Impact Factor ISSN: 1449-1907
Int J Med Sci 2021; 18(11):2262-2268. doi:10.7150/ijms.53945 This issue Cite
Research Paper
Term Newborns with relatively low Tissue Oxygen Saturation Levels soon after Birth are predisposed to Neonatal Respiratory Disorders in Low-risk, Elective Cesarean Sections
1. Department of Obstetrics and Gynecology, Hamamatsu University School of Medicine, Hamamatsu, Japan.
2. Department of Electrical and Electronics Engineering, Shizuoka University, Hamamatsu, Japan.
3. Research Institute of Electronics, Shizuoka University, Hamamatsu, Japan.
Received 2020-9-30; Accepted 2021-3-5; Published 2021-3-30
Abstract
Background: Neonatal respiratory disorders, such as transient tachypnea of the newborn and respiratory distress syndrome, occur frequently after an elective cesarean delivery. Although conventional pulse oximetry is recommended for neonatal resuscitation, it often requires several minutes after birth to obtain a reliable signal. In a previous study, we used novel tissue oximetry equipment to detect fetal and neonatal early tissue oxygen saturation (StO2) before and immediately after vaginal delivery. Therefore, we hypothesized that low neonatal StO2 levels measured by tissue oximetry may lead to neonatal respiratory disorder after a scheduled cesarean delivery. Hence, this study aimed to evaluate the StO2 levels measured by tissue oximetry in neonates with or without a respiratory disorder subsequently diagnosed after an elective cesarean delivery.
Materials and methods: We enrolled 78 pregnant Japanese women who underwent an elective cesarean section at ≥36 weeks' gestation. After combined spinal and epidural anesthesia were administered to the mother, fetal StO2 levels were measured by tissue oximetry using an examiner's finger-mounted sensor during a pelvic examination immediately before the cesarean section. We measured the neonatal StO2 levels at 1, 3, and 5 minutes after birth and retrospectively compared the fetal and neonatal StO2 levels with the incidence of subsequent diagnoses of neonatal respiratory disorders.
Results: The data of StO2 levels in 35 neonates were collected. Seven neonates (respiratory disorder (RD) group) were subsequently diagnosed with respiratory disorders by neonatal medicine specialists, whereas the 28 remaining neonates (NR group) were not. The median fetal StO2 (interquartile range) of the RD and NR groups was 52.0% (41.8%-60.8%) and 42.5% (39.0%-52.5%), respectively (P = 0.12). The median neonatal StO2 (interquartile range) of the RD and NR groups at 1 minute after birth was 42.0% (39.0%-44.0%) and 46.0% (42.0%-49.0%), respectively (P = 0.091). At 3 minutes after birth, the median neonatal StO2 (interquartile range) of the RD and NR groups was 41.0% (39.0%-46.0%) and 47.0% (44.3%-53.5%), respectively (P = 0.004). Finally, at 5 minutes after birth, the median neonatal StO2 (interquartile range) of the RD and NR groups was 45.0% (44.0%-52.0%) and 54.0% (49.3%-57.0%), respectively (P = 0.007).
Conclusions: The StO2 values in the RD group were lower than those in the NR group at 3 and 5 minutes after birth, suggesting that neonates with low StO2 levels soon after birth may be predisposed to clinically diagnosed neonatal respiratory disorders.
Keywords: fetal tissue oximetry, fetal tissue oxygen saturation, near-infrared spectroscopy, parturition
Introduction
Compared with term vaginal delivery, elective cesarean delivery is a risk factor for neonatal respiratory disorders such as transient tachypnea of the newborn (TTN) and respiratory distress syndrome [1,2]. Exposure to labor may initiate processes that enhance the resorption and clearance of alveolar fluid, thereby reducing the risk of neonatal respiratory disorders [3]. In a retrospective cohort study of 29 669 deliveries, TTN occurred approximately 3 times more often after a scheduled cesarean delivery than after a vaginal delivery (3.1% vs. 1.1%) [1]. However, diagnostic criteria or reliable tests on resuscitation involving neonatal respiratory disorders have not been completely established, especially given the high incidence of neonatal respiratory disorders after elective cesarean delivery [4]. Although TTN is a common disorder, it is diagnosed clinically by exclusion; in clinical studies, the “diagnostic criteria of transient tachypnea of the newborn included tachypnea and imaging studies characterized by nonspecific signs” [5].
The 2015 American Heart Association/American Academy of Pediatrics/International Liaison Committee on Resuscitation (AHA/AAP/ILCOR) guidelines recommend the use of pulse oximetry for measuring oxygen saturation (SpO2) during resuscitation because neonatal appearance and skin color (i.e., cyanosis) are poor indicators of neonatal hypoxia [6,7]. However, pulse oximetry does not provide an accurate signal immediately after birth [7-10]. The failure of pulse oximetry to obtain blood oxygen saturation (SpO2) measurements within 5 minutes of life has been pointed out [8,9]. Moreover, van Vonderen [11] reported that during the first 2 minutes after birth, pulse oximetry frequently displays a newborn heart rate that differs from that obtained by a simultaneous electrocardiogram.
We recently developed a noninvasive fetal and neonatal tissue oximetry system that uses an examiner's finger-mounted near-infrared spectroscopy sensor (NIRS) (KN-15, ASTEM Co., Ltd., Kawasaki, Japan). This 1 cm sensor provides immediate tissue oxygen saturation (StO2) data when an examiner touches the probe to an arbitrary site [12]. This novel device has been validated across various settings, such as animal experiments [13,14], fetal measurements [15,16], neonatal measurements [16,17], and limb measurements [18]. Uchida et al. [15] reported that fetal StO2 in the second stage of term labor significantly correlated with umbilical arterial pH analyzed after delivery. Moreover, Mukai et al. [16] reported a longitudinal assessment of StO2 using this novel device from the fetal to neonatal periods during vaginal delivery. Watanabe et al. [17] compared the neonatal StO2 measurements using this novel device with the SpO2 measurements obtained by pulse oximetry and found that the novel device provided a more stable measurement than pulse oximetry did.
In the present study, we hypothesized that fetal and neonatal StO2 levels measured by tissue oximetry using an examiner's finger-mounted NIRS sensor are low among neonates diagnosed with respiratory disorders after low-risk, term cesarean deliveries. This study specifically aimed to longitudinally measure fetal and neonatal StO2 levels (immediately before delivery and 1, 3, and 5 minutes after birth) in low-risk, term elective cesarean sections.
Materials and Methods
Participants
We conducted a nested case-control study and recruited pregnant women who underwent elective cesarean sections in the Perinatal and Neonatal Care Center in the Hamamatsu University Hospital from May 2016 to March 2018. We included elective cesarean deliveries performed ≥ 36 weeks, 0 days of gestation. The indications for an elective cesarean section included a previous uterine operation, breech presentation, or twin pregnancy. Conversely, the exclusion criteria were as follows: 1) rupture of membranes before cesarean section, 2) labor prior to cesarean section, 3) intact cervical os, 4) placenta previa detected by vaginal ultrasound, 5) twin pregnancy, 6) fetal malformations, or 7) nonreassuring fetal status or maternal complications before delivery.
Instruments
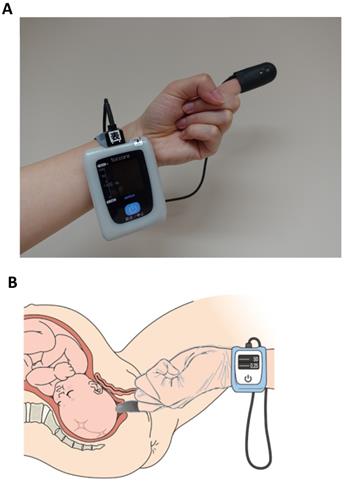
We utilized a novel tissue oximetry system that measures regional StO2 levels noninvasively using the spatially resolved NIRS. The wavelengths of the light from the light-emitting diodes (LEDs) were 770 and 830 nm (KN-15, ASTEM Co., Ltd., Kawasaki, Japan) (Figure 1A). A probe containing a sensor was mounted on an examiner's finger and covered by a sterile polyethylene glove (Singer disposable gloves, Utsunomiya Seisaku Co., Ltd., Osaka, Japan) to protect against infection. Light detectors were located 6 and 8 mm from the LEDs. We determined the oxyhemoglobin, deoxyhemoglobin, and StO2 values by using the spatially resolved NIRS [12]. The novel tissue oximetry device enabled us to noninvasively and immediately measure regional StO2 levels when the examiner touched the finger-mounted sensor to a target region, such as the fetal and neonatal head [15-17].
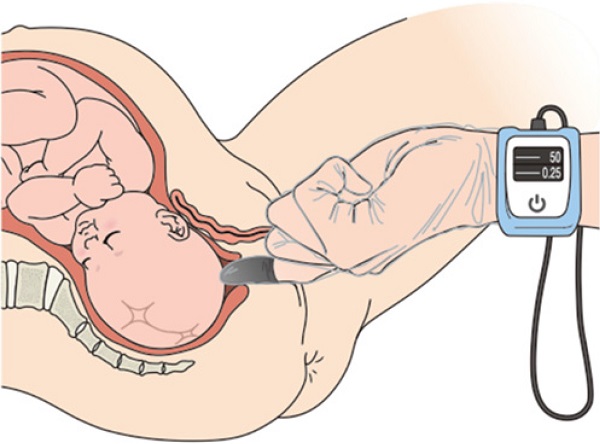
Tissue oximetry probe mounted on an examiner's finger (panel A). The examiner touches the probe to the fetus during a pelvic examination to measure the fetal tissue oxygen saturation levels (panel B). Panel B is cited by [27] Uchida T, Kanayama N, Kawai K, et al. Reevaluation of intrapartum fetal monitoring using fetal oximetry: A review. J Obstet Gynaecol Res. 2018;44(12):2127-2134. Copyright © 2018 Japan Society of Obstetrics and Gynecology. Re-use permitted.
Measurement protocol
Fetal and neonatal StO2 levels were measured for at least 5 seconds until the values stabilized. The values were measured after the combined spinal and epidural anesthesia was administered to the mother before the cesarean section (Figure 1B) and at 1, 3, and 5 minutes after delivery [15-17]. Because the fetal StO2 levels were measured during an elective cesarean section before the onset of labor, some cervical canals were still immature and partially opened. Therefore, probes mounted on the examiner's finger might have touched the maternal tissues (StO2 levels around 60%) instead of the fetal tissues (around 40%). Therefore, we measured the StO2 levels of the uterine cervix separately and excluded the subjects if the StO2 levels were more than 55% by analyzing the receiver operating character (ROC) curve (Supplementary material). All the subjects were measured by a maternal-fetal medicine specialist (KK). Routine neonatal care was given according to the 2015 AHA/AAP/ILCOR guidelines. Neonates who were clinically determined by neonatal specialists as needing any respiratory support on admission were retrospectively classified as belonging to the group with neonatal respiratory orders (the RD group), whereas the remaining neonates were classified as belonging to the NR group.
Statistics
The ratio scale values were expressed as median and interquartile ranges. The nominal scale values were expressed as numbers and percentages. Significant differences between 2 values were assessed using the Welch t-test or the Fisher exact test. Chronological changes of the values were examined using the Friedman test with the Wilcoxon signed-rank test for post hoc analysis adjusted with the Holm method. The cutoff values for StO2 were obtained by calculating the maximal Youden's J statistic in ROC analysis. The intra-rater and inter-rater reliability values were expressed using the intraclass correlation coefficient (ICC). Statistical analysis was performed using the statistical software R version 3.4.1 (R Core Team [2017]; R: A language and environment for statistical computing; R Foundation for Statistical Computing, Vienna, Austria. URL https://www.R-project.org/) and JMP version 13.2.1 (SAS Institute Inc., Cary, NC, USA).
Ethical considerations
This research related to human use complied with all the relevant national regulations and institutional policies, and in accordance with the tenets of the Helsinki Declaration, was approved by the authors' institutional review board. The Ethics Committee of the Hamamatsu University School of Medicine (Registration number 19-048) approved this study, and all participants provided written informed consent.
Results
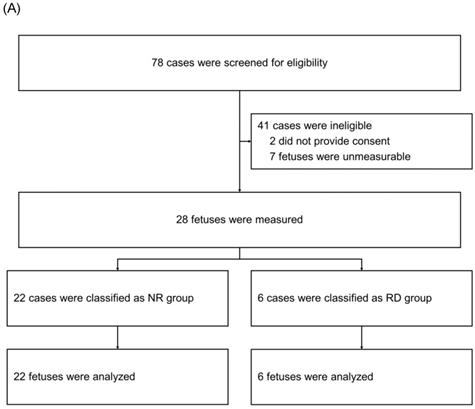
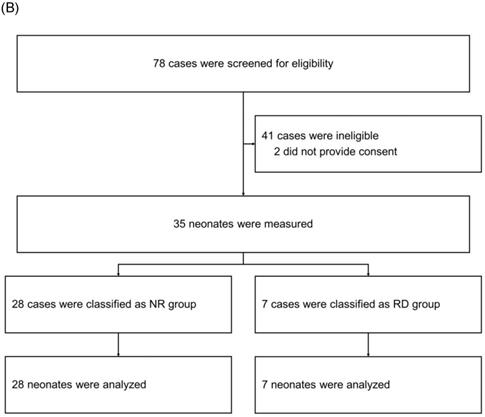
After screening 78 mothers for eligibility, we subsequently enrolled 35 mothers (Figure 2). Among the 35 neonatal cases, 7 were retrospectively classified as having a neonatal respiratory disorder (RD group), whereas the remaining 28 were categorized as having no such disorder (NR group). We compared the fetal and neonatal StO2 of both groups (Table 1, Figure 3A). Neonatal StO2 values were obtained in all 35 neonates; however, fetal StO2 values were only obtained in 28 neonates (RD group, n = 6; NR group, n = 22). Perinatal background, such as maternal age, gestational weeks, birth weight, umbilical arterial pH, and Apgar scores, were similar between the RD and NR groups. Notably, 85.7% of neonates in the RD group required resuscitation, including continuous positive airway pressure, positive pressure ventilation, oxygen supplementation, and/or intubation, but only 17.9% of those in the NR group required resuscitation (Table 1).
Enrollment and classification of fetuses (panel A) and neonates (panel B). NR Group: fetuses and neonates who were not subsequently diagnosed with respiratory disorders; RD Group: fetuses and neonates who were subsequently diagnosed with respiratory disorders.
Comparison of the baseline characteristics and measured data of fetuses and neonates who were subsequently diagnosed with a respiratory disorder (RD group) and those who were not (NR group)
| NR Group (n = 28) | RD Group (n = 7) | P value | |
|---|---|---|---|
| A. Characteristics | |||
| Maternal age, years | 35.0 (33.0-37.0) | 32.0 (26.0-40.0) | 0.52 |
| Gestational age, weeks | 37.9 (37.6-38.1) | 38.0 (37.7-38.3) | 0.33 |
| Birth weight, g | 2878 (2725-3177) | 2854 (2818-3068) | 0.95 |
| Umbilical arterial pH | 7.308 (7.282-7.334) | 7.332 (7.309-7.336) | 0.15 |
| Apgar score at 1 min | 8.0 (8.0-8.0) | 8.0 (8.0-8.0) | 0.84 |
| Apgar score at 5 min | 9.0 (9.0-9.0) | 9.0 (8.0-9.0) | 0.61 |
| Cervical dilation, cm | 2.0 (1.0-2.5) | 1.5 (1.0-2.0) | 0.66 |
| Neonatal resuscitation, n | 5 (17.9) | 6 (85.7) | 0.002 |
| Cephalic presentation, n | 20 (71.4) | 6 (85.7) | 0.65 |
| B. Measured data | |||
| StO2 of fetus, % | 42.5 (39.0-52.5)a | 52.0 (41.8-60.8)b | 0.12 |
| StO2 at 1 min after delivery, % | 46.0 (42.0-49.0) | 42.0 (39.0-44.0) | 0.091 |
| StO2 at 3 min after delivery, % | 47.0 (44.3-53.5) | 41.0 (39.0-46.0) | 0.004 |
| StO2 at 5 min after delivery, % | 54.0 (49.3-57.0) | 45.0 (44.0-52.0) | 0.007 |
Values are presented as median with interquartile range in parentheses, except for cephalic presentation, which shows n with percentage in parentheses. P values were calculated with the Welch t-test for all variables, except for cephalic presentation, where the Fisher exact test was used, an = 22, bn = 6.
NR Group: fetuses and neonates who were not subsequently diagnosed with respiratory disorders; RD Group: fetuses and neonates who were subsequently diagnosed with respiratory disorders; StO2: tissue oxygen saturation. Neonatal resuscitation includes continuous positive airway pressure, positive pressure ventilation, oxygen supplementation, and/or intubation.
Tissue oxygen saturation (StO2) values measured in fetuses and neonates who were subsequently diagnosed with or without respiratory disorders (RD and NR groups, respectively) at 1, 3, and 5 minutes after delivery. Fetal StO2 values were not significantly different between the 2 groups. At 3 and 5 minutes after delivery, the NR group had significantly higher StO2 values than the RD group (panel A). StO2 values measured in the NR group increased significantly (panel B), whereas those in the RD group did not change significantly (panel C). Box plots represent the lowest datum that is still within 1.5 interquartile range (IQR) of the lower quartile, the lower quartile, the median, and the upper quartile; the highest datum was still within 1.5 IQR of the upper quartile. Gray boxes represent the RD group, and the white boxes represent the NR group. P values were calculated using the Welch t-test or the Friedman test, and the Wilcoxon signed-rank test for post hoc analysis adjusted with the Holm method.
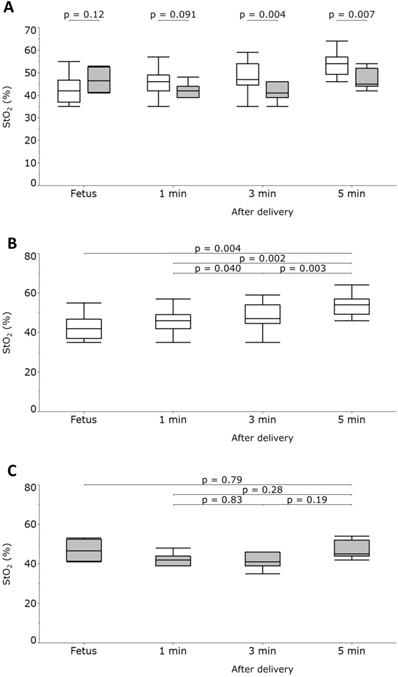
There were no statistically significant differences in fetal StO2 values or neonatal StO2 values at 1 minute after birth between the two groups (Table 1, Figure 3A).
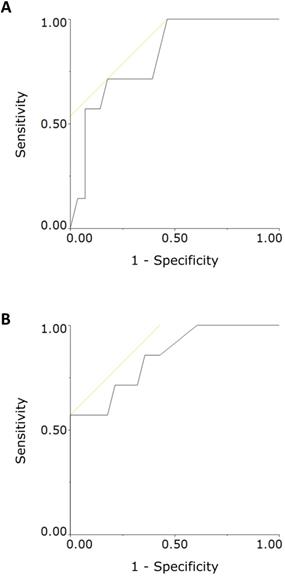
The longitudinal changes of the NR group's neonatal StO2 values were similar to those observed in our previous report [16,17]. However, the RD group had significantly lower neonatal StO2 values at 3 and 5 minutes after birth (Table 1, Figure 3A). Moreover, the NR group's neonatal StO2 values increased chronologically, whereas those of the RD group did not (Figure 3B, 3C). For the RD group, ROC analysis showed the cutoff values of StO2 at 3 and 5 minutes after delivery were 43%, and 45%, respectively (Figure 4). The intra-rater reliability for neonatal StO2 using the ICC (1, 1) and (1, 5) was 0.88 and 0.97, respectively, whereas the inter-rater reliability for neonatal StO2 using the ICC (3, 1) was 0.71.
Receiver operating characteristic (ROC) curve analysis of neonatal tissue oxygen saturation (StO2) values measured at 3 minutes (panel A) and 5 minutes (panel B) after birth. The cutoff values of 43% and 45% for distinguishing neonates who were and were not subsequently diagnosed with respiratory disorders were obtained by calculating the maximal Youden's J statistic in ROC curve analysis. The areas under the curve were 0.821 and 0.849 for the 43% and 45% cutoff values, respectively.
Discussion
Although we could not measure StO2 in some fetuses because of unfavorable cervical maturation, we successfully measured StO2 in all neonates at 1, 3, and 5 minutes after birth, indicating StO2 is a stable and reliable way to assess neonatal oxygenation soon after delivery. This study demonstrated that newborns who were clinically diagnosed with subsequent neonatal respiratory disorders had significantly lower neonatal StO2 values at 3 and 5 minutes after birth compared with those newborns without respiratory disorders (Figure 3). Moreover, the StO2 values before and after cesarean delivery did not increase chronologically in neonates with respiratory disorders (Figure 3). Additionally, there were no significant differences in fetal StO2 values before the elective cesarean sections between neonates with or without subsequently diagnosed neonatal respiratory disorders.
The 2015 AHA/AAP/ILCOR guidelines recommend the use of conventional pulse oximetry during resuscitation; however, pulse oximetry takes several minutes to obtain a reliable signal, especially immediately after birth [7-10]. Therefore, this method is not considered diagnostically useful because it remains unclear whether neonatal oxygenation levels during the first several minutes after birth as measured by pulse oximetry are reliable. Watanabe et al. [17] demonstrated that reliable StO2 levels measured by tissue oximetry (KN-15) could be obtained earlier than SpO2 levels measured by conventional pulse oximetry. We recently reported the ability to measure neonatal StO2 at 5 minutes after vaginal delivery [16], but in the present study, we successfully measured neonatal StO2 in all cases at 1, 3, and 5 minutes after cesarean birth. In the previous studies using tissue oximetry (KN-15), the mean or median value of StO2 at 5 minutes after vaginal delivery was approximately 55% [16,17], which was similar to the median value of 54.0% at 5 minutes after cesarean delivery in the current study's NR group. Meanwhile, the reference ranges for neonatal SpO2 in cesarean births were lower than those in vaginal births [9,19,20]. The reference value for SpO2 at 5 minutes after cesarean birth was reportedly 85% (60%-94%) [20]. Similar devices using NIRS have also been used for measuring neonatal oxygenation immediately after birth; however, not all the values obtained by such devices have been reliable [21-23]. Median values for the cerebral tissue oxygen saturation index (NIRO-200NX, Hamamatsu Photonics K.K., Hamamatsu, Japan) and for the regional cerebral tissue oxygen saturation (INVOS 5100, Somanetics Corp, Troy, MI, USA) were 65.6% and 66.8%, respectively. These values were higher than the StO2 values [22,23].
In this study, the neonatal StO2 values measured at 3 and 5 minutes after birth were significantly lower in neonates clinically diagnosed with subsequent neonatal respiratory disorders than in those without respiratory disorders. As mentioned, elective cesarean delivery at term is a risk factor for developing neonatal respiratory disorders such as TTN and respiratory distress syndrome as compared with vaginal delivery at term [1,2]. However, TTN has had no established criteria for diagnosis and continues to be diagnosed multilaterally according to clinical findings such as tachypnea and respiratory distress and imaging findings such as chest radiograph and ultrasonography [4,5,24,25]. In a previous study, there were no significant differences in neonatal SpO2 values at 10 minutes after birth between neonates with TTN and those in a control group [26]. The present findings suggest that the cutoff values of 43% (3 minutes), and/or 45% (5 minutes) might be useful in identifying newborns at high risk for developing subsequent neonatal respiratory disorders after cesarean delivery (Figure 4). A large multicenter study is necessary to reconfirm the cutoff values. The present findings suggest that the assessment of StO2 within 5 minutes after birth might provide guidance to help improve neonatal resuscitation, especially after an elective cesarean section. We have no clear explanation as to why fetal StO2 values in the RD group were higher than those in the NR group.
The present study, however, has some limitations. First, neither the neonatal SpO2 nor the partial pressure of arterial oxygen was available because the subjects were scheduled for low-risk elective cesarean sections. Second, some newborns in the RD group required resuscitation, including supplemental oxygen and positive pressure ventilation, while fewer newborns in the NR group required resuscitation, thereby possibly affecting the difference in StO2 levels. The StO2 values were measured independently by the author and concealed from the clinicians. Third, we only enrolled cases of elective cesarean section; therefore, further studies are necessary in cases of emergency cesarean section and vaginal delivery. Moreover, the present subjects underwent elective cesarean section without any clinical findings of nonreassuring fetal status. Our research group is conducting an ongoing study of the predictive value of low StO2 before an emergency cesarean section in cases of nonreassuring fetal status.
In conclusion, neonatal StO2 data were obtained in all cases after scheduled cesarean delivery. Neonatal StO2 values measured at 3 and 5 minutes after birth were significantly lower in neonates who were subsequently diagnosed with neonatal respiratory disorders than in those who were not, suggesting that neonates with low StO2 levels soon after birth might be predisposed to subsequent clinically diagnosed neonatal respiratory disorders.
Supplementary Material
Supplementary materials and figures.
Acknowledgements
The authors thank Ms. Naoko Hakamada, Ms. Yumiko Yamamoto, Ms. Naoko Kondo, Ms. Miuta Sawai, Ms. Kazuko Sugiyama, and Ms. Shiori Ebisuno for their secretarial or technical assistance.
Funding
This work was supported by the JSPS KAKENHI under Grant Number JP15K10667, JP18K09226.
Competing Interests
The authors have declared that no competing interest exists.
References
1. Levine EM, Ghai V, Barton JJ. et al. Mode of delivery and risk of respiratory diseases in newborns. Obstet Gynecol. 2001;97(3):439-442
2. Gerten KA, Coonrod DV, Bay RC. et al. Cesarean delivery and respiratory distress syndrome: does labor make a difference? Am J Obstet Gynecol. 2005;193(3):1061-1064
3. Avery ME, Gatewood OB, Brumley G. Transient tachypnea of newborn. Possible delayed resorption of fluid at birth. Am J Dis Child. 1966;111(4):380-385
4. Guglani L, Lakshminrusimha S, Ryan RM. Transient tachypnea of the newborn. Pediatr Rev. 2008;29(11):e59-65
5. Moresco L, Calevo MG, Baldi F. et al. Epinephrine for transient tachypnea of the newborn. Cochrane Database Syst Rev. 2016(5):CD011877
6. Wyckoff MH, Aziz K, Escobedo MB. et al. Part 13: Neonatal resuscitation: 2015 American Heart Association guidelines update for cardiopulmonary resuscitation and emergency cardiovascular care. Circulation. 2015;132(18):S543-560
7. Perlman JM, Wyllie J, Kattwinkel J, et al; Neonatal resuscitation chapter collaborators. Part 7: Neonatal resuscitation: 2015 international consensus on cardiopulmonary resuscitation and emergency cardiovascular care science with treatment recommendations. Circulation. 2015;132(16):S204-241
8. Dawson JA, Morley CJ. Monitoring oxygen saturation and heart rate in the early neonatal period. Semin Fetal Neonatal Med. 2010;15(4):203-207
9. Kamlin CO, O'Donnell CP, Davis PG. et al. Oxygen saturation in healthy infants immediately after birth. J Pediatr. 2006;148(5):585-589
10. Louis D, Sundaram V, Kumar P. Pulse oximeter sensor application during neonatal resuscitation: a randomized controlled trial. Pediatrics. 2014;133(3):476-482
11. van Vonderen JJ, Hooper SB, Kroese JK. et al. Pulse oximetry measures a lower heart rate at birth compared with electrocardiography. J Pediatr. 2015;166(1):49-53
12. Kanayama N, Niwayama M. Examiner's finger-mounted fetal tissue oximetry. J Biomed Opt. 2014;19(6):067008
13. Uchida T, Kanayama N, Kawai K. et al. Craniofacial tissue oxygen saturation is associated with blood pH using an examiner's finger-mounted tissue oximetry in mice. J Biomed Opt. 2016;21(4):40502
14. Hiwatashi K, Doi K, Mizuno R. et al. Examiner's finger-mounted near-infrared spectroscopy is feasible to analyze cerebral and skeletal muscle oxygenation in conscious Chihuahuas. J Biomed Opt. 2017;22(2):26006
15. Uchida T, Kanayama N, Mukai M. et al. Examiner's finger-mounted fetal tissue oximetry: a preliminary report on 30 cases. J Perinat Med. 2016;44(7):745-749
16. Mukai M, Uchida T, Itoh H. et al. Tissue oxygen saturation levels from fetus to neonate. J Obstet Gynaecol Res. 2017;43(5):855-859
17. Watanabe T, Ito M, Miyake F. et al. Measurement of brain tissue oxygen saturation in term infants using a new portable near-infrared spectroscopy device. Pediatr Int. 2017;59(2):167-170
18. Yata T, Sano M, Kayama T. et al. Utility of a finger-mounted tissue oximeter with near-infrared spectroscopy to evaluate limb ischemia in patients with peripheral arterial disease. Ann Vasc Dis. 2019;12(1):36-43
19. Suwattanaphim S, Yodavuhd S, Puangsa-art S. Time duration of oxygen adaptation immediately after birth; Monitoring by pulse oximeter in perinatal period of the infants at Charoenkrung Pracharak Hospital. J Med Assoc Thai. 2015;98(7):656-663
20. Dawson JA, Kamlin CO, Vento M. et al. Defining the reference range for oxygen saturation for infants after birth. Pediatrics. 2010;125(6):e1340-1347
21. Ziehenberger E, Urlesberger B, Binder-Heschl C. et al. Near-infrared spectroscopy monitoring during immediate transition after birth: time to obtain cerebral tissue oxygenation. J Clin Monit Comput. 2018;32(3):465-469
22. Baik N, Urlesberger B, Schwaberger B. et al. Reference ranges for cerebral tissue oxygen saturation index in term neonates during immediate neonatal transition after birth. Neonatology. 2015;108(4):283-286
23. Pichler G, Binder C, Avian A. et al. Reference ranges for regional cerebral tissue oxygen saturation and fractional oxygen extraction in neonates during immediate transition after birth. J Pediatr. 2013;163(6):1558-1563
24. Onal EE, Dilmen U, Adam B. et al. Serum atrial natriuretic peptide levels in infants with transient tachypnea of the newborn. J Matern Fetal Neonatal Med. 2005;17(2):145-149
25. Liu J, Chen XX, Li XW. et al. Lung ultrasonography to diagnose transient tachypnea of the newborn. Chest. 2016;149(5):1269-1275
26. Unal S, Ergenekon E, Aktas S. et al. Perfusion index assessment during transition period of newborns: an observational study. BMC Pediatr. 2016;16(1):164
27. Uchida T, Kanayama N, Kawai K. et al. Reevaluation of intrapartum fetal monitoring using fetal oximetry: A review. J Obstet Gynaecol Res. 2018;44(12):2127-2134
Author contact
Corresponding author: Toshiyuki Uchida MD, PhD, Department of Obstetrics and Gynecology, Hamamatsu University School of Medicine, 1-20-1 Handayama, Higashi-ku, Hamamatsu 431-3192, Japan. Phone: +81-53-435-2309, Fax: +81-53-435-2308, E-mail: uttyac.jp.

 Global reach, higher impact
Global reach, higher impact