Impact Factor ISSN: 1449-1907
Int J Med Sci 2023; 20(13):1705-1710. doi:10.7150/ijms.88720 This issue Cite
Research Paper
Utilization of sodium-glucose cotransporter 2 inhibitors on dry eye disease severity in patients with type 2 diabetes mellitus
1. Institute of Medicine, Chung Shan Medical University, Taichung, Taiwan.
2. Department of Ophthalmology, Show Chwan Memorial Hospital, Changhua, Taiwan.
3. Department of Family and Community Medicine, Chung Shan Medical University Hospital, Taichung, Taiwan.
4. School of Medicine, Chung Shan Medical University, Taichung, Taiwan.
5. Department of Ophthalmology, Nobel Eye Institute, Taipei, Taiwan.
6. Department of Ophthalmology, Jen-Ai Hospital Dali Branch, Taichung, Taiwan.
7. Department of Medical Research, Chung Shan Medical University Hospital, Taichung, Taiwan.
8. Department of Post-Baccalaureate Medicine, College of Medicine, National Chung Hsing University, Taichung, Taiwan.
9. Department of Optometry, Chung Shan Medical University, Taichung, Taiwan.
Received 2023-8-1; Accepted 2023-10-3; Published 2023-10-9
Abstract
Sodium-glucose cotransporter 2 (SGLT2) inhibitors have protective effects against various systemic diseases and neoplasms. This retrospective cohort study evaluated the severity of dry eye disease (DED) in patients with type 2 diabetes mellitus (T2DM) who were treated with SGLT2 inhibitors. Data were obtained from the National Health Insurance Research Database of Taiwan. Patients with T2DM who were treated with SGLT2 inhibitors were assigned to the SGLT2 group. Each patient in the SGLT2 group was matched to two individuals with T2DM who had not used SGLT2 inhibitors, constituting the control group. The primary outcomes were the development of DED and severe DED. A diagnosis of severe DED was indicated by the usage of cyclosporine. Cox proportional hazard regression was applied to yield adjusted hazard ratios (aHR) and 95% confidence intervals (CIs). In the SGLT2 group, 1864 new DED events and 147 severe DED events were recorded. Conversely, 4367 new DED events and 392 severe DED events were recorded in the control group. The incidence (aHR: 0.858, 95% CI: 0.811-0.908, p = 0.0010) and severity (aHR: 0.652, 95% CI: 0.481-0.777, p = 0.0006) of DED were significantly lower in the SGLT2 group than the control group after adjusting for multiple covariates. In subgroup analyses, the incidence and severity of DED were significantly lower in patients younger than 60 years old who were treated with SGLT2 inhibitors than in their older counterparts (p = 0.0008 and 0.0011, respectively). In conclusion, utilization of SGLT2 inhibitors in the T2DM population could reduce both the incidence and severity of DED.
Keywords: dry eye disease, SGLT2 inhibitors, epidemiology, diabetes mellitus, severity
Introduction
Type 2 diabetes mellitus (T2DM) is a widespread systemic disease, affecting approximately 10% of the global population [1, 2]. The primary treatments for T2DM are insulin injection and oral medication administration [3]. Of the available antidiabetic medications, sodium-glucose cotransporter 2 (SGLT2) inhibitors have gained attention for their effectiveness in controlling hyperglycemia [4, 5]. A relevant study revealed a 0.71% reduction in glycated hemoglobin levels when SGLT2 inhibitors were added to dipeptidyl peptidase-4 inhibitor monotherapy [6].
SGLT2 inhibitors offer therapeutic benefits in addition to hyperglycemia reduction for patients with T2DM [4]. The usage of SGLT2 inhibitors on mice with diabetes decreased the risk of myocardial infarction by suppressing cardiomyocyte autophagy [7]. These inhibitors were also associated with a reduced rate of chronic heart failure progression [8]. Moreover, for patients with T2DM who have chronic kidney disease, SGLT2 inhibitors were shown to lower glomerular capillary hyperfiltration and thereby reduce physical stress on the filtration barrier [9]. In addition to their role in managing hyperglycemia and heart-related problems, SGLT2 inhibitors may offer protective effects against other diseases.
Studies have also reported the influence of SGLT2 inhibitors on ocular diseases [10, 11]. Although these inhibitors have been suggested to lower the risk of diabetic retinopathy, some evidence has been collected from animal experiments, and a firm consensus has yet to be reached [12-15]. Other studies have demonstrated a decrease in the incidence of dry eye disease (DED), an inflammatory ocular disorder characterized by dryness and irritation, under the use of SGLT2 inhibitors [16, 17]. However, no study has yet evaluated the correlation between SGLT2 inhibitors and DED severity.
Consequently, our study aimed to evaluate the possible correlation between the use of SGLT2 inhibitors and DED severity. This investigation relied on data from the National Health Insurance Research Database (NHIRD) of Taiwan. Risk factors potentially related to DED development and severity was accounted for in the statistical analyses.
Materials and Methods
Data Source
This study adhered to principles outlined in the Declaration of Helsinki and its subsequent amendments. It received approval from both the National Health Insurance Administration of Taiwan and the Institutional Review Board of Chung Shan Medical University Hospital (Project code: CS1-20108). The requirement for written informed consent was waived by these institutions. The NHIRD of Taiwan served as the data source for this study. The database includes insurance claims data from Taiwan's health insurance system and encompasses medical records for approximately 23 million Taiwanese residents for the period January 1, 2000, to December 31, 2020. The NHIRD of Taiwan contains diagnostic codes from both the International Classification of Diseases Ninth Revision (ICD-9) and the International Classification of Diseases Tenth Revision (ICD-10). Additionally, it contains variables such as age, sex, education level, place of residence, imaging examination codes, laboratory examination codes, types of medical departments, procedure codes, surgical codes, and international anatomical therapeutic chemical (ATC) codes for medications.
Patient Selection
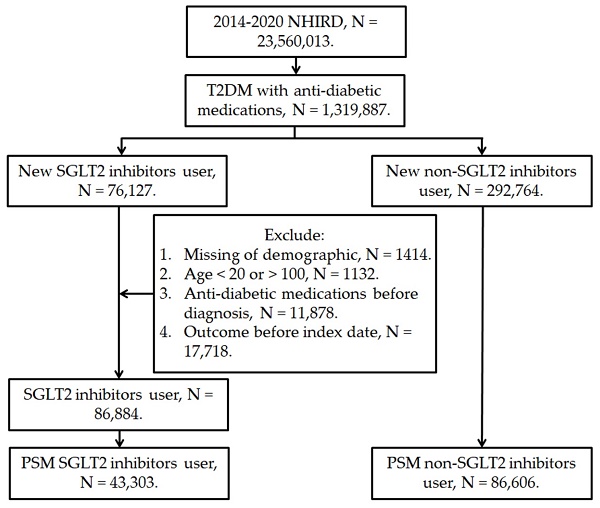
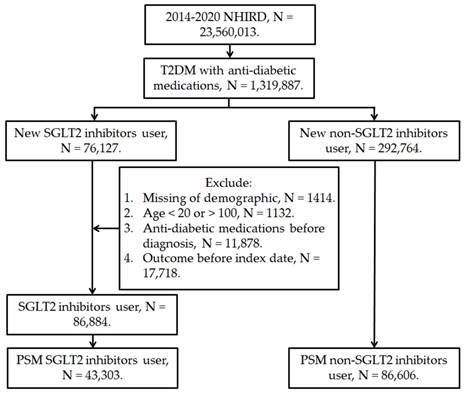
This retrospective cohort study enrolled patients with T2DM receiving SGLT2 inhibitor treatment if they fulfilled these conditions: (1) they had received a diagnosis of T2DM based on ICD-9 or ICD-10 codes between 2014 and 2019; (2) they underwent a minimum of 3 months of follow-up care in either an internal medicine or family medicine department; and (3) they were treated with an SGLT2 inhibitor—including dapagliflozin, canagliflozin, empagliflozin, and ertugliflozin—as identified by ATC codes. The index date for this study was set as the date 6 months after the initiation of SGLT2 inhibitor treatment. In addition, these exclusion criteria were applied: (1) the database contained incomplete demographic data; (2) the patient was prescribed an antidiabetic medication before they received their T2DM diagnosis; (3) the patient was younger than 20 years or older than 100 years; and (4) DED developed before the index date. For comparative purposes, this study also collected a control group of patients with T2DM who had not received an SGLT2 inhibitor. We matched the SGLT2 inhibitor group to the control group by using a PSM procedure that adjusted for demographic, systemic, and medication-related covariates in a 1:2 ratio. A total of 43,303 individuals in the SGLT2 inhibitor group and 86,606 individuals in the control group were selected (Figure 1).
The flowchart of participant selection. NHIRD: National Health Insurance Research Database, N: number, T2DM: type 2 diabetes mellitus, SGLT2: sodium-glucose cotransporter 2, PSM: propensity score-matching.
Primary Outcome Measurement
The primary outcome in this study was the development of DED, which was indicated by the following criteria: (1) assignment of DED-related ICD-9 or ICD-10 diagnostic codes; (2) performance of either a tear break-up time examination or a Schirmer test on or before the day of DED diagnosis, as revealed by procedure codes; (3) prescription of artificial tears following DED diagnosis, as indicated by ATC codes; and (4) diagnosis of DED by an ophthalmologist. The severe DED in this study was defined as the complement of above DED criteria plus the application of cyclosporine according to the ATC codes. If a patient did not receive cyclosporine therapy, the patient would be regarded as DED but not severe DED. Patients in the study were followed until either a DED diagnosis, withdrawal from the National Health Insurance program, or December 31, 2020.
Demographic and Systemic Covariate Enrollment
Multiple demographic data and systemic diseases were adjusted for in our multivariable model to reduce their influence on the outcome of DED development: age, sex, level of urbanization, hypertension, coronary heart disease, hyperlipidemia, cerebrovascular accidents, kidney disease, rheumatoid arthritis, systemic lupus erythematosus, Sjogren syndrome, and ankylosing spondylitis. The existence of these covariates was determined from the related codes in the NHIRD. To ensure that the systemic morbidities had persisted long enough to influence DED development, only those that been recorded for more than 2 years before index date were accounted for.
Statistical Analysis
SAS version 9.4 (SAS Institute, Cary, NC, USA) was used for statistical analyses. The statistical significance threshold was set at p < 0.05. Any p-value less than 0.0001 is presented as p < 0.0001.
Results
The baseline characteristics of the following two study groups are presented in Table 1: those treated with SGLT2 inhibitors and a control group. The groups had similar age distributions, with no significant differences discovered. The number of male participants was 28,410 in the SGLT2 group and 56,820 in the control group, the percentage of men was nonsignificantly different between the two groups. The distributions of covariates—including urbanization level, systemic factors, and types of antidiabetic medications used—were also similar in the two groups, as ensured by the propensity score matching (PSM) procedure (all absolute standardized differences < 0.1; Table 1).
The characteristic in SGLT2 group and matched diabetes population
| Characters | Control group (N = 86,606) | SGLT2 group (N = 43,303) | ASD |
|---|---|---|---|
| Age | |||
| 20-39 | 10746 (12.41%) | 6515 (15.05%) | 0.0012 |
| 40-49 | 23590 (27.24%) | 12066 (27.86%) | 0.0008 |
| 50-59 | 29582 (34.16%) | 13864 (32.02%) | 0.0017 |
| 60-69 | 18219 (21.04%) | 8598 (19.86%) | 0.0016 |
| 70-79 | 3763 (4.34%) | 1891 (4.37%) | 0.0004 |
| ≥ 80 | 706 (0.82%) | 369 (0.85%) | 0.0005 |
| Sex | |||
| Male | 56820 (65.61%) | 28410 (65.61%) | 0.0000 |
| Female | 29786 (34.39%) | 14893 (34.39%) | 0.0000 |
| Urbanization | |||
| Level 1 | 21506 (24.83%) | 11589 (26.76%) | 0.0056 |
| Level 2 | 28368 (32.76%) | 14387 (33.22%) | 0.0010 |
| Level 3 | 16882 (19.49%) | 8073 (18.64%) | 0.0015 |
| ≥ Level 4 | 19850 (22.92%) | 9254 (21.38%) | 0.0009 |
| Co-morbidity | |||
| Hypertension | 41859 (48.33%) | 22744 (52.52%) | 0.0245 |
| CHD | 5875 (6.78%) | 5506 (12.72%) | 0.0874 |
| Hyperlipidemia | 53466 (61.73%) | 29962 (69.19%) | 0.0659 |
| Cerebrovascular accident | 3347 (3.86%) | 1590 (3.67%) | 0.0008 |
| Kidney disease | 4238 (4.89%) | 2960 (6.84%) | 0.0437 |
| Rheumatoid arthritis | 421 (0.49%) | 160 (0.37%) | 0.0153 |
| Systemic lupus erythematosus | 56 (0.06%) | 35 (0.08%) | 0.0006 |
| Sjogren syndrome | 250 (0.29%) | 125 (0.29%) | 0.0000 |
| Ankylosing spondylitis | 681 (0.79%) | 368 (0.85%) | 0.0008 |
| Co-medication | |||
| Biguanides | 73225 (84.55%) | 38640 (89.23%) | 0.0077 |
| Sulfonylureas | 26345 (30.42%) | 19052 (44.00%) | 0.0561 |
| Alpha glucosidase inhibitors | 3133 (3.62%) | 2903 (6.70%) | 0.0704 |
| Thiazolidinediones | 4362 (5.04%) | 4036 (9.32%) | 0.0664 |
| dipeptidyl peptidase-4 inhibitor | 21935 (25.33%) | 18130 (41.87%) | 0.0833 |
| Insulin | 3969 (4.58%) | 3874 (8.95%) | 0.0992 |
N: number, SGLT2: sodium-glucose cotransporter 2, ASD: absolute standard difference, CHD: coronary heart disease
Over the course of a follow-up interval of up to 5 years, 1864 and 4367 new DED events were recorded in the SGLT2 and control groups, respectively, whereas 147 and 392 severe DED events were recorded (Table 2). The incidence (adjusted hazard ratio [aHR]: 0.858, 95% CI: 0.811-0.908, p = 0.0010) and severity (aHR: 0.652, 95% CI: 0.481-0.777, p = 0.0006) of DED were significantly lower in the SGLT2 group than in the control group after adjusting multiple demographic and systemic covariates (Table 2). Other factors that correlated with the development of DED included advanced age, female sex, Sjogren's syndrome, and ankylosing spondylitis (all p < 0.05; Table 3).
The risk of dry eye disease between SGLT2 and control groups
| Event | Control group | SGLT2 group | P |
|---|---|---|---|
| DED | |||
| Person-months | 1715969 | 877022 | |
| Event | 4367 | 1864 | |
| Crude HR (95% CI) | Reference | 0.837 (0.793-0.883)* | |
| aHR (95% CI) | Reference | 0.858 (0.811-0.908)* | 0.0010 |
| Severe DED | |||
| Person-months | 1715969 | 877022 | |
| Event | 392 | 147 | |
| Crude HR (95% CI) | Reference | 0.645 (0.463-0.759)* | |
| aHR (95% CI) | Reference | 0.652 (0.481-0.777)* | 0.0006 |
DED: dry eye disease, aHR: adjusted hazard ratio, CI: confidence interval
* Denotes significant difference between the two groups
The effect of each parameter on the development of dry eye disease
| Parameters | aHR | 95% CI | P |
|---|---|---|---|
| SGLT2 inhibitors | 0.652 | 0.481-0.777 | 0.0010* |
| Age | 1.875 | 1.013-2.651 | 0.0007* |
| Male sex | 2.007 | 1.285-3.428 | 0.0003* |
| Urbanization | 1.045 | 0.468-2.364 | 0.8769 |
| Co-morbidity | |||
| Hypertension | 1.232 | 0.782-1.964 | 0.6213 |
| CHD | 0.942 | 0.559-1.806 | 0.6652 |
| Hyperlipidemia | 1.367 | 0.656-2.228 | 0.5726 |
| Cerebrovascular accident | 1.003 | 0.877-1.380 | 0.7893 |
| Kidney disease | 0.737 | 0.661-1.482 | 0.0958 |
| Rheumatoid arthritis | 1.357 | 0.918-2.408 | 0.0738 |
| Systemic lupus erythematosus | 0.998 | 0.790-1.102 | 0.3542 |
| Sjogren syndrome | 1.495 | 1.113-2.091 | 0.0016* |
| Ankylosing spondylitis | 1.534 | 1.008-2.235 | 0.0334* |
aHR: adjusted hazard ratio, CI: confidence interval, SGLT2: sodium-glucose cotransporter 2, CHD: coronary heart disease
* Denotes significant difference between the two groups
Subgroup analyses comparing patients with T2DM under 60 years of age who received SGLT2 inhibitors with their counterparts aged 60 years and older demonstrated a significantly lower incidence (p = 0.0008) and severity (p = 0.0011) of DED in the younger patients (Table 4). However, no significant sex-based differences in DED incidence or severity were discovered (both p > 0.05; Table 4).
The subgroup analyses for dry eye disease development stratified by age and sex
| Parameters | aHR | 95% CI | P for interaction |
|---|---|---|---|
| DED | |||
| Age | 0.0008* | ||
| < 60 | 0.723 | 0.576-0.834 | |
| ≥ 60 | 0.920 | 0.878-0.993 | |
| Sex | 0.5381 | ||
| Male | 0.827 | 0.772-0.874 | |
| Female | 0.856 | 0.825-0.936 | |
| Severe DED | |||
| Age | 0.0011* | ||
| < 60 | 0.567 | 0.404-0.742 | |
| ≥ 60 | 0.715 | 0.555-0.798 | |
| Sex | 0.4076 | ||
| Male | 0.618 | 0.469-0.753 | |
| Female | 0.680 | 0.507-0.821 |
DED: dry eye disease, aHR: adjusted hazard ratio, CI: confidence interval, SGLT2: sodium-glucose cotransporter 2
* Denotes significant difference between the two groups
Discussion
In this study, the incidence of DED development was significantly lower in patients with T2DM who received SGLT2 therapy than in the control group, according to the multivariable analysis. The incidence of severe DED was lower in the SGLT2 group than in the control group. Patients with T2DM under the age of 60 years who received SGLT2 inhibitors were less likely to develop DED and have severe DED than were those 60 years and older.
Beyond blood sugar control, SGLT2 inhibitors have protective effects on various systemic diseases and neoplasms [18-20]. Specifically, the occurrence and progression of coronary heart disease can be retarded by the use of SGLT2 inhibitors [4]. A relevant study indicated that these inhibitors can mitigate microvascular damage and endothelial dysfunction in cardiac ischemic injury [21]. In addition, major CHD events and related mortality were found to be less likely in patients with T2DM who received SGLT2 inhibitors [22]. Patients with congestive heart failure, whether with preserved or reduced ejection fraction, were found to exhibit slower deterioration when they did rather than did not receive SGLT2 inhibitors [8]. In addition to cardiovascular benefits, SGLT2 inhibitors have protective effects on kidney function. Specifically, in patients with T2DM, SGLT2 inhibitors were discovered to reduce elevated serum creatinine levels, lower the rate of macroalbuminuria development, and reduce the likelihood of a need for hemodialysis [23]. Regarding its molecule pathways, SGLT2 inhibitors have been associated with both anti-inflammatory and antioxidant effects [24-26]. They can suppress the inflammatory process in certain types of cardiomyopathy, potentially through modulation of the HIF-2α signaling pathway [27, 28]. Moreover, oxidative stress levels were significantly lower in both in vitro and in vivo models of diabetic kidney disease when SGLT2 inhibitors were applied [29]. Concerning DED, this inflammatory disease is associated with increased levels of interleukins and tumor necrosis factor-alpha [30]. Elevated levels of reactive oxygen species were observed in the conjunctiva of patients with DED [30]. Various anti-inflammatory medications, such as cyclosporine and lifitegrast, are used to treat DED [31]. Given the broad protective effects of SGLT2 inhibitors and their specific role in inhibiting the molecule pathways in DED [29, 30], we proposed that the application of SGLT2 inhibitors may decrease both the incidence and severity of DED—a hypothesis supported by the results of this study.
In this study, the use of SGLT2 inhibitors was found to lead to both lower incidence and severity of DED in patients with T2DM. These results align with relevant studies that have examined similar populations treated with SGLT2 inhibitors [16, 17]. Additionally, our data suggest that the utilization of SGLT2 inhibitors correlates with lower DED severity, as evidenced by lower usage of cyclosporine in this study's participants. To our knowledge, this is a relatively preliminary finding indicating the protective effects of SGLT2 inhibitors regarding DED severity. Moreover, we excluded patients who had received a diagnosis of DED before the index data to eliminate the influence of pre-existing DED. The Cox proportional hazard regression models used in this study were adjusted for multiple known risk factors for DED development and progression. Consequently, the use of SGLT2 inhibitors may serve as an independent protective factor against high-severity DED. One study indicated that elevated tear film osmolarity and inflammatory responses contribute to high-severity DED [30]. Selenoprotein P mitigates DED by lowering oxidative stress [32]. Given the relevant anti-inflammatory and antioxidant mechanisms, SGLT2 inhibitors should reduce the likelihood of DED progression and high DED severity in patients with T2DM [23, 29]. Our findings highlight this possibility.
Concerning the subgroup analyses, the incidence and severity of DED were significantly lower in the patients with T2DM who were under the age of 60 years and treated with SGLT2 inhibitors than in those who were aged 60 years and older. Limited research has demonstrated this phenomenon. However, SGLT2 inhibitors were reported to reduce blood glucose levels across all ages, with significantly lower levels observed in patients less than 40 years old [33]. In one study, researchers attributed the enhanced glycemic control achieved under SGLT2 inhibitors to larger urinary glucose excretion [33]. Thus, in younger populations, the use of SGLT2 inhibitors may more effectively suppress hyperglycemia-related inflammation, leading to lower DED incidence and severity. Regarding the effect of SGLT2 inhibitors on different sexes, no conclusive evidence was obtained. SGLT2 inhibitors were found to produce similar heart failure control outcomes in men and women [34]. Likewise, the reduction in atrial fibrillation following SGLT2 inhibitor usage was nonsignificantly different between the sexes [35]. In this study, the aHRs for DED incidence and severity were not significantly different between the sexes, suggesting that sex may not affect the efficiency of SGLT2 inhibitors.
The global prevalence of T2DM and DED is substantial. In China, T2DM affects 8% of the population, whereas in Europe, its prevalence is 8.8% [2]. The highest prevalence of T2DM was found to be that in North America (13%) [2]. DED is also pervasive, diagnosed in 5%-50% of individuals aged 40 years or older [36]. Among visual display terminal users of all ages, the prevalence of DED can exceed 60% [37]. Moreover, T2DM can damage the corneal epithelium, leading to corneal erosion and DED development [38]. Because T2DM can lead to DED and both diseases affect a large population, whether SGLT2 inhibitors can be employed as a straightforward strategy to reduce the incidence and severity of DED in patients with T2DM warrants exploration.
This study had several limitations. First, the claimed-database design of our study let the blood sugar level, the duration of both DM and DED, the receipt of refractive surgery, the value of body mass index, the patient-reported symptoms and related questionnaires, the values of DED-related exam including the tear-break up time and Schirmer test, the types of DED, the external eye photography of DED patients, and the treatment outcome of DED become unavailable. Also, since the matching of too many covariates will cause over-fitting of the statistical analysis, we did not match all the immune disorder including thyroid disease in this study. Additionally, the severity of DED was assessed on the basis of the use of cyclosporine. However, some medications for advanced DED, including diquafosol and autologous serum, are self-funded and they are not recorded in the NHIRD. Consequently, some cases of severe DED may have been regarded as mild DED. In addition, although the duration of comorbidities was available for analysis, the severity of these co-morbidities, which could influence DED development and severity, was not. Finally, as nearly all the participants in this study were Asian, the external validity of our study could be limited.
In conclusion, the use of SGLT2 inhibitors in patients with T2DM correlates with significantly lower DED incidence and severity after adjusting for several DED risk factors. Furthermore, this protective effect appears to be stronger in patients under 60 years of age. Consequently, SGLT2 inhibitors might be recommended for patients with T2DM at high risk of DED. Further prospective, large-scale studies are required to evaluate the potential for SGLT2 inhibitors to reduce the numbers of treatments in patients with T2DM with DED.
Competing Interests
The authors have declared that no competing interest exists.
References
1. Xu B, Li S, Kang B, Zhou J. The current role of sodium-glucose cotransporter 2 inhibitors in type 2 diabetes mellitus management. Cardiovasc Diabetol. 2022;21:83
2. Lovic D, Piperidou A, Zografou I, Grassos H, Pittaras A, Manolis A. The Growing Epidemic of Diabetes Mellitus. Curr Vasc Pharmacol. 2020;18:104-109
3. Cho YK, Kang YM, Lee SE, Lee J, Park JY, Lee WJ. et al. Efficacy and safety of combination therapy with SGLT2 and DPP4 inhibitors in the treatment of type 2 diabetes: A systematic review and meta-analysis. Diabetes Metab. 2018;44:393-401
4. Tentolouris A, Vlachakis P, Tzeravini E, Eleftheriadou I, Tentolouris N. SGLT2 Inhibitors: A Review of Their Antidiabetic and Cardioprotective Effects. Int J Environ Res Public Health. 2019 16
5. Nelinson DS, Sosa JM, Chilton RJ. SGLT2 inhibitors: a narrative review of efficacy and safety. J Osteopath Med. 2021;121:229-239
6. Li D, Shi W, Wang T, Tang H. SGLT2 inhibitor plus DPP-4 inhibitor as combination therapy for type 2 diabetes: A systematic review and meta-analysis. Diabetes Obes Metab. 2018;20:1972-1976
7. Jiang K, Xu Y, Wang D, Chen F, Tu Z, Qian J. et al. Cardioprotective mechanism of SGLT2 inhibitor against myocardial infarction is through reduction of autosis. Protein Cell. 2022;13:336-359
8. van der Aart-van der Beek AB, de Boer RA, Heerspink HJL. Kidney and heart failure outcomes associated with SGLT2 inhibitor use. Nat Rev Nephrol. 2022;18:294-306
9. Vallon V, Verma S. Effects of SGLT2 Inhibitors on Kidney and Cardiovascular Function. Annu Rev Physiol. 2021;83:503-528
10. Li C, Zhou Z, Neuen BL, Yu J, Huang Y, Young T. et al. Sodium-glucose co-transporter-2 inhibition and ocular outcomes in patients with type 2 diabetes: A systematic review and meta-analysis. Diabetes Obes Metab. 2021;23:252-257
11. Zhou B, Shi Y, Fu R, Ni H, Gu L, Si Y. et al. Relationship Between SGLT-2i and Ocular Diseases in Patients With Type 2 Diabetes Mellitus: A Meta-Analysis of Randomized Controlled Trials. Front Endocrinol (Lausanne). 2022;13:907340
12. Sha W, Wen S, Chen L, Xu B, Lei T, Zhou L. The Role of SGLT2 Inhibitor on the Treatment of Diabetic Retinopathy. J Diabetes Res. 2020;2020:8867875
13. Mudaliar S, Hupfeld C, Chao DL. SGLT2 Inhibitor-Induced Low-Grade Ketonemia Ameliorates Retinal Hypoxia in Diabetic Retinopathy-A Novel Hypothesis. J Clin Endocrinol Metab. 2021;106:1235-1244
14. Ma Y, Lin C, Cai X, Hu S, Zhu X, Lv F. et al. The association between the use of sodium glucose cotransporter 2 inhibitor and the risk of diabetic retinopathy and other eye disorders: a systematic review and meta-analysis. Expert Rev Clin Pharmacol. 2022;15:877-886
15. Herat LY, Matthews JR, Ong WE, Rakoczy EP, Schlaich MP, Matthews VB. Determining the Role of SGLT2 Inhibition with Dapagliflozin in the Development of Diabetic Retinopathy. Front Biosci (Landmark Ed). 2022;27:321
16. Su YC, Hung JH, Chang KC, Sun CC, Huang YH, Lee CN. et al. Comparison of Sodium-Glucose Cotransporter 2 Inhibitors vs Glucagonlike Peptide-1 Receptor Agonists and Incidence of Dry Eye Disease in Patients With Type 2 Diabetes in Taiwan. JAMA Netw Open. 2022;5:e2232584
17. Pan LY, Kuo YK, Chen TH, Sun CC. Dry eye disease in patients with type II diabetes mellitus: A retrospective, population-based cohort study in Taiwan. Front Med (Lausanne). 2022;9:980714
18. Komatsu S, Nomiyama T, Numata T, Kawanami T, Hamaguchi Y, Iwaya C. et al. SGLT2 inhibitor ipragliflozin attenuates breast cancer cell proliferation. Endocr J. 2020;67:99-106
19. Karagiannis T, Tsapas A, Athanasiadou E, Avgerinos I, Liakos A, Matthews DR. et al. GLP-1 receptor agonists and SGLT2 inhibitors for older people with type 2 diabetes: A systematic review and meta-analysis. Diabetes Res Clin Pract. 2021;174:108737
20. Pawlos A, Broncel M, Woźniak E, Gorzelak-Pabiś P. Neuroprotective Effect of SGLT2 Inhibitors. Molecules. 2021 26
21. Ma L, Zou R, Shi W, Zhou N, Chen S, Zhou H. et al. SGLT2 inhibitor dapagliflozin reduces endothelial dysfunction and microvascular damage during cardiac ischemia/reperfusion injury through normalizing the XO-SERCA2-CaMKII-coffilin pathways. Theranostics. 2022;12:5034-5050
22. Brown E, Heerspink HJL, Cuthbertson DJ, Wilding JPH. SGLT2 inhibitors and GLP-1 receptor agonists: established and emerging indications. Lancet. 2021;398:262-276
23. Salvatore T, Galiero R, Caturano A, Rinaldi L, Di Martino A, Albanese G. et al. An Overview of the Cardiorenal Protective Mechanisms of SGLT2 Inhibitors. Int J Mol Sci. 2022 23
24. Cowie MR, Fisher M. SGLT2 inhibitors: mechanisms of cardiovascular benefit beyond glycaemic control. Nat Rev Cardiol. 2020;17:761-772
25. Xing YJ, Liu BH, Wan SJ, Cheng Y, Zhou SM, Sun Y. et al. A SGLT2 Inhibitor Dapagliflozin Alleviates Diabetic Cardiomyopathy by Suppressing High Glucose-Induced Oxidative Stress in vivo and in vitro. Front Pharmacol. 2021;12:708177
26. Li C, Zhang J, Xue M, Li X, Han F, Liu X. et al. SGLT2 inhibition with empagliflozin attenuates myocardial oxidative stress and fibrosis in diabetic mice heart. Cardiovasc Diabetol. 2019;18:15
27. Long Q, Li L, Yang H, Lu Y, Yang H, Zhu Y. et al. SGLT2 inhibitor, canagliflozin, ameliorates cardiac inflammation in experimental autoimmune myocarditis. Int Immunopharmacol. 2022;110:109024
28. Yang Z, Li T, Xian J, Chen J, Huang Y, Zhang Q. et al. SGLT2 inhibitor dapagliflozin attenuates cardiac fibrosis and inflammation by reverting the HIF-2α signaling pathway in arrhythmogenic cardiomyopathy. Faseb j. 2022;36:e22410
29. Winiarska A, Knysak M, Nabrdalik K, Gumprecht J, Stompór T. Inflammation and Oxidative Stress in Diabetic Kidney Disease: The Targets for SGLT2 Inhibitors and GLP-1 Receptor Agonists. Int J Mol Sci. 2021 22
30. Bron AJ, de Paiva CS, Chauhan SK, Bonini S, Gabison EE, Jain S. et al. TFOS DEWS II pathophysiology report. Ocul Surf. 2017;15:438-510
31. O'Neil EC, Henderson M, Massaro-Giordano M, Bunya VY. Advances in dry eye disease treatment. Curr Opin Ophthalmol. 2019;30:166-178
32. Kojima T, Dogru M, Kawashima M, Nakamura S, Tsubota K. Advances in the diagnosis and treatment of dry eye. Prog Retin Eye Res. 2020 100842
33. Nakamura Y, Nagai Y, Terashima Y, Nishine A, Ishii S, Kato H. et al. Better response to the SGLT2 inhibitor dapagliflozin in young adults with type 2 diabetes. Expert Opin Pharmacother. 2015;16:2553-2559
34. Butler J, Packer M, Filippatos G, Ferreira JP, Zeller C, Schnee J. et al. Effect of empagliflozin in patients with heart failure across the spectrum of left ventricular ejection fraction. Eur Heart J. 2022;43:416-426
35. Zelniker TA, Bonaca MP, Furtado RHM, Mosenzon O, Kuder JF, Murphy SA. et al. Effect of Dapagliflozin on Atrial Fibrillation in Patients With Type 2 Diabetes Mellitus: Insights From the DECLARE-TIMI 58 Trial. Circulation. 2020;141:1227-1234
36. Rouen PA, White ML. Dry Eye Disease: Prevalence, Assessment, and Management. Home Healthc Now. 2018;36:74-83
37. Uchino M, Yokoi N, Uchino Y, Dogru M, Kawashima M, Komuro A. et al. Prevalence of dry eye disease and its risk factors in visual display terminal users: the Osaka study. Am J Ophthalmol. 2013;156:759-766
38. Han SB, Yang HK, Hyon JY. Influence of diabetes mellitus on anterior segment of the eye. Clin Interv Aging. 2019;14:53-63
Author contact
Corresponding authors: Shun-Fa Yang, Ph.D. or Hung-Yu Lin, MD, Ph.D., Institute of Medicine, Chung Shan Medical University, Taichung 402, Taiwan; Tel: +886-4-24739595 ext. 34253; Fax: +886-4-24723229; E-mail: ysfedu.tw (Shun-Fa Yang); E-mail: anthonyhungyulincom (Hung-Yu Lin).

 Global reach, higher impact
Global reach, higher impact