Impact Factor
ISSN: 1449-1907
Int J Med Sci 2022; 19(8):1290-1299. doi:10.7150/ijms.73645 This issue Cite
Research Paper
A proprietary blend of standardized Punica granatum fruit rind and Theobroma cocoa seed extracts mitigates aging males' symptoms: A randomized, double-blind, placebo-controlled study
1. Shetty's Hospital, Kaveri Nagar, Bengaluru-560068, Karnataka, India.
2. Narayana Hrudayalaya, Bommasandra Industrial Area, Bengaluru-560068, Karnataka, India.
3. D2L Clinical Solutions, Sadaramangala Industrial Area, Bengaluru-560048, Karnataka, India.
4. Sri Venkateshwara Hospital, Madiwala, Bengaluru-560068, Karnataka, India.
Abstract
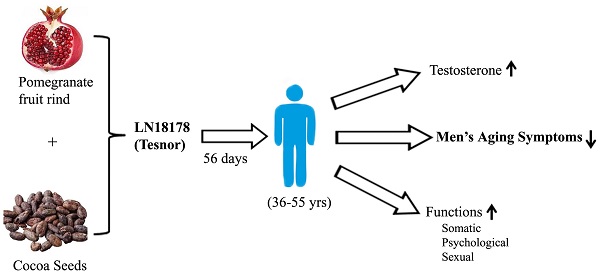
Objective: We evaluated the safety and efficacy of a novel combination of Punica granatum fruit rind and Theobroma cocoa seed extracts (LN18178 or Tesnor®) in enhancing serum testosterone level and reducing aging males' symptoms (AMS) in a randomized, double-blind, placebo-controlled investigation (CTRI Reg. No. CTRI/2019/02/017506).
Methods: One hundred twenty healthy male participants (age 36-55 years) were randomized (n=40) to placebo, 200 or 400 mg of LN18178 for a period of fifty-six consecutive days of supplementation. The primary efficacy parameter was the AMS score. The secondary parameters were serum testosterone (free and total) levels, hand-grip strength, and perceived stress scale (PSS-10) score. Measurement of other hormones included in the study was serum dihydrotestosterone (DHT), cortisol, and 17β-estradiol (E2). Total blood chemistry parameters, vital signs, and urinalysis were parts of the safety assessment.
Results: Both doses of LN18178 significantly reduced the mean AMS scores after 56 days of supplementation. Furthermore, it significantly improved general, psychological, and sexual well-being. Serum levels of free testosterone and total testosterone levels were significantly increased in LN18178 supplemented (200 and 400 mg) participants compared to the baseline and placebo. Significant improvements in hand-grip strength and reduced PSS-10 scores were also observed.
Conclusion: LN18178 supplementation reduced AMS scores and improved sexual performance. Also, LN18178 groups exhibited superior muscular strength and reduction in perceived stress. Total blood chemistry and urine analysis demonstrated the broad-spectrum safety.
Keywords: Aging males' symptoms, LN18178, Muscular strength, Tesnor®, Testosterone, Stress alleviation

 Global reach, higher impact
Global reach, higher impact