Impact Factor
ISSN: 1449-1907
Int J Med Sci 2022; 19(8):1265-1274. doi:10.7150/ijms.72655 This issue Cite
Research Paper
Exosomes with FOXP3 from gene-modified dendritic cells ameliorate the development of EAE by regulating the balance of Th/Treg
1. Department of Neurology, The Second Hospital of Hebei Medical University, Shijiazhuang, Hebei, China.
2. Neurological Laboratory of Hebei Province, Shijiazhuang, Hebei, China.
3. Department of Neurology, Dongzhimen Hospital, Beijing University of Chinese Medicine, Beijing, China.
4. Institute for Brain Disorders, Beijing University of Chinese Medicine, Beijing, China.
Abstract
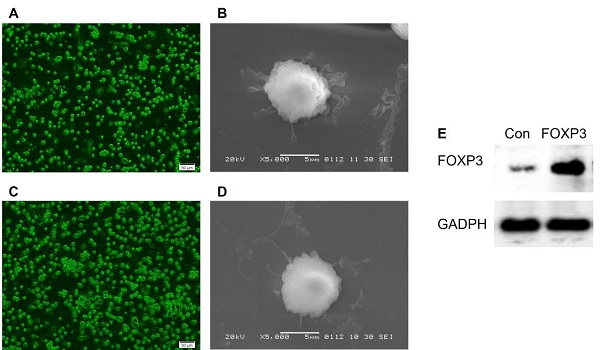
Objective: To investigate the efficiency and potential mechanisms of exosomes from dendritic cells (DCs) transfected with Forkhead box protein P3 (FOXP3) in the development of experimental autoimmune encephalomyelitis (EAE).
Method: Mouse bone marrow-derived immature DCs were loaded with adenovirus carrying FOXP3 gene, and exosomes were generated. Then the exosomes with FOXP3 (FOXP3-EXOs) were co-cultured with CD4+T cell in vitro to evaluate their potential on CD4+T cell proliferation and differentiation, and injected into EAE mice to assess their effects on the development of EAE.
Result: FOXP3-EXOs were effective to inhibit the CD4+T cell proliferation and the production of Interferon gamma (IFN-γ), interleukin (IL)-6, and IL-17, while they promoted the production of IL-10 in vitro. Moreover, FOXP3-EXOs treatment significantly decreased the neurological scores, reduced the infiltration of inflammatory cells into the spinal cord, and decreased demyelination in comparison to saline and Con-EXOs treated EAE mice. Moreover, the FOXP3-EXOs treatment resulted in obvious increases in the levels of regulatory T (Treg) cells and IL-10, whereas levels of T helper 1 (Th1) cells, Th17 cells, IFN-γ, IL-6, and IL-17 decreased significantly in the splenocyte culture of EAE mice.
Conclusion: The present study preliminarily investigated the effects and potential mechanisms of FOXP3-EXOs in EAE and revealed that the FOXP3-EXOs could inhibit the production of Th1 and Th17 cells and promote the production of Treg cells as well as ameliorate the development of EAE. The neuroprotective effects of FOXP3-EXOs on EAE are likely due to the regulation of Th/Treg balance.
Keywords: Multiple sclerosis, experimental autoimmune encephalomyelitis, exosomes, dendritic cell, FOXP3

 Global reach, higher impact
Global reach, higher impact