Impact Factor
ISSN: 1449-1907
Int J Med Sci 2021; 18(6):1382-1389. doi:10.7150/ijms.50919 This issue Cite
Research Paper
Ginsenoside Rg1 alleviates acute liver injury through the induction of autophagy and suppressing NF-κB/NLRP3 inflammasome signaling pathway
1. Department of Infectious Diseases, The First Affiliated Hospital of Chongqing Medical University, Chongqing, 400016, China.
2. Department of Orthopedics, The First Affiliated Hospital of Chongqing Medical University, Chongqing, 400016, China.
3. Clinical Molecular Medicine Testing Center, The First Affiliated Hospital of Chongqing Medical University, Chongqing, 400016, China.
Abstract
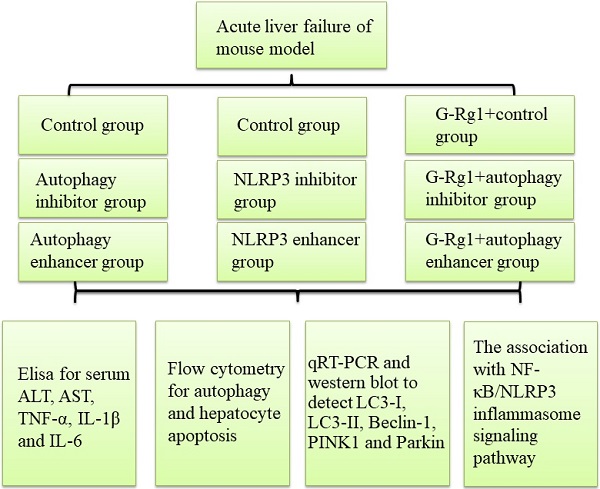
Background: Severe hepatitis is a common cause of chronic or acute liver disease and autophagy might play an important role in cellular response to inflammation and injury. It has been reported that Ginsenoside-Rg1 (G-Rg1) has strong hepatoprotective effects for acute liver injury, but its protective mechanisms have not yet been elucidated. This study aims to explore the detailed molecular mechanisms of G-Rg1 on acute liver injury via autophagy.
Methods: The role of G-Rg1 by autophagic induction was studied in the mouse model of acute liver injury which induced by carbon tetrachloride (CCl4). Liver function, inflammatory reaction and apoptosis were detected when autophagy has been inhibited by 3-MA or stimulated by RPA. MCC950 and ATP were applied to investigate the role of NLRP3 inflammasome in acute liver injury. The differential expression of NF-κB, NLRP3 inflammasome, caspase 1, caspase 3, IL-1β, IL-18, LC3-I, LC3-II, Beclin-1, PINK1 and Parkin have been detected by the quantitative real-time polymerase chain reaction (qRT-PCR) and Western blot.
Results: G-Rg1 could decrease ALT, AST, TNF-α, IL-1β and IL-6 in mice with CCl4-induced acute liver injury. The change of autophagy and apoptosis after the treatment of 3-MA or RPA demonstrated that the autophagy played a key role in the protective effect of G-Rg1 in acute liver injury. The enhancement of G-Rg1 promoted-autophagy resulted in the significant decrease in NF-κB, NLRP3 inflammasome, caspase 1, caspase 3, IL-1β and IL-18, which suggesting that NF-κB/NLRP3 inflammasome signaling pathway was associated with the autophagy induced by G-Rg1 in acute liver injury.
Conclusion: G-Rg1 ameliorated acute liver injury via the autophagy, which may be related to NF-κB/NLRP3 inflammasome signaling pathway.
Keywords: Ginsenoside-Rg1, acute liver injury, autophagy, NF-κB, NLRP3 inflammasome.

 Global reach, higher impact
Global reach, higher impact