Impact Factor ISSN: 1449-1907
Int J Med Sci 2021; 18(6):1456-1464. doi:10.7150/ijms.51256 This issue Cite
Research Paper
LY3214996 relieves acquired resistance to sorafenib in hepatocellular carcinoma cells
1. Medical school, Anhui University of Science and Technology, Huainan 232001, China.
2. Institute of Environment-friendly Materials and Occupational Health of Anhui University of Science and Technology (Wuhu), Wuhu, 241003, China.
3. Department of Clinical Laboratory Medicine, The Fourth Affiliated Hospital, Zhejiang University School of Medicine, Yiwu 322000, China.
*These authors contributed equally to this work.
Abstract
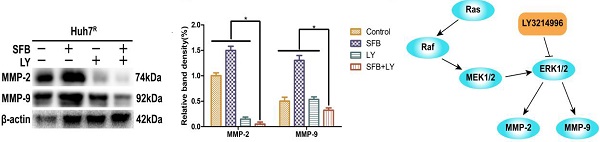
Background: Sorafenib, an oral multi-kinase inhibitor of rapidly accelerated fibrosarcoma; vascular endothelial growth factor receptor-2/3, platelet-derived growth factor receptor, c-Kit, and Flt-3 signaling, is approved for treatment of advanced hepatocellular carcinoma (HCC). However, the benefit of sorafenib is often diminished because of acquired resistance through the reactivation of ERK signaling in sorafenib-resistant HCC cells. In this work, we investigated whether adding LY3214996, a selective ERK1/2 inhibitor, to sorafenib would increase the anti-tumor effectiveness of sorafenib to HCC cells.
Methods: The Huh7 cell line was used as a cell model for treatment with sorafenib, LY3214996, and their combination. Phosphorylation of the key kinases in the Ras/Raf/MAPK and PI3K/Akt pathways, protein expression of the cell cycle, and apoptosis migration were assessed with western blot. MTT and colony-formation assays were used to evaluate cell proliferation. Wound-healing assay was used to assess cell migration. Cell cycle and apoptosis analyses were conducted with flow cytometry.
Results: LY3214996 decreased phosphorylation of the Ras/Raf/MAPK and PI3K/Akt pathways, including p-c-Raf, p-P90RSK, p-S6K and p-eIF4EBP1 activated by sorafenib, despite increased p-ERK1/2 levels. LY3214996 increased the anti-proliferation, anti-migration, cell-cycle progression, and pro-apoptotic effects of sorafenib on Huh7R cells.
Conclusions: Reactivation of ERK1/2 appears to be a molecular mechanism of acquired resistance of HCC to sorafenib. LY3214996 combined with sorafenib enhanced the anti-tumor effects of sorafenib in HCC. These findings form a theoretical basis for trial of LY3214996 combined with sorafenib as second-line treatment of sorafenib-resistant in advanced HCC.
Keywords: sorafenib, LY3214996, hepatocellular carcinoma, Ras/Raf/MAPK pathway, acquired resistance

 Global reach, higher impact
Global reach, higher impact