Impact Factor ISSN: 1449-1907
Int J Med Sci 2022; 19(13):1977-1988. doi:10.7150/ijms.77193 This issue Cite
Research Paper
Expression and function of Siglec-15 in RLPS and its correlation with PD-L1: Bioinformatics Analysis and Clinicopathological Evidence
1. Key Laboratory of Carcinogenesis and Translational Research (Ministry of Education/Beijing), Department of Hepato-Pancreato-Biliary Surgery, Peking University Cancer Hospital & Institute, Beijing, People's Republic of China.
2. Key Laboratory of Carcinogenesis and Translational Research (Ministry of Education/Beijing), Department of Critical Care Medicine, Peking University Cancer Hospital and Institute, Beijing, People's Republic of China.
3. Key Laboratory of Carcinogenesis and Translational Research (Ministry of Education/Beijing), Central Laboratory, Peking University Cancer Hospital & Institute, Beijing, People's Republic of China.
4. Key Laboratory of Carcinogenesis and Translational Research (Ministry of Education/Beijing), Department of Pathology, Peking University Cancer Hospital & Institute, Beijing, People's Republic of China.
#These authors contributed equally to this work.
Abstract
Purpose: Retroperitoneal liposarcoma (RLPS) is a rare malignancy without effective treatment. Since current treatment for unresectable RLPS is unsatisfactory, immunotherapy and targeted therapy are urgently needed. Siglec-15 is a transmembrane protein highly homologous to PD-L1 and is involved in tumor immune escape. The biological function of Siglec-15 in RLPS, its prognostic relevance and its relationship with PD-L1 need to be further clarified. In this study, we aimed to explore the biological function of Siglec-15 in sarcomas through bioinformatics analysis, and we also evaluated Siglec-15 and PD-L1 expression in RLPS samples. The relationship between the expression of Siglec-15 and PD-L1 and their clinicopathological relevance and prognostic value were also investigated in clinical RLPS patients.
Methods: The RNA sequencing data of 259 sarcoma cases and 48 RLPS cases from TCGA were used to analyze the Siglec-15 expression and the differentially expressed genes (DEG) related with Siglec-15 expression. In addition, DEGs were subsequently analyzed through the gene ontology (GO)/ Kyoto Encyclopedia of Genes and Genomes (KEGG) and protein-protein interaction (PPI) network. Tumor specimens were obtained from 91 RLPS patients of our sarcoma center, and Siglec-15 and PD-L1 expression were evaluated using immunohistochemistry. The correlation between the expression level of these two markers as well as their correlation with clinicopathological factors and prognosis of RLPS patients was also assessed.
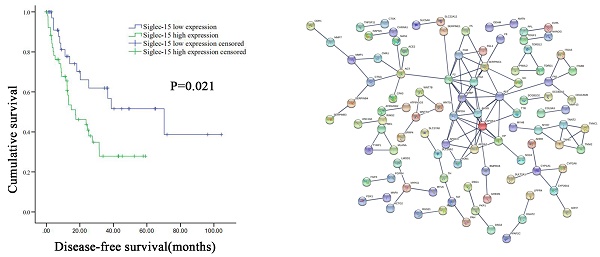
Results: GEPIA analysis showed that the high expression of Siglec-15 was associated with poor sarcoma OS (P=0.034). A total of 682 differential genes were identified between the high and low expression groups of Siglec-15 in RLPS. Enrichment analysis of the KEGG pathway showed that Siglec-15 was related to the Hippo signaling pathway and the neuroactive ligand-receptor interaction. GO annotation analysis showed that the expression of Siglec-15 may thus be able to affect serine hydrolase activity, alongside signal receptor activator activity. The top 5 genes with the largest number of connection points are APOA1, F2, AHSG, AMBP, SERPINC1. In subsequent studies, we used 91 liposarcoma samples from our center for verification. Siglec-15 was expressed in 84.6% of RLPS cases, whereas PD-L1 was expressed in 17.6% of RLPS cases. A negative correlation was observed between Siglec-15 and PD-L1 expression (P=0.020). In this group of RLPS patients, high Siglec-15 expression was correlated with poorer disease-free survival (DFS) (P=0.021), and it was an independent predictor of DFS (hazard ratio: 2.298; 95% confidence interval: 1.154-4.576; P=0.018). However, we did not find a correlation between PD-L1 expression and overall survival or DFS in RLPS patients.
Conclusion: The DEG and signaling pathways identified in the study could provide a preliminary understanding of the underlying molecular mechanisms of Siglec-15 in the development and progression of RLPS. High expression of Siglec-15 was a negative independent predictive factor for DFS of RLPS. The negative relationship between Siglec-15 and PD-L1 expression suggested that the Siglec-15 pathway might be an important supplement to PD-L1 treatment.
Keywords: Siglec-15, PD-L1, Retroperitoneal liposarcoma, Prognosis

 Global reach, higher impact
Global reach, higher impact