Impact Factor ISSN: 1449-1907
Int J Med Sci 2024; 21(3):483-491. doi:10.7150/ijms.89901 This issue Cite
Research Paper
Transcription factor 21 rs12190287 polymorphism is related to stable angina and ST elevation myocardial infarction in a Chinese Population
1. Division of Cardiology, Department of Internal Medicine, E-Da Hospital, I-Shou University, Kaohsiung 82445 Taiwan.
2. School of Medicine, College of Medicine, I-Shou University, Kaohsiung, 82445 Taiwan.
3. School of Medicine for International Students, College of Medicine, I-Shou University, Kaohsiung, 82445 Taiwan.
4. Department of Emergency, E-Da Hospital, I-Shou University, Kaohsiung 82445 Taiwan.
5. Division of Cardiology, Department of Internal Medicine, E-Da Dachang Hospital, I-Shou University, Kaohsiung 807066, Taiwan.
6. Division of Endocrinology and Metabolism, Department of Internal Medicine, E-Da Hospital, I-Shou University, Kaohsiung 82445 Taiwan.
7. Division of Cardiology, Department of Internal Medicine, Taipei Veterans General Hospital, Yuli Branch, Hualien 98142 Taiwan.
8. Faculty of Medicine, School of Medicine, National Yang Ming Chiao Tung University, Taipei 112304 Taiwan.
9. Division of General Surgery, Department of Surgery, E-Da Hospital, I-Shou University, Kaohsiung 82445 Taiwan.
10. The School of Chinese Medicine for Post Baccalaureate, College of Medicine, I-Shou University, Kaohsiung 82445 Taiwan.
11. Lee's Endocrinologic Clinic, Pingtung 90000 Taiwan.
12. Division of Cardiology, Department of Internal Medicine, E-Da Cancer Hospital, I-Shou University, Kaohsiung 82445 Taiwan.
Received 2023-9-6; Accepted 2023-12-20; Published 2024-1-1
Abstract
Background: Transcription factor 21 (TCF21, epicardin, capsuling, pod-1) is expressed in the epicardium and is involved in the regulation of cell fate and differentiation via epithelial-mesenchymal transformation during development of the heart. In addition, TCF21 can suppress the differentiation of epicardial cells into vascular smooth muscle cells and promote cardiac fibroblast development. This study aimed to explore whether TCF21 gene (12190287G/C) variants affect coronary artery disease risk.
Methods: We enrolled 381 patients who had stable angina, 138 with ST elevation myocardial infarction (STEMI), and 276 healthy subjects. Genotyping of rs12190287 of the TCF21 gene was performed.
Results: Higher frequencies of the CC genotype were found in the patients with stable angina/STEMI than in the healthy controls. After adjusting for diabetes mellitus, hypertension, age, sex, smoking, body mass index and hyperlipidemia, the patients with the CC genotype of the TCF21 gene were associated with 2.49- and 9.19-fold increased risks of stable angina and STEMI, respectively, compared to the patients with the GG genotype. Furthermore, TCF21 CC genotypes showed positive correlations with both stable angina and STEMI, whereas TCF21 GG genotypes exhibited a negative correlation with STEMI. Moreover, the stable angina and STEMI patients with the CC genotype had significantly elevated high-sensitivity C-reactive protein levels than those with the GG genotype. In addition, significant associations were found between type 2 diabetes mellitus, hypertension, and hyperlipidemia with TCF21 gene polymorphisms (p for trend < 0.05).
Conclusion: TCF21 gene polymorphisms may increase susceptibility to stable angina and STEMI.
Keywords: transcription factor 21, polymorphism, stable angina, ST elevation myocardial infarction
Introduction
Coronary artery disease (CAD) is still a leading cause of death in industrialized countries even though advances in its prevention and treatment have been made. In the United States, over 18.2 million people have CAD [1,2]; in Europe, CAD results in over 3.9 million deaths [3]. In Taiwan, more than 17,000 people die of CAD each year [4]. Many risk factors contribute to the pathogenesis of CAD, including both modifiable (physical inactivity, diabetes, smoking, hypertension, stress, hypercholesterolemia, and obesity) and non-modifiable (gender, age, race, family history of CAD, and genotype) risk factors [5-10]. Behavioral as well as metabolic and environmental risks are leading cause of CAD development [11]. Many studies have demonstrated the role of genetic polymorphisms in CAD development, suggesting that a combination of genetic and environmental factors may increase susceptibility to CAD [10,12,13].
Previous genome-wide association studies have reported associations between common variants with the risks of myocardial infarction and CAD [14]. Transcription factor 21 (TCF21), a basic helix-loop-helix transcription factor family member, has been demonstrated to play key roles in cell fate and differentiation [15]. TCF21 lineage- traced cells were shown to transform into periostin-expressing myofibroblasts in a mice model of myocardial infarction, a process essential to promote adaptive fibrosis and facilitate cardiac healing [16]. On the other hand, a lack of cardiac fibroblast production and cardiac repair have been demonstrated in TCF21-deficient mice [17]. Furthermore, in a cardiac fibroblast zebrafish model, TCF21 was shown to both contribute to a fibrotic response after injury and also cardiomyocyte proliferation during regeneration [18]. In humans of various ethnicity, a higher risk of CAD has been associated with rs12190287, a single nucleotide polymorphism (SNP) located in the 3′untranslated region of TCF21 [19-21]. Hamed et al. indicated that the CC genotype and C allele of TCF21 (12190287G/C) polymorphisms may be genetic risk factors for CAD [22]. Furthermore, in a Chinese population, the TCF21 rs12190287 polymorphism was also shown to confer susceptibility to ventricular septal defects [23]. Moreover, a Japanese study reported that TCF21 rs12190287 (G→C) may be a susceptibility locus for hypertension [24]. Another study found significant associations between the GG genotype and G allele of rs12190287 in TCF21 and an increased TCF21 concentration with the onset and recurrence of paroxysmal atrial fibrillation post ablation [25]. Considering these findings, study aimed to further explore whether TCF21 genetic polymorphisms affected susceptibility to stable angina and ST elevation myocardial infarction (STEMI) and their relationships with clinical and biochemical characteristics.
Methods
Participants
Patients with stable angina (n=381) and STEMI (n = 138) were enrolled from the Emergency Room and Cardiovascular Ward at Kaohsiung E-Da Hospital, Taiwan, from January to December 2022. STEMI was identified according to prolonged chest pain (more than 30 minutes), typical increases in circulating biochemical markers (troponin-I and creatine kinase-MB (CK-MB)/creatine-phospho-kinase) with symptoms of ischemia persisting for ≥ 30 min, and ST-segment elevation ≥ 2.0 mm in two or more contiguous electrocardiogram leads. Stable angina was identified as patients with rest or effort-related chest pain which had not progressed in the last 6 months. All of the patients with STEMI underwent primary percutaneous coronary interventions within 12 hours of symptom onset. Patients with renal dysfunction, hepatic dysfunction, heart failure and valvular heart disease were excluded. Healthy controls (n = 276) were unrelated patients who entered the health examination program of the hospital. None the controls had detectable cardiovascular risk factors or were not taking any medication. The Human Research Ethics Committee of E-Da Hospital gave approval for the study, and all of the enrolled patients signed informed consent forms.
Data collection
All of the enrolled participants lived in southern Taiwan and were of Han Chinese origin. Data on smoking, drinking, and personal disease history were obtained using standardized questionnaires. Smoking and alcohol drinking status were classified as never, former (stopped for at least 1 year), or current. We analyzed former and current drinkers as one group [26]. Body mass index (BMI) and waist (between the lowest rib and top of the hip) and hip (at the widest part) circumferences were measured in all of the patients, and the average of two measurements was used to calculate the waist-to-hip ratio. A digital sphygmomanometer (HEM-907, Omron, Japan) was used to measure blood pressure (BP) after a 5-min rest. The modified Simpson's method was used to compute left ventricular ejection fraction with apical 4-chamber views [27]. The ATP III criteria were used to diagnose hyperlipidemia as the presence of one of the following: (1) total cholesterol ≥ 200 mg/dL, (2) low-density lipoprotein-cholesterol (LDL-C) ≥ 130 mg/dL, (3) triglyceride level ≥ 150 mg/dL, (4) high-density lipoprotein-cholesterol (HDL-C) < 35/39 mg/dL (men/women), and (5) prescriptions for lipid disorder medications [28]. Patients were defined as being hypertensive if they had prescriptions for anti-hypertension medications, systolic BP (SBP) ≥ 140 mmHg, or diastolic BP (DBP) ≥ 90 mmHg. Patients were defined as having diabetes mellitus if they had prescriptions for anti-diabetes medications or a fasting glucose level > 126 mg/dl [29].
Laboratory measurements
Levels of total cholesterol, LDL-C, complete blood cell count, serum triglycerides, HDL-C, albumin, blood urine nitrogen (BUN), uric acid, and glucose were measured from fasting blood samples [30]. The Jaffe method was used to calculate serum creatinine. Hemoglobin A1c was measured using an automated analyzer (HLC-723G8, Tosoh Corp., Tokyo, Japan). CK-MB and troponin I serum levels were measured using chemiluminescent microparticle immunoassays. Total leukocyte count and the lymphocyte, neutrophil and monocytes proportions were measured using an automated cell counter (XE-2100 Hematology Alpha Transportation System; Sysmex, Kobe, Japan). Absolute leukocyte subtype counts were calculated as the product of its proportion and total leukocyte count. The CKD-EPI formula was utilized to determine estimated glomerular filtration rate (eGFR) [31]. An immunochemistry system (Beckman Coulter IMMAGE) was used to evaluate plasma levels of high-sensitivity C-reactive protein (hs-CRP). The detection limit was 0.2 mg/L, and all measurements were made twice.
Determining the TCF21 genotype (rs12190287)
A QIAamp DNA Blood Mini kit (Qiagen, Valencia, CA) was used to extract genomic DNA from whole blood samples following the manufacturer's protocol. DNA was reconstituted in TE buffer [1 mM EDTA, 10 mM Tris (pH 7.8)], quantified using OD260 measurements, and stored at -20°C until use as a PCR template. Genotyping of rs12190287 of the TCF21 gene was performed by TagMan® SNP Genotyping Assay (Thermo Fisher Scientific) using an Applied Biosystems PRISM7900 Sequence Detection System. In brief, 5 ng of genomic DNA was amplified in a reaction volume of 10 μl containing 0.25 ml of 40× Predesigned TaqMan SNP Genotyping assay (FAM™ dye labeled MGB probe + VIC® dye-labeled MGB probe + 2 unlabeled primers), 5 ml of 2× TaqMan® GTXpress™ Master Mix. The cycling conditions were: initial denaturation for 2 minutes at 95°C; 40 cycles at 95°C for 15 seconds, and 60°C for 40 seconds.
Statistical analysis
Data normality was assessed using the Kolmogorov-Smirnov test. Normally distributed continuous data are shown as means (SD). Non-normally distributed data are shown as median (interquartile range). Plasma triglyceride, BUN, CK-MB, troponin I, and hs-CRP values were logarithmically transformed before the analysis due to skewed distribution. The unpaired Student's t-test and chi-square test were used to compare means and proportions between the control, STEMI, and stable angina groups when appropriate. A Pearson correlation heat map was employed to investigate the relationship between TCF21 genotypes and the occurrence of stable angina and STEMI among patients. Associations of genotype frequencies with the risk of stable angina and STEMI were assessed using logistic regression analysis after controlling for age, sex, smoking, BMI, hypertension, hyperlipidemia, and diabetes mellitus, and reported as odds ratios (ORs) with 95% confidence intervals (CIs). Continuous variables among the three genotypes of TCF21 were compared with one-way analysis of variance. Prevalence rates of type 2 diabetes mellitus (T2DM), hypertension, and hyperlipidemia status and trends in the three genotypes of TCF21 groups were analyzed using the chi-square test and Cochran-Armitage trend test respectably. JMP version 7.0 for Windows (SAS Institute, Cary, NC) was used for the statistical analysis. A p value <0.05 was considered to be statistically significant.
Results
Demographic characteristics of the control, stable angina and STEMI groups
The patients with STEMI were significantly older, predominantly male, and had higher rates of hypertension, T2DM, hyperlipidemia, current smoker, drinking, BMI, SBP than the control group. In addition, the patients with stable angina were significantly older, predominantly male, and had higher rates of hypertension, T2DM, hyperlipidemia, current smoker, drinking, BMI, and SBP than the control group. Furthermore, the STEMI group had significantly higher rates of single-vessel disease, stenosis in right coronary and left anterior descending arteries, and higher Gensini score than the stable angina group (Table 1).
Distributions of demographic characteristics in 276 controls and 519 stable angina or STEMI patients
| Parameters | Controls | STEMI | Stable angina | p-valuea | p-valueb | p-valuec |
|---|---|---|---|---|---|---|
| N | 276 | 138 | 381 | |||
| Age (years) | 58.4 ± 11.2 | 65.9 ± 12.9 | 63.9 ± 10.9 | < 0.0001 | < 0.0001 | 0.079 |
| Sex (male/female) | 72/204 | 114/24 | 297/84 | < 0.0001 | < 0.0001 | 0.248 |
| Hypertension (n, %) | 94 (34.1) | 84 (60.9) | 265 (69.6) | < 0.0001 | < 0.0001 | 0.063 |
| Type 2 diabetes mellitus (n, %) | 49 (17.8) | 52 (37.7) | 148 (38.9) | < 0.0001 | < 0.0001 | 0.810 |
| Hyperlipidemia (n, %) | 42 (15.2) | 101 (73.2) | 249 (65.4) | < 0.0001 | < 0.0001 | 0.092 |
| Current smoker (n, %) | 62 (22.5) | 56 (40.6) | 157 (41.2) | 0.0001 | < 0.0001 | 0.872 |
| Drinking (n, %) | 50 (18.1) | 39 (28.3) | 124 (32.6) | 0.016 | < 0.0001 | 0.308 |
| Body mass index (kg/m2) | 23.7 ± 4.9 | 25.8 ± 3.7 | 26.3 ± 3.7 | < 0.0001 | < 0.0001 | 0.176 |
| Systolic blood pressure (mmHg) | 122 ± 15 | 134 ± 23 | 132 ± 20 | < 0.0001 | < 0.0001 | 0.247 |
| Diastolic blood pressure (mmHg) | 75 ± 12 | 76 ± 16 | 77 ± 13 | 0.534 | 0.169 | 0.728 |
| LVMI | - | 134.7 ± 49.3 | 123.4 ± 40.5 | - | - | 0.059 |
| LVEF (%) | - | 59.5 ± 13.4 | 60.1 ± 13.3 | - | - | 0.736 |
| Number of diseased vessels (%) | ||||||
| Single-vessel disease | - | 48 (34.8) | 98 (25.7) | - | - | 0.044 |
| Two-vessel disease | - | 45 (32.6) | 101 (26.5) | - | - | 0.178 |
| Three-vessel disease | - | 41 (29.7) | 110 (28.9) | - | - | 0.866 |
| Location of stenosis | - | - | ||||
| Left anterior descending | - | 106 (76.8) | 251 (65.9) | - | - | 0.018 |
| Left circumflex | - | 64 (46.4) | 171 (44.9) | - | - | 0.781 |
| Right coronary artery | - | 86 (62.3) | 189 (49.6) | - | - | 0.011 |
| Gensini score | - | 40.0 (14.5-76.5) | 22.5 (8.0-52.0) | - | - | 0.012 |
Data are expressed as mean ± SD, number (percentage), or median (interquartile range). STEMI, ST elevation myocardial infarction; LVMI, left ventricular mass index; LVEF, left ventricular ejection fraction; a: Data were compared between ST elevation myocardial infarction patients and controls. b: Data were compared between stable angina patients and controls. c: Data were compared between ST elevation myocardial infarction patients and stable angina patients.
Biochemical characteristics in controls and stable angina or STEMI patients
Patients with STEMI had a significantly higher levels of fasting sugar, HbA1C, total cholesterol, triglyceride, LDL-C, uric acid, BUN, creatinine, total WBC/neutrophil/ monocyte/lymphocyte counts, and hs-CRP, and lower HDL-C, eGFR, and albumin compared to the control group. In addition, the stable angina group had significantly higher fasting sugar, HbA1C, total cholesterol, triglyceride, LDL-C, uric acid, BUN, creatinine, total WBC/neutrophil/monocyte/lymphocyte counts, and hs-CRP, and lower HDL-C, eGFR, and hemoglobin than the control group. Furthermore, the STEMI group had significantly higher fasting sugar, hemoglobin, total WBC/neutrophil/monocyte/lymphocyte counts, CK-MB, and troponin I, and lower HDL-C, eGFR, and albumin than the stable angina group (Table 2).
Biochemical characteristics in 276 controls and 519 stable angina or ST elevation myocardial infarction patients
| Parameters | Controls | STEMI | Stable angina | p-valuea | p-valueb | p-valuec |
|---|---|---|---|---|---|---|
| No. | 276 | 138 | 381 | |||
| Fasting sugar (mg/dL) | 96.1 ± 31.5 | 168.3 ± 80.1 | 136.6 ± 69.9 | < 0.0001 | < 0.0001 | <0.0001 |
| HbA1C (%) | 5.7 ± 0.7 | 7.0 ± 1.9 | 6.9 ± 1.7 | < 0.0001 | < 0.0001 | 0.789 |
| Total cholesterol (mg/dL) | 157.2 ± 32.3 | 181.5 ± 42.6 | 176.3 ± 45.3 | < 0.0001 | < 0.0001 | 0.241 |
| Triglyceride (mg/dL) | 84.0 (63.0 - 116.5) | 127.0 (86.8 - 197.5) | 118.0 (90.0 - 179.0) | < 0.0001 | < 0.0001 | 0.329 |
| HDL-cholesterol (mg/dL) | 54.7 ± 15.3 | 37.0 ± 12.0 | 40.8 ± 11.0 | < 0.0001 | < 0.0001 | 0.001 |
| LDL-cholesterol (mg/dL) | 90.9 ± 27.1 | 110.8 ± 36.0 | 104.0 ± 38.4 | < 0.0001 | < 0.0001 | 0.070 |
| Uric acid (mg/dL) | 4.7 ± 1.9 | 6.8 ± 2.0 | 6.5 ± 2.2 | < 0.0001 | < 0.0001 | 0.348 |
| BUN (mg/dl) | 12.0 (9.0 - 15.0) | 18.1 (15.6 - 23.7) | 17.2 (13.5 - 22.1) | < 0.0001 | < 0.0001 | 0.092 |
| Creatinine (mg/dl) | 0.8 ± 0.5 | 1.5 ± 0.9 | 1.4 ± 1.1 | < 0.0001 | < 0.0001 | 0.534 |
| eGFR (ml/min/1.73m2) | 124.2 ± 44.3 | 60.7 ± 24.2 | 66.4 ± 25.9 | < 0.0001 | < 0.0001 | 0.024 |
| Hemoglobin (g/dl) | 14.3 ± 1.1 | 14.0 ± 2.4 | 13.6 ± 2.0 | 0.824 | 0.041 | 0.046 |
| Albumin (g/L) | 4.2 ± 0.4 | 3.8 ± 0.4 | 4.0 ± 0.4 | < 0.0001 | 0.784 | < 0.0001 |
| Total WBC count (109/L) | 6.367 ± 2.754 | 10.957 ± 4.301 | 7.632 ± 2.642 | < 0.0001 | < 0.0001 | < 0.0001 |
| Neutrophil count (109/L) | 4057 ± 2618 | 7762 ± 3841 | 4802 ± 2169 | < 0.0001 | 0.0001 | < 0.0001 |
| Monocyte count (109/L) | 380 ± 156 | 632 ± 371 | 448 ± 202 | < 0.0001 | < 0.0001 | < 0.0001 |
| Lymphocyte count (109/L) | 1761 ± 716 | 2367 ± 1340 | 2118 ± 1004 | < 0.0001 | < 0.0001 | 0.028 |
| Creatine kinase-MB (ng/mL) | - | 7.1 (2.4 - 73.9) | 2.4 (1.1 - 7.1) | - | - | < 0.0001 |
| Troponin I (ng/mL) | - | 1.1 (0.1 - 6.5) | 0.1 (0.0 - 0.8) | - | - | < 0.0001 |
| hs-CRP (mg/L) | 0.2 (0.1 - 0.5) | 3.7 (0.3 - 8.3) | 2.2 (0.8 - 6.2) | < 0.0001 | <0.0001 | 0.325 |
Data are expressed as mean ± SD, or median (interquartile range). STEMI, ST elevation myocardial infarction; HDL, high-density lipoprotein; LDL, low-density lipoprotein; BUN, blood urine nitrogen; eGFR, estimated glomerular filtration rate; WBC, white blood cell; hs-CRP, high sensitivity C-reactive protein. a: Data were compared between ST elevation myocardial infarction patients and controls. b: Data were compared between stable angina patients and controls. c: Data were compared between ST elevation myocardial infarction patients and stable angina patients.
Association between TCF21 genotypes and stable angina and STEMI
Distributions of the TCF21 gene G/C genotype in the stable angina, STEMI and control groups are shown in Table 3, which shows that they were in Hardy-Weinberg equilibrium. The TCF21 genotype distributions were significantly different between the stable angina and STEMI groups when compared to the control group (p < 0.01). Higher CC genotype distributions were found in the stable angina and STEMI groups, suggesting an association between TCF21 gene polymorphisms with stable angina and STEMI (CC vs. GG: crude OR = 2.00, 95% CI = 1.26-3.21, p = 0.004 for stable angina; OR = 4.25, 95% CI = 2.14-9.00, p < 0.0001 for STEMI). These associations between TCF21 CC genotypes and stable angina/STEMI persisted after logistic regression adjustments for age, sex, smoking, BMI, hypertension, hyperlipidemia, and diabetes mellitus (CC vs. GG: adjusted OR = 2.49, 95% CI = 1.24-5.07, p = 0.010 for stable angina; adjusted OR = 9.19, 95% CI = 4.36-16.33, p < 0.0001 for STEMI).
Adjusted odds ratio and 95% confidence interval of stable angina and ST elevation myocardial infarction with TCF21 genotypic frequencies
| Parameter | Controls (n=276) | Stable angina (n=381) | Crude OR | 95% CI | AORa | 95% CI | p-value |
|---|---|---|---|---|---|---|---|
| TCF21 genotypes | |||||||
| GG genotypeb | 56 (20.3) | 58 (15.2) | 1.00 | - | 1.00 | - | - |
| GC genotype | 153 (55.4) | 184 (48.3) | 1.16 | 0.76 - 1.78 | 1.61 | 0.87 - 3.02 | 0.133 |
| CC genotype | 67 (24.3) | 139 (36.5) | 2.00 | 1.26 - 3.21 | 2.49 | 1.24 - 5.07 | 0.010 |
| GG/GC genotypeb | 209 (75.7) | 242 (63.5) | 1.00 | - | 1.00 | - | - |
| CC genotype | 67 (24.3) | 139 (36.5) | 1.79 | 1.27 - 2.54 | 1.76 | 1.04 - 3.02 | 0.036 |
| Controls (n = 276) | STEMI (n = 138) | Crude OR | 95% CI | AORa | 95% CI | p-value | |
| TCF21 genotypes | |||||||
| GG genotypeb | 56 (20.3) | 12 (8.7) | 1.00 | - | 1.00 | - | - |
| GC genotype | 153 (55.4) | 65 (47.1) | 1.98 | 1.03 - 4.10 | 4.54 | 1.43 - 12.39 | 0.009 |
| CC genotype | 67 (24.3) | 61 (44.2) | 4.25 | 2.14 - 9.00 | 9.19 | 4.36 - 16.33 | < 0.0001 |
| GG/GC genotypeb | 209 (75.7) | 77 (55.8) | 1.00 | - | 1.00 | - | - |
| CC genotype | 67 (24.3) | 61 (44.2) | 2.47 | 1.60 - 3.82 | 4.44 | 2.05 - 10.09 | 0.0001 |
STEMI, ST elevation myocardial infarction; TCF21, transcription factor 21. AOR = adjusted odds ratio; CI = confidence interval. a: The adjusted ORs with their 95% CI were estimated by employing multiple logistic regression models, after controlling for age, sex, smoking, body mass index, hypertension, hyperlipidemia, and diabetes mellitus. b: as a reference group.
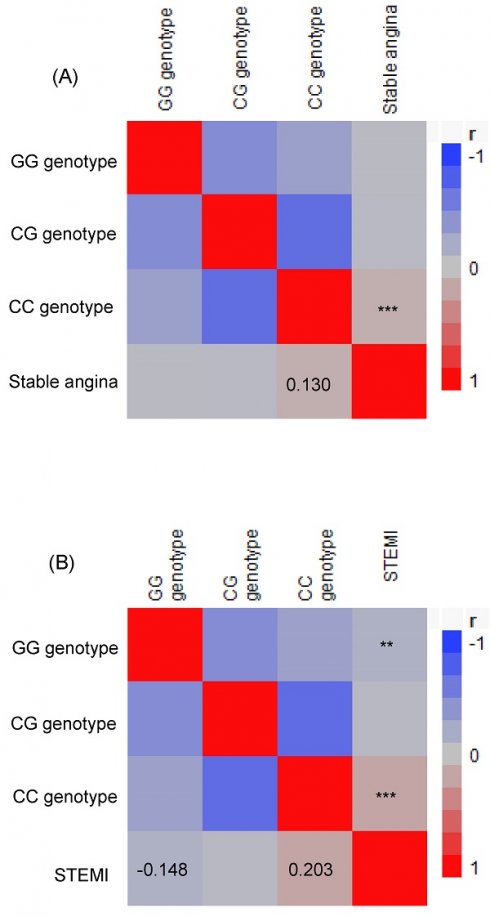
Comparing the risk of stable angina and STEMI between the CC vs. GG/GC genotypes showed that the CC genotype was significantly associated with a higher risk (Table 3). Furthermore, using Pearson's correlation analysis, TCF21 CC genotypes showed positive correlations with stable angina (r = 0.130, p = 0.001) and STEMI (r = 0.203, p < 0.0001), while TCF21 GG genotypes exhibited a negative correlation with STEMI (r = -0.148, p = 0.003; Figure 1).
A Pearson correlation heat map was generated to explore the relationship between the genotypes of the transcription factor 21 and the occurrence of stable angina (A) and ST elevation myocardial infarction (B) among patients. The red color indicates a positive correlation, whereas the blue and grey colors indicate a negative correlation. **p < 0.01 and ***p< 0.001. STEMI, ST elevation myocardial infarction.
Clinical and biochemical characteristics of the CAD patients by different genotypes of TCF21
Table 4 shows the clinical and biochemical characteristics of the 519 enrolled CAD patients according to TCF21 genotypes. The prevalence rates of GG genotype, GC genotype, and CC genotype were 13.5%, 48.0%, and 38.5%, respectively. The CC genotype group had higher level of hs-CRP than those with GG genotype. No significant differences were found in age, sex, BMI, waist circumference, waist-to-hip ratio, SBP, DBP, fasting glucose, HbA1c, total cholesterol, triglyceride, LDL-C, HDL-C, uric acid, BUN, creatinine, eGFR, hemoglobin, albumin, total WBC count, neutrophil count, monocyte count, and lymphocyte count among the three TCF21 genotypes.
Clinical and biochemical parameters of the studied patients with CAD in different genotypes of transcription factor 21
| Parameters | GG | GC | CC | p-value |
|---|---|---|---|---|
| No. | 70 | 249 | 200 | |
| Age | 64.0 ± 11.7 | 64.2 ± 11.4 | 64.8 ± 11.5 | 0.813 |
| Sex (male/female) | 54/16 | 196/53 | 161/39 | 0.810 |
| Body mass index (kg/m2) | 26.5 ± 3.9 | 26.1 ± 3.7 | 26.2 ± 3.7 | 0.726 |
| Waist circumference (cm) | 92.6 ± 8.3 | 91.9 ± 9.4 | 92.3 ± 10.9 | 0.862 |
| Waist-to-hip ratio | 0.94 ± 0.07 | 0.94 ± 0.07 | 0.94 ± 0.09 | 0.922 |
| Systolic blood pressure (mmHg) | 131 ± 21 | 133 ± 21 | 133 ± 21 | 0.874 |
| Diastolic blood pressure (mmHg) | 77± 13 | 78 ± 14 | 76 ± 14 | 0.351 |
| Fasting sugar (mg/dL) | 152.8 ± 92.1 | 143.9 ± 65.6 | 144.4 ± 77.3 | 0.678 |
| HbA1C (%) | 7.1 ± 2.1 | 6.8 ± 1.6 | 7.0 ± 1.7 | 0.472 |
| Total-cholesterol (mg/dL) | 178.5 ± 43.1 | 179.5 ± 43.7 | 175.0 ± 46.3 | 0.562 |
| Triglyceride (mg/dL) | 115.0 (85.5 - 178.3) | 119.5 (89.0 - 179.8) | 125.5 (93.3 - 190.8) | 0.742 |
| HDL-cholesterol (mg/dL) | 39.3 ± 10.3 | 39.7 ± 11.0 | 40.2 ± 12.2 | 0.792 |
| LDL-cholesterol (mg/dL) | 106.6 ± 34.1 | 107.8 ± 39.6 | 103.0 ± 36.9 | 0.412 |
| Uric acid (mg/dL) | 6.2 ± 1.7 | 6.8 ± 2.4 | 6.6 ± 2.0 | 0.249 |
| BUN (mg/dl) | 17.6 (14.4 - 25.3) | 17.8 (13.5 - 22.0) | 17.7 (14.0 - 23.0) | 0.082 |
| Creatinine (mg/dl) | 1.2 (1.0 - 1.4) | 1.2 (1.1 - 1.4) | 1.2 (1.1 - 1.4) | 0.177 |
| eGFR (ml/min/1.73m2) | 66.9 ± 29.4 | 66.1 ± 24.6 | 62.8 ± 25.3 | 0.320 |
| Hemoglobin (g/dl) | 13.4 ± 2.2 | 13.8 ± 2.0 | 13.7 ± 2.2 | 0.528 |
| Albumin (g/L) | 3.9 ± 0.5 | 4.0 ± 0.4 | 3.9 ± 0.4 | 0.270 |
| Total WBC count (109/L) | 8.341 ± 3.361 | 8.380 ± 3.520 | 8.747 ± 3.500 | 0.490 |
| Neutrophil count (109/L) | 5218 ± 2797 | 5541 ± 3147 | 5709 ± 2857 | 0.500 |
| Monocyte count (109/L) | 471 ± 216 | 492 ± 295 | 508 ± 249 | 0.592 |
| Lymphocyte count (109/L) | 2235 ± 1155 | 2114 ± 976 | 2245 ± 1225 | 0.431 |
| hs-CRP (mg/L) | 1.9 (0.7-5.2) | 3.1 (0.8 - 10.0) | 3.2 (0.9 - 9.2) | 0.032 |
Data are expressed as mean ± SD, or median (interquartile range). HDL, high-density lipoprotein; LDL, low-density lipoprotein; BUN, blood urine nitrogen; eGFR, estimated glomerular filtration rate; WBC, white blood cell; hs-CRP, high sensitivity C-reactive protein.
Association between TCF21 genotypes and T2DM, hypertension, and hyperlipidemia status
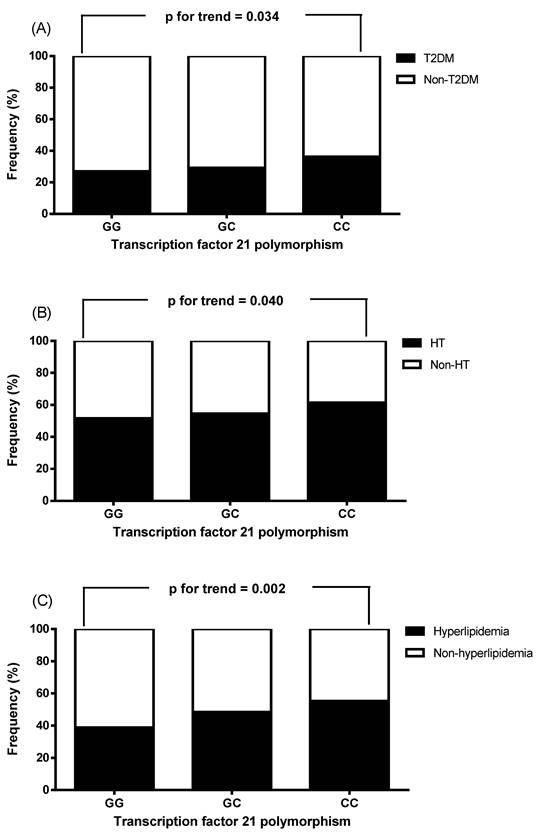
We then analyzed the frequencies of without and with T2DM, hypertension, and hyperlipidemia status of the all study participates stratified by genotypes of TCF21, and found that T2DM, hypertension, and hyperlipidemia status were significantly associated with genotypes of TCF21 (without T2DM vs. with T2DM, GG/GC/CC = 73.0%/70.7%/ 63.7% vs. 27.0%/29.4%/36.3%, p for trend = 0.034; without hypertension vs. with hypertension, GG/GC/CC = 48.4%/45.5%/38.6% vs. 51.6%/54.5%/61.4%, p for trend = 0.040; without hyperlipidemia vs. with hyperlipidemia, GG/GC/CC = 61.1%/51.5%/ 44.6% vs. 38.9%/48.5%/55.4%, p for trend = 0.002, Figure 2).
The frequencies of with or without type 2 diabetes mellitus (T2DM), hypertension, and hyperlipidemia status of the all study participates stratified by genotypes of transcription factor 21 (TCF21). T2DM, hypertension, and hyperlipidemia status were significantly associated with genotypes of TCF21.
Discussion
Our results showed significantly higher distributions of the TCF21 CC homozygote genotype in the stable angina and STEMI patients compared to the controls. In addition, independent associations between the TCF21 CC homozygote genotype with stable angina and STEMI were shown in univariate and multivariate logistic regression analyses after adjusting for age, sex, smoking, BMI, hypertension, hyperlipidemia, and diabetes mellitus. Furthermore, TCF21 CC genotypes demonstrated positive correlations with both stable angina and STEMI, while TCF21 GG genotypes exhibited a negative correlation with STEMI. Even though previous epidemiological studies have indicated that environmental factors play key roles in the development of cardiovascular disease [32,33], not all people develop CAD [13,34]. Molecular epidemiological research has shown that both genetic and environmental factors can influence susceptibility to CAD [35,36].
A previous study reported a significant association between the TCF21 gene G/C polymorphism with CAD in an Egypt population [22]. Santos et al. reported that the TCF21 polymorphism was a risk factor for CAD events in a Portuguese population [37]. Our results further suggest an association between the TCF21 CC homozygote genotype in both the occurrence of stable angina and STEMI in a Han Taiwanese cohort. The precise role of TCF21 polymorphisms in stable angina and STEMI has yet to be confirmed, although chronic inflammatory cytokines have been implicated [38-40]. Previous studies have shown that chemokines, including macrophage inflammatory protein-alpha, IFN-γ-inducible protein, monocyte chemoattractant protein-1, and eotaxin, play key roles in the pathogenesis of CAD via chronic inflammation [41].
A variation of TCF21 at 6q23.2 has been associated with CAD in Han Chinese and Caucasian populations [35,42]. A previous study showed that TCF21 modulates and interacts with aryl-hydrocarbon receptor (AHR), a well-known environmental sensor, and that this regulates the expressions of pro-inflammatory genes in coronary artery smooth muscle cells [43]. In addition, the study found that oxidized LDL, a widely recognized contributor to atherosclerosis within plaques, can induce the AHR pathway [43]. Taken together with our findings, TCF21 gene polymorphisms may play important roles with a heritable form of gene-environment interaction in stable angina and STEMI. In addition, Kim et al. also suggested that TCF21 can cooperate with AHR to activate an inflammatory gene expression program that is exacerbated by environmental stimuli, and may contribute to the overall risk of CAD [43]. In the present study, we found that the TCF21 CC genotype group had higher level of hs-CRP than those with TCF21 GG genotype group. Thus, patients with the TCF21 gene CC genotype may, through the increased effect of AHR, on inflammatory gene expression activation, contribute to the increased risk of stable angina and STEMI.
The TCF21 gene has been associated with CAD, promoting the stability of plaques and reducing clinical events by modulating the phenotypic transition from smooth muscle cells to fibromyocytes in atherosclerosis [44]. The present study shows that TCF21 gene G/C polymorphism is significantly associated with stable angina and STEMI both in non-adjusted and adjusted models (Table 3). In agreement with our results, Wang et al. [45], Hamed et al. [22] and Schunkert et al. [35] reported significant associations between TCF21 rs12190287 with CAD. Furthermore, Santos et al. also demonstrated that TCF21 rs12190287 may be a risk factor for major adverse cardiovascular events, and suggested that the TCF21 gene may affect fundamental smooth muscle cell processes in response to vascular stress, thereby facilitating the progression of atherosclerosis [37]. The mechanisms between rs12190287 risk C allele and CAD could be explained two key pathways. First, TCF21 risk alleles (rs12524865-C or rs12190287-C) may increase the expression of TCF21 on activation of platelet-derived growth factor (PDGF) signaling in coronary vascular smooth muscle cells (VSMCs) [46]. PDGF signaling mediated by PDGF receptor beta has been shown to play a major role in epithelial-mesenchymal transition, epicardial fate, VSMC migration, proliferation, and atherosclerosis [47]. This increase in PDGF signaling may then promote aggressive formation of plaques in the thoracic aorta and coronary arteries [48]. Second, the major risk C variants of TCF21 rs12190287 preferentially combine with miRNA-224 due to the altered secondary RNA structure, leading to downregulation of the expression of TCF21 [49]. Furthermore, TGF-β1 and PDGF signaling may be upstream miRNA-224 mediators and influence allele-specific TCF21 rs12190287 expression. More importantly, the rs12190287 C allele may interact with miR-224 leading to a reduction in TCF21 expression through mechanisms linked to miRNA, playing important roles in the risk of stable angina and STEMI.
In the present study, we showed that T2DM, hypertension, and hyperlipidemia status were significantly associated with genotypes of TCF21 are consistent with previous studies [22,24]. Fujimaki et al. showed that after adjusting for age, sex, BMI and smoking status in multivariable logistic regression analysis, TCF21 rs12190287 was significantly correlated with hypertension (OR 1.21, p=0.0014), and that the C allele was a risk factor for hypertension [24]. Furthermore, Hamed et al. reported that patients with CC genotype predominance showed higher levels of triglycerides, LDL-C, and total cholesterol [22]. T2DM, hypertension, and hyperlipidemia were risk factor of atherosclerosis, which contributes to progression of myocardial infarction. Therefore, it is possible that TCF21 CC genotype may be associated with hypertension, hyperlipidemia, and diabetes mellitus, thereby contributing to risk of stable angina and STEMI in the present study. However, further studies are required to ascertain the role of TCF21 polymorphism in diabetes mellitus.
Some limitations of this study need to be considered. The sample size was relatively small. In addition, the cross-sectional design limits our ability to infer a causal relationship between TCF21 gene (12190287G/C) variants and stable angina and STEMI. Long-term follow-up studies are needed to verify the roles of TCF21 gene polymorphisms with regards to the risk of stable angina and STEMI. Furthermore, as the results of the present study were not replicated, validation of the findings is required in other ethnic groups or in other independent subject panels. Further prospective studies are required to verify our results.
Conclusions
The results of the current study indicate that the CC genotype of TCF21 (12190287G/C) polymorphism is associated with stable angina and STEMI. Our findings suggest that TCF21 rs12190287 polymorphisms could be potential genetic markers for the susceptibility to stable angina and STEMI in Chinese populations.
Acknowledgements
The authors would like to thank E-Da Hospital of the Republic of China, Taiwan, for financially supporting this research under contracts EDAHI111001, EDAHI112001, and EDCHP112002.
Competing Interests
The authors have declared that no competing interest exists.
References
1. Heart Disease Statistics 2022. Gerardo Sison Editor. Available online: https://www.singlecare.com/blog/news/heart-diseasestatistics/ (accessed on 21 January 2022).
2. Muse ED, Chen SF, Torkamani A. Monogenic and Polygenic Models of Coronary Artery Disease. Curr Cardiol Rep. 2021;23:107
3. European Heart References Network (EHN). European Cardiovascular Disease Statistics. 2017. Available online: https://ehnheart.org/cvd-statistics.html/ (accessed on 21 January 2022).
4. Statistics on causes of death. 2020 [Available from: https://www.mohw.gov.tw/lp-5256-2.html]
5. Mach F, Baigent C, Catapano AL, Koskinas KC, Casula M, Badimon L, et al.; ESC Scientific Document Group. 2019 ESC/EAS Guidelines for the management of dyslipidaemias: lipid modification to reduce cardiovascular risk. Eur Heart J. 2020;41:111-88
6. Visseren FLJ, Mach F, Smulders YM, Carballo D, Koskinas KC, Bäck M. et al. ESC Scientific Document Group. 2021 ESC Guidelines on cardiovascular disease prevention in clinical practice: Developed by the Task Force for cardiovascular disease prevention in clinical practice with representatives of the European Society of Cardiology and 12 medical societies with the special contribution of the European Association of Preventive Cardiology (EAPC). Rev Esp Cardiol (Engl Ed). 2022;75:429
7. Brown JC, Gerhardt TE, Kwon E. Risk Factors for Coronary Artery Disease. In StatPearls; StatPearls Publishing: Treasure Island, FL, USA. 2021 Available online: https://www.ncbi.nlm.nih.gov/books/NBK554410/ (accessed on 22 January 2022)
8. Knuuti J, Wijns W, Saraste A, Capodanno D, Barbato E, Funck-Brentano C. et al. ESC Scientific Document Group. 2019 ESC Guidelines for the diagnosis and management of chronic coronary syndromes. Eur Heart J. 2020;41:407-77
9. Pechlivanis S, Lehmann N, Hoffmann P, Nöthen MM, Jöckel KH, Erbel R. et al. Risk prediction for coronary heart disease by a genetic risk score-results from the Heinz Nixdorf Recall study. BMC Med Genet. 2020;21:178
10. Dai X, Wiernek S, Evans JP, Runge MS. Genetics of coronary artery disease and myocardial infarction. World J Cardiol. 2016;8:1-23
11. Vaduganathan M, Mensah GA, Turco JV, Fuster V, Roth GA. The Global Burden of Cardiovascular Diseases and Risk: A Compass for Future Health. J Am Coll Cardiol. 2022;80:2361-71
12. Schunkert H, Exdmann J, Samani N. Genetics of myocardial infarction: a progress report. Eur Heart J. 2010;31:918-25
13. Lanktree MB, Hegele RA. Gene-gene and gene-environment interactions: new insights into the prevention, detection and management of coronary artery disease. Genome Med. 2009;1:28
14. Mehta N. Large-scale association analysis identifies 13 new susceptibility loci for coronary artery disease. Nat Genet. 2011;43:333-8
15. Massari ME, Murre C. Helix-loop-helix proteins: regulators of transcription in eucaryotic organisms. Mol Cell Biol. 2000;20:429-40
16. Kanisicak O, Khalil H, Ivey MJ, Karch J, Maliken BD, Correll RN. et al. Genetic lineage tracing defines myofibroblast origin and function in the injured heart. Nat Commun. 2016;7:12260
17. Acharya A, Baek ST, Huang G, Eskiocak B, Goetsch S, Sung CY. et al. The bHLH transcription factor Tcf21 is required for lineage-specific EMT of cardiac fibroblast progenitors. Development. 2012;139:2139-49
18. Sánchez-Iranzo H, Galardi-Castilla M, Sanz-Morejón A, González-Rosa JM, Costa R, Ernst A. et al. Transient fibrosis resolves via fibroblast inactivation in the regenerating zebrafish heart. Proc Natl Acad Sci U S A. 2018;115:4188-93
19. Wirka RC, Wagh D, Paik DT, Pjanic M, Nguyen T, Miller CL. et al. Atheroprotective roles of smooth muscle cell phenotypic modulation and the TCF21 disease gene as revealed by single-cell analysis. Nat Med. 2019;25:1280-9
20. Zhao Q, Wirka R, Nguyen T, Nagao M, Cheng P, Miller CL. et al. TCF21 and AP-1 interact through epigenetic modifications to regulate coronary artery disease gene expression. Genome Med. 2019;11:23
21. Bastami M, Ghaderian SM, Omrani MD, Mirfakhraie R, Vakili H, Parsa SA. et al. MiRNA-related polymorphisms in miR-146a and TCF21 are associated with increased susceptibility to coronary artery disease in an Iranian population. Genet Test Mol Biomarkers. 2016;20:241-8
22. Hamed WA, Hammouda GE, El-Hefnawy SM, Abd El Gayed EM. Transcription factor 21 gene polymorphism in patients with coronary artery disease. Research Reports in Clinical Cardiology. 2017;8:13-8 DOI https://doi.org/10.2147/RRCC.S121617
23. Yang L, Gao X, Luo H, Huang Q, Su D, Tan X. et al. TCF21 rs12190287 polymorphisms are associated with ventricular septal defects in a Chinese population. Genet Test Mol Biomarkers. 2017;21:312-5
24. Fujimaki T, Oguri M, Horibe H, Kato K, Matsuoka R, Abe S. et al. Association of a transcription factor 21 gene polymorphism with hypertension. Biomed Rep. 2015;3:118-22
25. Zhang X, Huang J, Li J, Lu Q, Huang Y, Lu D. et al. Association Between TCF21 Gene Polymorphism with the Incidence of Paroxysmal Atrial Fibrillation and the Efficacy of Radiofrequency Ablation for Patients with Paroxysmal Atrial Fibrillation. Int J Gen Med. 2022;15:4975-83
26. Chung FM, Yang YH, Shieh TY, Shin SJ, Tsai JC, Lee YJ. Effect of alcohol consumption on estimated glomerular filtration rate and creatinine clearance rate. Nephrol Dial Transplant. 2005;20:1610-6
27. Bozkurt B, Coats AJ, Tsutsui H, Abdelhamid M, Adamopoulos S, Albert N. et al. Universal Definition and Classification of Heart Failure: A Report of the Heart Failure Society of America, Heart Failure Association of the European Society of Cardiology, Japanese Heart Failure Society and Writing Committee of the Universal Definition of Heart Failure. J Card Fail. 2021: S1071-9164(21)00050-6.
28. Expert Panel on Detection, Evaluation, Treatment of High Blood Cholesterol in Adults. Executive summary of the third report of the National Cholesterol Education Program (NCEP) Expert Panel on Detection, Evaluation, And Treatment of High Blood Cholesterol In Adults (Adult Treatment Panel III). JAMA. 2001;285:2486-97
29. American Diabetes Association. Diagnosis and classification of diabetes mellitus. Diabetes Care. 2012;35(Suppl 1):S64-71
30. Yu TH, Hsuan CF, Wu CC, Hung WC, Lee TL, Tsai IT. et al. Association of plasma fatty acid-binding protein 3 with estimated glomerular filtration rate in patients with type 2 diabetes mellitus. Int J Med Sci. 2022;19:82-8
31. Kong X, Ma Y, Chen J, Luo Q, Yu X, Li Y, et al; Chinese eGFR Investigation Collaboration. Evaluation of the chronic kidney disease epidemiology collaboration equation for estimating glomerular filtration rate in the Chinese population. Nephrol Dial Transplant. 2013;28:641-51
32. Münzel T, Hahad O, Sørensen M, Lelieveld J, Duerr GD, Nieuwenhuijsen M. et al. Environmental risk factors and cardiovascular diseases: a comprehensive expert review. Cardiovasc Res. 2022;118:2880-902
33. Reeves F, Potter BJ. Toward a Cardio-Environmental Risk Model: Environmental Determinants of Cardiovascular Disease. Can J Cardiol. 2023 S0828-282X(23) 01449-6
34. McPherson R, Tybjaerg-Hansen A. Genetics of Coronary Artery Disease. Circ Res. 2016;118:564-78
35. Schunkert H, König IR, Kathiresan S, Reilly MP, Assimes TL, Holm H. et al. Large-scale association analysis identifies 13 new susceptibility loci for coronary artery disease. Nat Genet. 2011;43:333-8
36. Coronary Artery Disease (C4D) Genetics Consortium. A genome-wide association study in Europeans and South Asians identifies five new loci for coronary artery disease. Nat Genet. 2011;43:338-44
37. Santos MR, Mendonça MI, Temtem M, Sá D, Sousa AC, Freitas S. et al. Transcription factor 21 gene and prognosis in a coronary population. Rev Port Cardiol. 2023: S0870-2551(23)00352-9.
38. Yang C, Deng Z, Li J, Ren Z, Liu F. Meta-analysis of the relationship between interleukin-6 levels and the prognosis and severity of acute coronary syndrome. Clinics (Sao Paulo). 2021;76:e2690
39. Al Shahi H, Shimada K, Miyauchi K, Yoshihara T, Sai E, Shiozawa T. et al. Elevated Circulating Levels of Inflammatory Markers in Patients with Acute Coronary Syndrome. Int J Vasc Med. 2015;2015:805375
40. Amin MN, Siddiqui SA, Ibrahim M, Hakim ML, Ahammed MS, Kabir A. et al. Inflammatory cytokines in the pathogenesis of cardiovascular disease and cancer. SAGE Open Med. 2020;8:2050312120965752
41. Rothenbacher D, Müller-Scholze S, Herder C, Koenig W, Kolb H. Differential expression of chemokines, risk of stable coronary heart disease, and correlation with established cardiovascular risk markers. Arterioscler Thromb Vasc Biol. 2006;26:194-9
42. Wang Y, Wang L, Liu X, Zhang Y, Yu L, Zhang F. et al. Genetic variants associated with myocardial infarction and the risk factors in Chinese population. PLoS One. 2014;9:e86332
43. Kim JB, Pjanic M, Nguyen T, Miller CL, Iyer D, Liu B. et al. TCF21 and the environmental sensor aryl-hydrocarbon receptor cooperate to activate a pro-inflammatory gene expression program in coronary artery smooth muscle cells. PLoS Genet. 2017;13:e1006750
44. Pan H, Reilly MP. A protective smooth muscle cell transition in atherosclerosis. Nat Med. 2019;25:1194-5
45. Wang Y, Wang L, Liu X, Zhang Y, Yu L, Zhang F. et al. Genetic variants associated with myocardial infarction and the risk factors in Chinese population. PLoS One. 2004;9:e86332
46. Miller CL, Anderson DR, Kundu RK, Raiesdana A, Nürnberg ST, Diaz R. et al. Disease-related growth factor and embryonic signaling pathways modulate an enhancer of TCF21 expression at the 6q23.2 coronary heart disease locus. PLoS Genet. 2013;9:e1003652
47. Smith CL, Baek ST, Sung CY, Tallquist MD. Epicardial-derived cell epithelial-to- mesenchymal transition and fate specification require PDGF receptor signaling. Circ Res. 2011;108:e15-26
48. He C, Medley SC, Hu T, Hinsdale ME, Lupu F, Virmani R. et al. PDGFRβ signalling regulates local inflammation and synergizes with hypercholesterolaemia to promote atherosclerosis. Nat Commun. 2015;6:7770
49. Miller CL, Haas U, Diaz R, Leeper NJ, Kundu RK, Patlolla B. et al. Coronary heart disease-associated variation in TCF21 disrupts a miR-224 binding site and miRNA-mediated regulation. PLoS Genet. 2014;10:e1004263
Author contact
Corresponding author: Dr. Cheng-Ching Wu; E-Da Hospital, I-Shou University, No. 1, Yi-Da Rd., Jiau-Shu Village, Yan-Chao Township, Kaohsiung, 82445, Taiwan; Tel: +886-7-615-1100 ext. 5914 or 5018; E-mail: maxvic24isu.edu.tw.

 Global reach, higher impact
Global reach, higher impact