ISSN: 1449-1907
Int J Med Sci 2022; 19(7):1131-1137. doi:10.7150/ijms.72241 This issue Cite
Research Paper
Catechin-7-O-α-L-rhamnopyranoside can reduce α-MSH-induced melanogenesis in B16F10 melanoma cells through competitive inhibition of tyrosinase
1. College of Pharmacy and Research Institute of Pharmaceutical Sciences, Gyeongsang National University, Jinju 52828, Republic of Korea.
2. Department of Orthopedic Surgery and Institute of Health Sciences, School of Medicine and Hospital, Gyeongsang National University, Jinju 52727, Korea Republic of Korea.
3. Axigen Co. Ltd., Seoul, Republic of Korea.
Abstract
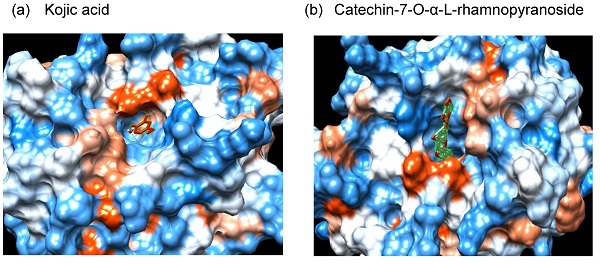
Although melanogenesis is a defense mechanism against ultraviolet (UV)-induced skin damage, abnormally excessive melanin production causes pigmentation disorders. Tyrosinase, as a key factor for melanin synthesis, plays an important role in inducing skin pigmentation. Therefore, the inhibition of tyrosinase is crucial in preventing skin pigmentation in the cosmetics and medicine fields. However, the majority of well-known tyrosinase inhibitors have been discontinued due to toxic effects on the skin or lack of selectivity and/or stability. In this study, we evaluated possible anti-melanogenic effects of catechin-7-O-α-L-rhamnopyranoside (C7R) isolated from the stem bark of Ulmus parvifolia, to discover a new tyrosinase inhibitor that has both safety and stability. When C7R was pretreated in B16F10 melanoma cells stimulated by α-melanocyte-stimulating hormone, this compound reduced melanin accumulation and murine tyrosinase activity. In line with these results, C7R inhibits tyrosinase purified from a mushroom in vitro like kojic acid and arbutin. Furthermore, C7R exhibited a competitive inhibition on a Lineweaver-Burk plot. Next, the underlying mechanisms of the C7R-mediated tyrosinase inhibitory effect were sought through docking simulation and pharmacophore analysis between tyrosinase residues and C7R. The results of these analyses showed that C7R had binding energy of -14.5kcal/mol, and indicated that C7R interacts with tyrosinase through an aromatic ring and various hydrophobic and hydrogen bonds. Together, our results suggest that C7R can be applied as a novel natural anti-melanogenic agent that inhibits tyrosinase.
Keywords: Anti-melanogenic agent, Catechin-7-O-α-L-rhamnopyranoside, Melanogenesis, Skin aging, Tyrosinase, α-MSH

 Global reach, higher impact
Global reach, higher impact