with optical interventions like orthokeratology (OK) or defocus-incorporated multiple segments (DIMS) spectacles [96-99]. Proposed protocols suggest instilling 0.01% atropine 5-15 minutes before OK lens insertion [100]. Combination therapy may be particularly beneficial for rapid progressors insufficiently controlled by monotherapy.
• Alternative Agents: For children intolerant to atropine, pirenzepine gel (a relatively selective M1/M4 antagonist) demonstrated efficacy in early trials with reduced cycloplegic effects [101]. 7-methylxanthine (7-MX), an adenosine receptor antagonist that increases scleral collagen synthesis, shows promise in oral formulation but remains investigational [102].
6 Unresolved Issues and Future Directions
6.1 Critical Knowledge Gaps
Despite substantial advances, several fundamental questions remain unresolved:
• Mechanistic Uncertainty: The precise cellular and molecular pathways through which atropine influences ocular growth require further elucidation. Particularly puzzling is the concentration paradox: how do minute concentrations insufficient for complete muscarinic blockade exert profound anti-myopic effects? Advanced techniques like single-cell RNA sequencing in primate models and proteomic analyses of human scleral fibroblasts may provide insights [40].
• Variable Treatment Response: Approximately 10-15% of children show minimal response ("non-responders") to atropine therapy [103]. Predictive biomarkers—potentially genetic variants in muscarinic receptors, dopamine pathways, or scleral remodeling enzymes—could enable personalized therapy. Pharmacogenomic studies embedded in large trials are underway.
• Rebound Phenomenon: The post-cessation acceleration of myopia progression remains management challenge, particularly with higher concentrations and abrupt discontinuation. The biological basis of rebound—whether compensatory growth signaling or unmasking of suppressed progression—requires clarification to develop mitigation strategies [74].
• Prevention vs. Control: The promising LAMP2 results for 0.05% atropine in pre-myopes raise important questions: can atropine truly prevent myopia or merely delay onset? Long-term follow-up beyond adolescence is needed to determine whether delayed onset translates to reduced high myopia prevalence. It also remains unclear for the mechanism of action of atropine for pre-myopia prevention. Although successful interventions are true, the precise mechanism of some treatments, such as low-dose atropine, remains unclear. There is no definite consensus on the best approach to prevent pre-myopia. When is the best to initiate, combine, and discontinue treatments remains unknown. In addition, the long-term effectiveness and safety of atropine for pre-myopia prevention requires further investigation. Large scale and long-term clinical trials are needed for pre-myopia research. Studies, such as ATOM3 Trial for low-dose atropine, are critical for the efficacy and safety of such interventions for pre-myopic children. A precise understanding of atropine's mechanisms of action in pre-myopia is needed to guide development of novel mechanism-based treatments. In addition, personalized pre-myopia prevention strategies, based on combined factors including age, genetics, environment, and control methods, are needed. New research-based guidelines and strategies for effective interventions are required to maximize efficacy of atropine on pre-myopia prevention. Furthermore, known risk factors of pre-myopia such as ethnicity, age, genetics, and outdoor time require further investigation.
Rank of atropine clinical readiness is listed as below:
Ranking Emerging Atropine Innovations by Clinical Readiness
| Rank | Innovation | Clinical Readiness & Evidence Level | Feasibility Discussion |
|---|---|---|---|
| 1 | Sustained-release systems | High Readiness. These systems are in advanced clinical development, including Phase 3 trials and ongoing research. They have strong foundational evidence in overcoming limitations of traditional drops, such as low bioavailability and poor compliance. | Highly feasible and a transformative frontier in myopia management. They leverage existing contact lens technology to provide consistent drug delivery, potentially improving efficacy and adherence. |
| 2 | AI-powered dosing | Moderate Readiness. While AI is not yet used for dosing, the concept of individualized treatment based on patient response (e.g., age, ethnicity, progression rate) is widely discussed and implemented in clinical practice. Clinical data drives these personalized decisions (e.g., adjusting concentration from 0.01% to 0.05%). | Feasible as it relies on existing clinical data and algorithms rather than a novel drug or device. The challenge lies in standardizing protocols and ensuring consistent data input for AI optimization. |
| 3 | Receptor-specific analogs | Early-Stage Research. These are primarily in animal models and in vitro studies, aiming to target specific muscarinic receptors (M1/M4) involved in eye growth without affecting accommodation (near vision) or pupil size. This research is still foundational. | Less clinically ready, as the specific compounds need extensive preclinical testing before entering human clinical trials. The primary challenge is identifying a compound with the desired specificity and safety profile. |
6.2 Emerging Innovations
Several promising developments may shape future clinical practice:
• Sustained-Release Delivery: Biodegradable implants, drug-eluting contact lenses, and punctal plug delivery systems could enhance adherence and provide steady drug levels, potentially improving efficacy and reducing peak-dose side effects. Early-phase clinical trials are evaluating a bimatoprost ring for sustained atropine delivery.
• Receptor-Specific Analogs: Compounds selectively targeting M1, M4, or dopamine D2 receptors may retain atropine's anti-myopic efficacy while minimizing unwanted effects like cycloplegia and mydriasis. Animal studies show promising results for compounds like pirenzepine and himbacine derivatives [104].
• Combination Formulations: Fixed-ratio combinations with dopamine agonists (e.g., apomorphine) or GABA enhancers might target multiple growth-signaling pathways simultaneously, potentially yielding synergistic effects that permit lower individual drug concentrations. Such approaches are in preclinical development.
• Personalized Concentration Titration: Adaptive dosing strategies using artificial intelligence algorithms that integrate individual progression rates, axial elongation velocity, and side effect profiles could optimize the benefit-risk ratio. Pilot implementation studies are exploring this precision medicine approach.
7. Conclusion
Low-concentration atropine has transformed the therapeutic landscape for childhood myopia, providing a safe, effective, and clinically practical intervention that slows axial elongation and reduces the risk of sight-threatening complications. The optimal concentration balances efficacy, tolerability, and rebound potential: 0.05% demonstrates superior efficacy in most trials, while 0.01% offers excellent tolerability with minimal rebound. Concentrations of 0.025-0.03% present promising intermediate options. Individualized management, taking into account age, progression rate, baseline refraction, and risk factors, remains essential. Ongoing research into mechanisms, alternative delivery systems, and combination therapies promises to further enhance outcomes. Ultimately, integrating atropine therapy with environmental modifications (e.g., increased outdoor time) and optical interventions offers the most comprehensive approach to combating the global myopia epidemic. As long-term safety data accumulate and novel treatment formulations emerge, atropine-based strategies will undoubtedly remain central to myopia management for the foreseeable future.
Acknowledgements
This work is supported by Basic Research Joint Special Project of the Department of Science and Technology, Yunnan Province and Kunming Medical University, China (Grant Number 202401AY070001-289, to Qin Zhu).
AI usage statement
During the preparation of this review article, artificial intelligence (AI) tools were used for assisting in drafting an outline of this article at the initiating stage, in language polishing, in grammatical improvement, and in readability enhancement. The AI did not contribute to the development of core arguments, literature selection, critical analysis, or the formation of original academic viewpoints. All intellectual content, scientific judgment, and final manuscript were completed and verified solely by the authors, who take full responsibility for the accuracy and academic integrity of this work.
Competing Interests
The authors have declared that no competing interest exists.
References
1. Shahzad I Mian M. Visual impairment in adults: Refractive disorders and presbyopia. Sep 17, 2025 ed. uptodate.com: Wolters Kluwer. 2025
2. David K Coats M, Evelyn A Paysse M. Refractive Errors in Children. Nov 13, 2025 ed. uptodate.com: UpToDate. 2025
3. Russo A, Boldini A, Romano D, Mazza G, Bignotti S, Morescalchi F. et al. Myopia: Mechanisms and Strategies to Slow Down Its Progression. J Ophthalmol. 2022;2022:1004977
4. Medina A. The cause of myopia development and progression: Theory, evidence, and treatment. Surv Ophthalmol. 2022;67:488-509
5. Sun L, Zhu L, Chen S, Li J, Li X, Wang K. et al. Mechanism of myopic defocus or atropine for myopia control: different or similar ways? Ophthalmic research. 2022: 65: 698-711.
6. Zhang XJ, Zaabaar E, French AN, Tang FY, Kam KW, Tham CC. et al. Advances in myopia control strategies for children. Br J Ophthalmol. 2025;109:165-76
7. Zhu Q, Chen L, Liang JR, Hu M, Xue LP, Zhou GL. et al. Retardation of myopia by atropine regimes. Int J Ophthalmol. 2024;17:2129-40
8. Surico PL, Parmar UPS, Singh RB, Farsi Y, Musa M, Maniaci A. et al. Myopia in Children: Epidemiology, Genetics, and Emerging Therapies for Treatment and Prevention. Children (Basel). 2024;11:1446
9. Organization WH. The Impact of Myopia and High Myopia In: Myopia JWHO-BHVIGSMo, editor. WHO Library: World Health Organization. 2016
10. Chen M, Wu A, Zhang L, Wang W, Chen X, Yu X. et al. The increasing prevalence of myopia and high myopia among high school students in Fenghua city, eastern China: a 15-year population-based survey. BMC Ophthalmol. 2018;18:159
11. Flitcroft DI. The complex interactions of retinal, optical and environmental factors in myopia aetiology. Prog Retin Eye Res. 2012;31:622-60
12. Williams K, Hammond C. High myopia and its risks. Community Eye Health. 2019;32:5-6
13. Xiang ZY, Zou HD. Recent Epidemiology Study Data of Myopia. J Ophthalmol. 2020;2020:4395278
14. Morgan IG, Jan CL. China Turns to School Reform to Control the Myopia Epidemic: A Narrative Review. Asia Pac J Ophthalmol (Phila). 2022;11:27-35
15. Zaabaar E, Zhang Y, Kam KW, Zhang XJ, Tham CC, Chen LJ. et al. Low-concentration atropine for controlling myopia onset and progression in East Asia. Asia Pac J Ophthalmol (Phila). 2024;13:100122
16. Sankaridurg P, Tahhan N, Kandel H, Naduvilath T, Zou H, Frick KD. et al. IMI Impact of Myopia. Investigative ophthalmology & visual science. 2021;62:2
17. Martinez-Albert N, Bueno-Gimeno I, Gene-Sampedro A. Risk Factors for Myopia: A Review. J Clin Med. 2023;12:6062
18. Chung YW, Park SH, Shin SY. Graduated atropine protocol effects on spherical equivalent and axial length in premyopic children. Sci Rep. 2025;15:15210
19. Fang PC, Chung MY, Yu HJ, Wu PC. Prevention of myopia onset with 0.025% atropine in premyopic children. Journal of ocular pharmacology and therapeutics: the official journal of the Association for Ocular Pharmacology and Therapeutics. 2010;26:341-5
20. Jethani J. Efficacy of low-concentration atropine (0.01%) eye drops for prevention of axial myopic progression in premyopes. Indian J Ophthalmol. 2022;70:238-40
21. Wang W, Zhang F, Yu S, Ma N, Huang C, Wang M. et al. Prevention of myopia shift and myopia onset using 0.01% atropine in premyopic children - a prospective, randomized, double-masked, and crossover trial. Eur J Pediatr. 2023;182:2597-606
22. Yam JC, Zhang XJ, Zhang Y, Yip BHK, Tang F, Wong ES. et al. Effect of Low-Concentration Atropine Eyedrops vs Placebo on Myopia Incidence in Children: The LAMP2 Randomized Clinical Trial. JAMA. 2023;329:472-81
23. Carlson AB, Kraus GP. Physiology, Cholinergic Receptors. StatPearls. Treasure Island (FL) ineligible companies. Disclosure: Gregory Kraus declares no relevant financial relationships with ineligible companies. 2025
24. Haga T. Molecular properties of muscarinic acetylcholine receptors. Proc Jpn Acad Ser B Phys Biol Sci. 2013;89:226-56
25. Lawrenson JG, Shah R, Huntjens B, Downie LE, Virgili G, Dhakal R. et al. Interventions for myopia control in children: a living systematic review and network meta-analysis. The Cochrane database of systematic reviews. 2023;2:CD014758
26. Upadhyay A, Beuerman RW. Biological Mechanisms of Atropine Control of Myopia. Eye & contact lens. 2020;46:129-35
27. Barathi VA, Beuerman RW. Molecular mechanisms of muscarinic receptors in mouse scleral fibroblasts: Prior to and after induction of experimental myopia with atropine treatment. Mol Vis. 2011;17:680-92
28. Tao YX, Conn PM. Chaperoning G protein-coupled receptors: from cell biology to therapeutics. Endocr Rev. 2014;35:602-47
29. Kudlak M, Tadi P. Physiology, Muscarinic Receptor. StatPearls. Treasure Island (FL) ineligible companies. Disclosure: Prasanna Tadi declares no relevant financial relationships with ineligible companies. 2025
30. North RV, Kelly ME. A review of the uses and adverse effects of topical administration of atropine. Ophthalmic & physiological optics: the journal of the British College of Ophthalmic Opticians (Optometrists). 1987;7:109-14
31. Cristaldi M, Olivieri M, Pezzino S, Spampinato G, Lupo G, Anfuso CD. et al. Atropine Differentially Modulates ECM Production by Ocular Fibroblasts, and Its Ocular Surface Toxicity Is Blunted by Colostrum. Biomedicines. 2020;8:78
32. Zadnik K, Mutti DO. How applicable are animal myopia models to human juvenile onset myopia? Vision Res. 1995;35:1283-8
33. Avetisov ES, Savitskaya NF, Vinetskaya MI, Iomdina EN. A study of biochemical and biomechanical qualities of normal and myopic eye sclera in humans of different age groups. Metab Pediatr Syst Ophthalmol. 1983;7:183-8
34. McBrien NA, Lawlor P, Gentle A. Scleral remodeling during the development of and recovery from axial myopia in the tree shrew. Investigative ophthalmology & visual science. 2000;41:3713-9
35. McBrien NA, Jobling AI, Gentle A. Biomechanics of the sclera in myopia: extracellular and cellular factors. Optom Vis Sci. 2009;86:E23-30
36. McBrien NA, Gentle A. Role of the sclera in the development and pathological complications of myopia. Prog Retin Eye Res. 2003;22:307-38
37. Norton TT, Rada JA. Reduced extracellular matrix in mammalian sclera with induced myopia. Vision Res. 1995;35:1271-81
38. Curtin BJ. Physiopathologic aspects of scleral stress-strain. Trans Am Ophthalmol Soc. 1969;67:417-61
39. Yu Q, Zhou JB. Scleral remodeling in myopia development. Int J Ophthalmol. 2022;15:510-4
40. Huang L, Zhang J, Luo Y. The role of atropine in myopia control: insights into choroidal and scleral mechanisms. Front Pharmacol. 2025;16:1509196
41. Hansen NC, Hvid-Hansen A, Bek T, Moller F, Jacobsen N, Kessel L. The Macular Choroidal Thickness in Danish Children with Myopia After Two-Year Low-Dose Atropine and One-Year Wash-Out: A Placebo-Controlled, Randomized Clinical Trial. Ophthalmol Ther. 2024;13:3111-22
42. Barathi VA, Chaurasia SS, Poidinger M, Koh SK, Tian D, Ho C. et al. Involvement of GABA transporters in atropine-treated myopic retina as revealed by iTRAQ quantitative proteomics. J Proteome Res. 2014;13:4647-58
43. Horn D, Salzano AD, Jenewein EC, Weise KK, Schaeffel F, Mathis U. et al. Topical review: Potential mechanisms of atropine for myopia control. Optom Vis Sci. 2025;102:260-70
44. Kaiti R, Shyangbo R, Sharma IP. Role of Atropine in the control of Myopia Progression- A Review. Beyoglu Eye J. 2022;7:157-66
45. Buels KS, Fryer AD. Muscarinic receptor antagonists: effects on pulmonary function. Handb Exp Pharmacol. 2012:317-41
46. Wall SJ, Yasuda RP, Li M, Ciesla W, Wolfe BB. The ontogeny of m1-m5 muscarinic receptor subtypes in rat forebrain. Brain research Developmental brain research. 1992;66:181-5
47. Cembala TM, Forde SC, Appadu BL, Lambert DG. Allosteric interaction of the neuromuscular blockers vecuronium and pancuronium with recombinant human muscarinic M2 receptors. Eur J Pharmacol. 2007;569:37-40
48. Nelson CP, Nahorski SR, Challiss RA. Constitutive activity and inverse agonism at the M2 muscarinic acetylcholine receptor. J Pharmacol Exp Ther. 2006;316:279-88
49. McBrien NA, Metlapally R, Jobling AI, Gentle A. Expression of collagen-binding integrin receptors in the mammalian sclera and their regulation during the development of myopia. Investigative ophthalmology & visual science. 2006;47:4674-82
50. Li X, Hu J, Peng Z, Chen S, Sun L, Wang K. et al. Association between choriocapillaris perfusion and axial elongation in children using defocus incorporated multiple segments (DIMS) spectacle lenses. Eye (Lond). 2023;37:3847-53
51. Boulamery A, Simon N, Vidal J, Bruguerolle B. Effects of L-Dopa on circadian rhythms of 6-OHDA striatal lesioned rats: a radiotelemetric study. Chronobiol Int. 2010;27:251-64
52. Kutty RK, Samuel W, Boyce K, Cherukuri A, Duncan T, Jaworski C. et al. Proinflammatory cytokines decrease the expression of genes critical for RPE function. Mol Vis. 2016;22:1156-68
53. Chen B, Ni Y, Chen J, Xing S, Zhang Z. Comparison of different concentrations atropine in controlling children and adolescent myopia: an umbrella review of systematic reviews and meta-analyses. Front Ophthalmol (Lausanne). 2024;4:1447558
54. Chia A, Chua WH, Cheung YB, Wong WL, Lingham A, Fong A. et al. Atropine for the treatment of childhood myopia: safety and efficacy of 0.5%, 0.1%, and 0.01% doses (Atropine for the Treatment of Myopia 2). Ophthalmology. 2012;119:347-54
55. Chua WH, Balakrishnan V, Chan YH, Tong L, Ling Y, Quah BL. et al. Atropine for the treatment of childhood myopia. Ophthalmology. 2006;113:2285-91
56. Tong L, Huang XL, Koh AL, Zhang X, Tan DT, Chua WH. Atropine for the treatment of childhood myopia: effect on myopia progression after cessation of atropine. Ophthalmology. 2009;116:572-9
57. Chia A, Lu QS, Tan D. Five-Year Clinical Trial on Atropine for the Treatment of Myopia 2: Myopia Control with Atropine 0.01% Eyedrops. Ophthalmology. 2016;123:391-9
58. Wu P-C, Chuang M-N, Choi J. et al. Update in myopia and treatment strategy of atropine use in myopia control. Eye (Lond). 2018;33:3-13
59. Yen MY, Liu JH, Kao SC, Shiao CH. Comparison of the effect of atropine and cyclopentolate on myopia. Ann Ophthalmol. 1989;21:180-2
60. Shih YF, Hsiao CK, Chen CJ, Chang CW, Hung PT, Lin LL. An intervention trial on efficacy of atropine and multi-focal glasses in controlling myopic progression. Acta ophthalmologica Scandinavica. 2001;79:233-6
61. Yi S, Huang Y, Yu SZ, Chen XJ, Yi H, Zeng XL. Therapeutic effect of atropine 1% in children with low myopia. Journal of AAPOS: the official publication of the American Association for Pediatric Ophthalmology and Strabismus / American Association for Pediatric Ophthalmology and Strabismus. 2015;19:426-9
62. Wang YR, Bian HL, Wang Q. Atropine 0.5% eyedrops for the treatment of children with low myopia: A randomized controlled trial. Medicine (Baltimore). 2017;96:e7371
63. Zhu Q, Tang Y, Guo L, Tighe S, Zhou Y, Zhang X. et al. Efficacy and Safety of 1% Atropine on Retardation of Moderate Myopia Progression in Chinese School Children. Int J Med Sci. 2020;17:176-81
64. Li Y, Yip M, Ning Y, Chung J, Toh A, Leow C. et al. Topical Atropine for Childhood Myopia Control: The Atropine Treatment Long-Term Assessment Study. JAMA Ophthalmol. 2024;142:15-23
65. Yam JC, Jiang Y, Tang SM, Law AKP, Chan JJ, Wong E. et al. Low-Concentration Atropine for Myopia Progression (LAMP) Study: A Randomized, Double-Blinded, Placebo-Controlled Trial of 0.05%, 0.025%, and 0.01% Atropine Eye Drops in Myopia Control. Ophthalmology. 2019;126:113-24
66. Zhang XJ, Zhang Y, Yip BHK, Kam KW, Tang F, Ling X. et al. Five-Year Clinical Trial of Low-concentration Atropine for Myopia Progression (LAMP) Study: Phase 4 Report. Ophthalmology. 2024;131:1011-20
67. Loughman J, Lingham G, Nkansah EK, Kobia-Acquah E, Flitcroft DI. Efficacy and Safety of Different Atropine Regimens for the Treatment of Myopia in Children: Three-Year Results of the MOSAIC Randomized Clinical Trial. JAMA Ophthalmol. 2025;143:134-44
68. Simonaviciute D, Grzybowski A, Gelzinis A, Zemaitiene R. Efficacy and Safety of 0.03% Atropine Eye Drops in Controlling Myopia Progression: A One-Year Prospective Clinical Study. J Clin Med. 2024;13:3218
69. Kumaran A, Htoon HM, Tan D, Chia A. Analysis of Changes in Refraction and Biometry of Atropine- and Placebo-Treated Eyes. Investigative ophthalmology & visual science. 2015;56:5650-5
70. Li FF, Yam JC. Low-Concentration Atropine Eye Drops for Myopia Progression. Asia Pac J Ophthalmol (Phila). 2019;8:360-5
71. Zadnik K, Schulman E, Flitcroft I, Fogt JS, Blumenfeld LC, Fong TM. et al. Efficacy and Safety of 0.01% and 0.02% Atropine for the Treatment of Pediatric Myopia Progression Over 3 Years: A Randomized Clinical Trial. JAMA Ophthalmol. 2023;141:990-9
72. Biswas S, Lanca C, Kynman-Sprinks D, Grzybowski A. Ethnic and dose-dependent differences in atropine efficacy for myopia control: a systematic review and meta-analysis. Br J Ophthalmol. 2025 Nov 10:bjo-2025-328518. doi: 10.1136/bjo-2025-328518
73. Wang X, Zhang L, Gan J, Wang Y, Yang W. Effectiveness of various atropine concentrations in myopia control for Asian children: a network meta-analysis. Front Pharmacol. 2024;15:1503536
74. Yam JC, Khanal S, Phillips JR. Does 0.01% atropine have a place as a myopia control therapy? Ophthalmic & physiological optics: the journal of the British College of Ophthalmic Opticians (Optometrists). 2025;45:929-35
75. Maulvi FA, Desai DT, Kalaiselvan P, Shah DO, Willcox MDP. Current and emerging strategies for myopia control: a narrative review of optical, pharmacological, behavioural, and adjunctive therapies. Eye (Lond). 2025;39:2635-44
76. Zhang X, Wang Y, Zhou X, Qu X. Analysis of Factors That May Affect the Effect of Atropine 0.01% on Myopia Control. Front Pharmacol. 2020;11:01081
77. Biswas S, El Kareh A, Qureshi M, Lee DMX, Sun CH, Lam JSH. et al. The influence of the environment and lifestyle on myopia. J Physiol Anthropol. 2024;43:7
78. Loughman J, Kobia-Acquah E, Lingham G, Butler J, Loskutova E, Mackey DA. et al. Myopia outcome study of atropine in children: Two-year result of daily 0.01% atropine in a European population. Acta Ophthalmol. 2024;102:e245-e56
79. Navarra P, Buzzonetti L, Amico V, Cro M, Federico B. A systematic review with meta-analysis on the efficacy of 0.01% atropine eyedrops in preventing myopia progression in worldwide children's populations. Front Pharmacol. 2025;16:1497667
80. Chen PJ, Hsia Y, Tsai TH, Su CC, Huang JY, Wang TH. Impact of atropine use for myopia control on intraocular pressure in children: A comprehensive review including postpupil dilation intraocular pressure changes. Taiwan J Ophthalmol. 2024;14:179-89
81. Li S, Li SM, Wang XL, Kang MT, Liu LR, Li H. et al. Distribution and associations of intraocular pressure in 7- and 12-year-old Chinese children: The Anyang Childhood Eye Study. PloS one. 2017;12:e0181922
82. Moriche-Carretero M, Revilla-Amores R, Gutierrez-Blanco A, Moreno-Morillo FJ, Martinez-Perez C, Sanchez-Tena MA. et al. Five-year results of atropine 0.01% efficacy in the myopia control in a European population. Br J Ophthalmol. 2024;108:715-9
83. Kuo HY, Ke CH, Chen ST, Sun HY. The Impact of Clinical Atropine Use in Taiwanese Schoolchildren: Changes in Physiological Characteristics and Visual Functions. Children (Basel). 2021;8:1054
84. Li L. Interpretation of expert consensus on the application of low-concentration atropine eye drops in the prevention and control of myopia in children and adolescents. Pediatr Investig. 2025;9:107-13
85. Zhao C, Cai C, Ding Q, Dai H. Efficacy and safety of atropine to control myopia progression: a systematic review and meta-analysis. BMC ophthalmology. 2020;20:478
86. Tricard D, Marillet S, Ingrand P, Bullimore MA, Bourne RRA, Leveziel N. Progression of myopia in children and teenagers: a nationwide longitudinal study. Br J Ophthalmol. 2022;106:1104-9
87. Recko M, Stahl ED. Childhood myopia: epidemiology, risk factors, and prevention. Mo Med. 2015;112:116-21
88. Erdinest N, Atar-Vardi M, London N, Landau D, Smadja D, Pras E. et al. Treatment of Rapid Progression of Myopia: Topical Atropine 0.05% and MF60 Contact Lenses. Vision (Basel). 2024;8:3
89. Zhu Q, Tang GY, Hua ZJ, Xue LP, Zhou Y, Zhang JY. et al. 0.05% atropine on control of myopia progression in Chinese school children: a randomized 3-year clinical trial. Int J Ophthalmol. 2023;16:939-46
90. Wang Z, Li T, Zuo X, Zhang T, Liu L, Zhou C. et al. 0.01% Atropine Eye Drops in Children With Myopia and Intermittent Exotropia: The AMIXT Randomized Clinical Trial. JAMA Ophthalmol. 2024;142:722-30
91. Rajavi Z, Kheiri B, Sheibani K, Sabbaghi H. Myopia Controlling using Low Dose Atropine Eye Drop. J Curr Ophthalmol. 2024;36:82-8
92. Simonaviciute D, Grzybowski A, Lanca C, Pang CP, Gelzinis A, Zemaitiene R. The Effectiveness and Tolerability of Atropine Eye Drops for Myopia Control in Non-Asian Regions. J Clin Med. 2023;12:2314
93. Repka MX, Weise KK, Chandler DL, Wu R, Melia BM, Manny RE. et al. Low-Dose 0.01% Atropine Eye Drops vs Placebo for Myopia Control: A Randomized Clinical Trial. JAMA Ophthalmol. 2023;141:756-65
94. Gifford KL, Richdale K, Kang P, Aller TA, Lam CS, Liu YM. et al. IMI - Clinical Management Guidelines Report. Investigative ophthalmology & visual science. 2019;60:M184-M203
95. Enaholo ES, Musa MJ, Zeppieri M. Optical Coherence Tomography. StatPearls. Treasure Island (FL) ineligible companies. Disclosure: Mutali Musa declares no relevant financial relationships with ineligible companies. Disclosure: Marco Zeppieri declares no relevant financial relationships with ineligible companies. 2025
96. Yang Y, Xiao Z, Ouyang J, Guo Y. Synergistic efficacy of orthokeratology and 0.01% atropine in controlling pediatric myopia progression: A retrospective analysis. J Investig Med. 2025;73:290-9
97. Yum HR, Han SY, Park SH, Shin SY. Synergistic Effect of Dual-Focus Soft Contact Lenses and 0.05% Atropine on Myopia Control in Children With Rapidly Progressing Myopia. Eye & contact lens. 2025;51:92-7
98. Xu S, Li Z, Zhao W, Zheng B, Jiang J, Ye G. et al. Effect of atropine, orthokeratology and combined treatments for myopia control: a 2-year stratified randomised clinical trial. Br J Ophthalmol. 2023;107:1812-7
99. Nucci P, Lembo A, Schiavetti I, Shah R, Edgar DF, Evans BJW. A comparison of myopia control in European children and adolescents with defocus incorporated multiple segments (DIMS) spectacles, atropine, and combined DIMS/atropine. PloS one. 2023;18:e0281816
100. Tsai HR, Wang JH, Huang HK, Chen TL, Chen PW, Chiu CJ. Efficacy of atropine, orthokeratology, and combined atropine with orthokeratology for childhood myopia: A systematic review and network meta-analysis. J Formos Med Assoc. 2022;121:2490-500
101. Siatkowski RM, Cotter S, Miller JM, Scher CA, Crockett RS, Novack GD. et al. Safety and efficacy of 2% pirenzepine ophthalmic gel in children with myopia: a 1-year, multicenter, double-masked, placebo-controlled parallel study. Arch Ophthalmol. 2004;122:1667-74
102. Lai L, Trier K, Cui DM. Role of 7-methylxanthine in myopia prevention and control: a mini-review. Int J Ophthalmol. 2023;16:969-76
103. Wu PC, Chuang MN, Choi J, Chen H, Wu G, Ohno-Matsui K. et al. Update in myopia and treatment strategy of atropine use in myopia control. Eye (Lond). 2019;33:3-13
104. Arumugam B, McBrien NA. Muscarinic antagonist control of myopia: evidence for M4 and M1 receptor-based pathways in the inhibition of experimentally-induced axial myopia in the tree shrew. Investigative ophthalmology & visual science. 2012;53:5827-37
Author contact
Corresponding authors: Yuezu Li, Department of Refractive Surgery Center, Affiliated Hospital of Yunnan University; Kunming 650021, China; Telephone: 0118615925166452; Fax: 011860871-65156650; E-mail: 267089197com. Yingting Zhu, Research and Development Department, BioTissue (TissueTech, Inc.), 7230 Corporate Center Drive, Suite B, Miami, FL 33126, USA. Telephone: (786) 456-7632; Fax: (305) 274-1297; E-mail: yzhucom

 Global reach, higher impact
Global reach, higher impact