Impact Factor ISSN: 1449-1907
Int J Med Sci 2021; 18(1):128-136. doi:10.7150/ijms.48214 This issue Cite
Research Paper
MicroRNAs as potential biomarkers for the diagnosis of Traumatic Brain Injury: A systematic review and meta-analysis
1. Department of Emergency and Trauma Center, The International Medical Center, The First Affiliated Hospital, College of Medicine, Zhejiang University, 79 Qingchun Road, Hangzhou 310003, Zhejiang Province, China.
2. Department of Rehabilitation Medicine, The First Affiliated Hospital, Zhejiang University, 79 Qingchun Road, Hangzhou 310003, Zhejiang Province, China.
Received 2020-5-15; Accepted 2020-10-15; Published 2021-1-1
Abstract
Background: Traumatic brain injury (TBI) is a sudden trauma on the head, in which severe TBI (sTBI) is usually associated with death and long-term disability. MicroRNAs (miRNAs) are potential biomarkers of diverse diseases, including TBI. However, few systematic reviews and meta-analyses have been conducted to determine the clinical value of miRNAs expression in TBI patients.
Methods: We conducted this systematic review and meta-analysis study according to Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines. We searched PubMed, Embase, the Cochrane Library, Web of Science, from inception to August 26, 2020. We included articles written in English that have reported on the diagnostic value of miRNAs expression in TBI patients. We excluded studies that did not provided sufficient information to construct the 2×2 contingency table.
Results: Eight studies investigating the diagnostic value of miRNA in TBI were analyzed in this study. The overall sensitivity, specificity and area under the curve (AUC) of miRNAs in diagnosis of TBI were 89% [95% confidence interval (CI): 0.84-0.93], 92% (95% CI 0.82-0.97) and 95% (95% CI 0.93-0.97). We found that panels of multiple miRNAs could improve the diagnostic accuracy of TBI. Samples from blood and brain tissue have significantly enhanced diagnostic accuracy, when compared with saliva. The AUC of miRNAs in severe TBI was 0.97, with 91% sensitivity and 92% specificity.
Conclusion: This systematic review and meta-analysis demonstrated that miRNAs could be potential diagnostic markers in TBI patients. MiRNAs detected in blood and brain tissue display high accuracy for TBI diagnosis.
Keywords: miRNAs, TBI, diagnosis, biomarker, meta-analysis
Introduction
Traumatic brain injury (TBI) is a prevalent form of nervous system ailment that inflicts more than 50 million people each year worldwide [1]. TBI, particularly severe TBI (sTBI), cause an enormous socio-economic and health care burden because of its associated mortalities and long-term disability among patients. Due to their clinical value in the diagnosis, prognosis and treatment of TBI patients, biomarkers have attracted considerable attention [2].
MicroRNAs (miRNAs) are a class of small non-coding RNAs with a length of 19-22 nucleotides. Many studies have found that miRNAs play a significant role in the maintenance and regulation of physiological function in TBI [3]. Recently, the expression of miRNAs in TBI has been extensively examined. Many studies have revealed that some miRNAs have diagnostic and prognostic value in TBI. For example, Pietrao et al. showed that miR-425-5p is significantly downregulated in mild TBI (mTBI) and is a good diagnostic and prognostic indicator for TBI [4]. Furthermore, Redell et al. detected substantial plasma quantities of miR-16, miR-92a, and miR-765 in sTBI patients, and found that the miRNAs have high diagnostic value in sTBI [5]. Another study proved that miR-3195 and miR-328-5p may be used to distinguish mild and moderate TBI from sTBI [6]. These studies have shown that miRNA can be used to diagnose TBI, including distinguishing sTBI from healthy controls and mild to moderate TBI. However, there are no meta-analyses of the clinical values of miRNAs in TBI patients. In this study, we conducted a meta-analysis to identify the potential diagnostic values of miRNAs in TBI patients. The purpose of this study was to explore the key miRNAs and provide useful information and direction for future study in the clinical value of miRNAs in TBI, such as prognosis of outcome.
Materials and Methods
Search strategy
Relevant studies published before August 26, 2020 was comprehensively searched through the English databases PubMed, Cochrane Library, Web of Science, and EMBASE. We used “TBI” and “miRNA” as the main key words and the following strategy to search PubMed: (((“Brain Injuries, Traumatic”[Mesh]) OR (((((((((((((((Brain Injury, Traumatic[Title/Abstract]) OR Traumatic Brain Injuries[Title/Abstract]) OR Trauma, Brain[Title/Abstract]) OR Brain Trauma[Title/Abstract]) OR Brain Traumas[Title/Abstract]) OR Traumas, Brain[Title/Abstract]) OR TBI (Traumatic Brain Injury)[Title/Abstract]) OR Encephalopathy, Traumatic[Title/Abstract]) OR Encephalopathies, Traumatic[Title/Abstract]) OR Traumatic Encephalopathies[Title/Abstract]) OR Injury, Brain, Traumatic[Title/Abstract]) OR Traumatic Encephalopathy[Title/Abstract]) OR TBIs (Traumatic Brain Injuries)[Title/Abstract]) OR TBI (Traumatic Brain Injuries)[Title/Abstract]) OR Traumatic Brain Injury[Title/Abstract]))) AND ((“MicroRNAs”[Mesh]) OR (((((((((((((((((MicroRNA[Title/Abstract]) OR miRNAs[Title/Abstract]) OR Micro RNA[Title/Abstract]) OR RNA, Micro[Title/Abstract]) OR miRNA[Title/Abstract]) OR Primary MicroRNA[Title/Abstract]) OR MicroRNA, Primary[Title/Abstract]) OR Primary miRNA[Title/Abstract]) OR miRNA, Primary[Title/Abstract]) OR pri-miRNA[Title/Abstract]) OR pri miRNA[Title/Abstract]) OR RNA, Small Temporal[Title/Abstract]) OR Temporal RNA, Small[Title/Abstract]) OR stRNA[Title/Abstract]) OR Small Temporal RNA[Title/Abstract]) OR pre-miRNA[Title/Abstract]) OR pre miRNA[Title/Abstract])).
Eligibility criteria and Data extraction
The inclusion criteria are as follows: (1) articles provided diagnostic capacity of miRNA for TBI; (2) articles provided enough data such as true positives (TP), false positives (FP), false negatives (FN), true negatives (TN) or sensitivity and specificity to construct the 2×2 contingency table; (3) Publications written in English. The exclusion criteria are as follows: (1) Non-TBI or miRNAs researches; (2) Non-English Articles; (3) Animal or cell experiments; (4) meeting records, reviews and letters.
We extracted the following data according to our previous protocol: the first author's name; study population, sample sizes and regions; year of publication; and the false and true positives and negatives [7].
Statistical analysis
Two reviewers (Zhou and Yin) extracted the number of TP, FP, FN and TN, which was provided in the articles. We used the numbers to calculate the pooled sensitivity, specificity, positive likelihood ratio (PLR), negative likelihood ratio (NLR), diagnostic odds ratio (DOR) and corresponding 95% confidence intervals (CIs). The summary receiver operating characteristic (SROC) curve and the area under the SROC curve (AUC) were also calculated to evaluate the pooled diagnostic value of miRNAs. We did not test for the publication bias because only seven articles were ultimately included in this meta-analysis. We used the chi-square and I2 tests to access heterogeneity in this study. If P < 0.1 or I2 > 50%, we defined heterogeneity as significant and would conduct meta-regression, subgroup and sensitivity analyses to discover the sources of heterogeneity. We performed all statistical analyses using Stata 12.0 (StataCorp, College Station, TX, USA). We defined P<0.05 as statistically significant.
Results
Study characteristics
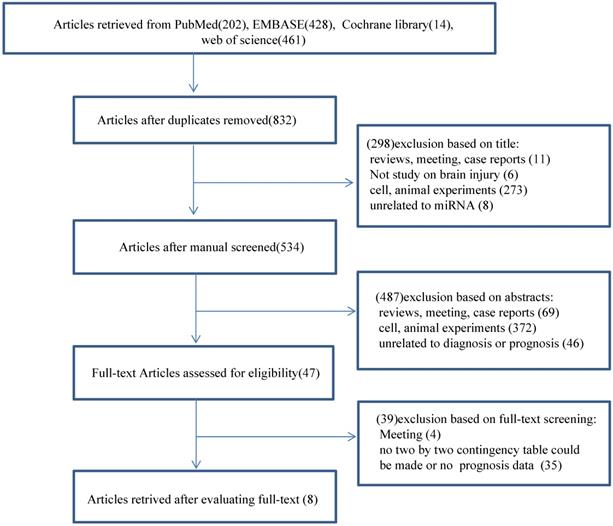
We did a search through PubMed, EMBASE, the Cochrane Library and Web of Science and identified 1105 records. Among these records, 273 were duplicate studies and were, therefore, excluded. We excluded 298 articles after reading the titles and another 487 publications after reviewing the abstracts (Fig. 1). The remaining 47 full-text articles were assessed for relevance according to our pre-determined inclusion and exclusion criteria. Subsequently, we excluded 39 publications, including four meetings and 35 without clinical data, and made a two-by-two contingency table to calculate sensitivity, specificity and a corresponding CI. We attempted to calculate pooled hazard ratios (HRs) to analyze the prognostic performance of miRNAs; however, we only extracted enough data to calculate the diagnostic value of miRNAs. Eight diagnostic-related articles were ultimately included in this study [4, 5, 8-13]. A flow chart of the selection process for this study presented in Fig. 1.
Flow diagram of the study selection for the meta-analysis.
These eight articles included (ranging from the year 2010 to 2020) reported 44 experiments, including different single miRNAs and panel miRNAs (Tables 1 & 2). Those articles totally included 215 TBI patients and 152 controls composed of healthy controls and other diseases (Table 1). Among the 44 experiments, 29 reported a single miRNA, while the additional 15 discussed a panel miRNAs (Table 2). Out of the 44 experiments, ten detected miRNA in plasma, twelve detected miRNA in serum, one detected miRNA in blood and urine, six detected miRNA in saliva, one identified miRNA in brain‐derived extracellular vesicles, and six evaluated the brain tissue. Of the 8 articles, the populations of 6 studies were Caucasian, whereas two studies were Asian. 24 experiments conducted in severe TBI patients, eleven in mild TBI patients, and the remaining nine experiments focused on TBI patients.
Characteristics of studies included in meta-analysis
| First author | Publish year | Ethnicity | Patients | Controls | Patients/controls | miRNAS | Detected sample |
|---|---|---|---|---|---|---|---|
| Schober | 2014 | Caucasian | Severe TBI | Non-TBI | 8/7 | miR-138, miR-504, miR-16, miR-376a, miR-195, miR-370, miR-320b, miR-135b; miR-744, miR-324-5p, miR-455-3p, let-7a, miR-193b. | Cerebellar tissue |
| Di Pietro | 2017 | Caucasian | Mild TBI | healthy volunteers | 30/30 | miR-425-5p, miR-502, miR-335 | Serum |
| Di Pietro | 2018 | Caucasian | Concussion | healthy volunteers | 22/10 | let-7i-5p, miR-142-3p, miR-107, miR-27b-3p, miR-135b-5p. | Salivary |
| Redell | 2010 | Caucasian | Severe TBI/mild TBI | healthy volunteers/orthopedic injury patients. | 18/16 | miR-16, miR-92a, miR-765. | Plasma |
| Ko | 2018 | Caucasian | TBI | Healthy controls | 16/20 | miR‐203b‐5p, miR‐203a‐3p, miR‐206, miR‐185‐5p | EV |
| Yang | 2016 | Asian | TBI | Healthy controls | 76/38 | miR-93 | serum |
| Pan | 2019 | Asian | TBI | Non-TBI | 35/21 | miR-155 | Blood and urine |
| O'Connell | 2020 | Caucasian | TBI | Healthy controls | 10/10 | miR-124-3p, miR-219a-5p, miR-9-5p, miR-9-3p, miR-137, and miR-128-3p | Serum |
TBI: traumatic brain injury; EV: brain‐derived extracellular vesicles.
False and true positives and negatives of total 44 experiments from 8 included articles
| First author | Year | miRNA(s) | TP | FP | FN | TN |
|---|---|---|---|---|---|---|
| Single miRNA | ||||||
| Schober | 2014 | miR-138 | 6 | 0 | 2 | 7 |
| Schober | 2014 | miR-504 | 6 | 1 | 1 | 5 |
| Schober | 2014 | miR-16 | 8 | 1 | 0 | 3 |
| Schober | 2014 | miR-376a | 8 | 3 | 0 | 4 |
| Schober | 2014 | miR-195 | 6 | 0 | 2 | 7 |
| Schober | 2014 | miR-370 | 6 | 0 | 1 | 7 |
| Schober | 2014 | miR-320b | 7 | 3 | 0 | 3 |
| Schober | 2014 | miR-135b | 6 | 1 | 2 | 6 |
| Schober | 2014 | miR-744 | 7 | 2 | 1 | 5 |
| Schober | 2014 | miR-324-5p | 7 | 1 | 1 | 6 |
| Schober | 2014 | miR-455-3p | 5 | 0 | 3 | 7 |
| Schober | 2014 | let-7a | 7 | 2 | 1 | 5 |
| Schober | 2014 | miR-193b | 7 | 1 | 1 | 6 |
| Di Pietro | 2017 | miR-425-5p | 30 | 0 | 0 | 30 |
| Di Pietro | 2017 | miR-502 | 30 | 0 | 0 | 30 |
| Di Pietro | 2017 | miR-335 | 30 | 0 | 0 | 30 |
| Di Pietro | 2018 | let-7i-5p | 19 | 9 | 3 | 1 |
| Di Pietro | 2018 | miR-142-3p | 16 | 8 | 6 | 2 |
| Di Pietro | 2018 | miR-107 | 15 | 9 | 7 | 1 |
| Di Pietro | 2018 | miR-27b-3p | 15 | 7 | 7 | 3 |
| Di Pietro | 2018 | miR-135b-5p | 16 | 8 | 6 | 2 |
| Yang | 2016 | miR-93 | 76 | 0 | 0 | 38 |
| Yang | 2016 | miR-93 | 25 | 0 | 0 | 38 |
| Pan | 2019 | miR-155 | 27 | 7 | 8 | 14 |
| O'Connell | 2020 | miR-124-3p | 5 | 1 | 5 | 9 |
| O'Connell | 2020 | miR-219a-5p | 8 | 1 | 2 | 9 |
| O'Connell | 2020 | miR-9-5p | 8 | 0 | 2 | 10 |
| O'Connell | 2020 | miR-9-3p | 7 | 0 | 3 | 10 |
| O'Connell | 2020 | miR-137 | 9 | 2 | 1 | 8 |
| O'Connell | 2020 | miR-128-3p | 4 | 0 | 6 | 10 |
| miRNA panel | ||||||
| Schober | 2014 | miR-138,miR-744 | 8 | 0 | 0 | 7 |
| Schober | 2014 | miR-195,miR-324-5p | 8 | 0 | 0 | 7 |
| Di Pietro | 2018 | let-7i-5p,miR-142-3p,miR-107,miR-27b-3p,miR-135b-5p. | 16 | 9 | 6 | 1 |
| Redell | 2010 | miR-16,miR-92a | 5 | 1 | 2 | 6 |
| Redell | 2010 | miR-16,miR-765 | 6 | 0 | 1 | 8 |
| Redell | 2010 | miR-92a,miR-765 | 7 | 0 | 0 | 8 |
| Redell | 2010 | miR-92a,miR-765,miR-16, | 7 | 0 | 0 | 8 |
| Redell | 2010 | miR-16,miR-92a | 7 | 1 | 0 | 7 |
| Redell | 2010 | miR-16,miR-765 | 7 | 0 | 0 | 8 |
| Redell | 2010 | miR-92a,miR-765 | 6 | 2 | 1 | 6 |
| Redell | 2010 | miR-92a,miR-765,miR-16 | 7 | 0 | 0 | 8 |
| Redell | 2010 | miR-16,miR-92a | 9 | 2 | 2 | 6 |
| Redell | 2010 | miR-16,miR-92a | 8 | 6 | 3 | 2 |
| Ko | 2018 | miR-203b-5p,miR-203a-3p,miR-206,miR-185-5p. | 15 | 3 | 1 | 17 |
| O'Connell | 2020 | miR-124-3p, miR-219a-5p, miR-9-5p, miR-9-3p, miR-137, and miR-128-3p. | 9 | 0 | 1 | 10 |
TP: true positive; FP: false positive; FN: false negative; TN: true negative.
Diagnosis
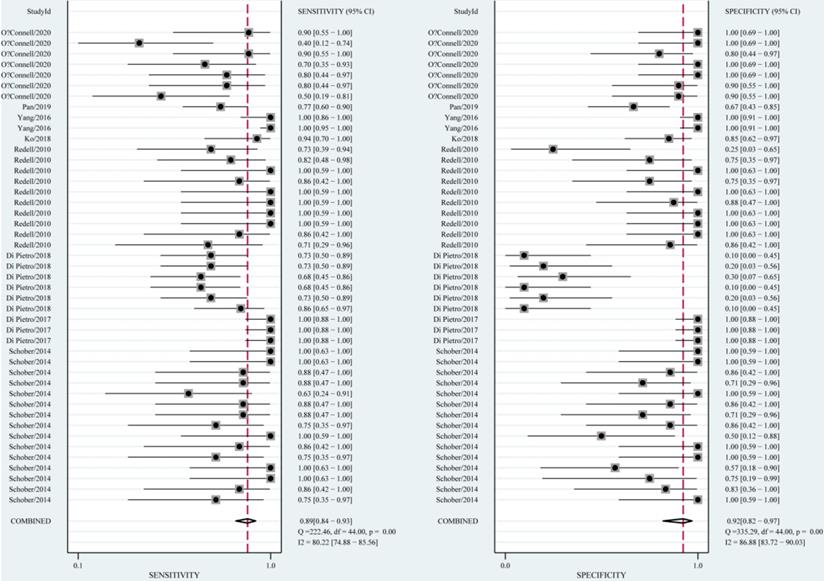
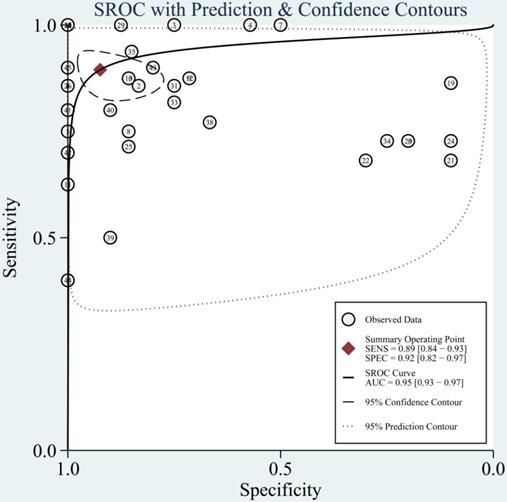
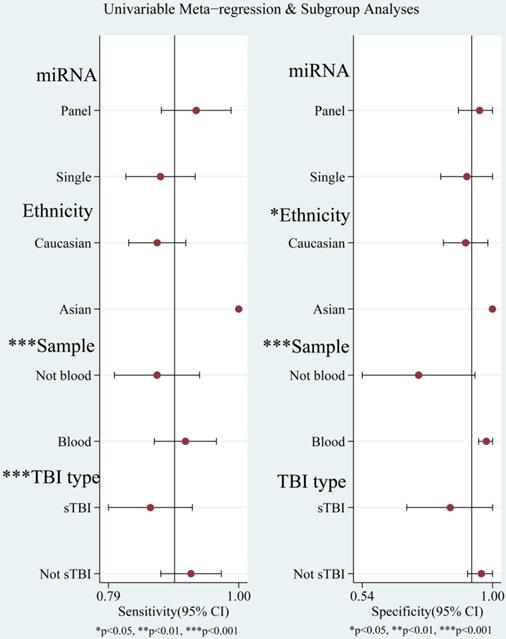
The diagnostic value of miRNAs for all TBI is shown in Fig. 2. Forest plots revealed a significant heterogeneity and we, therefore, used the mixed effect model in this meta-analysis. We also summarized sensitivity, specificity, and diagnostic accuracy of all miRNAs in TBI (Table 3). The sensitivity, specificity, PLR, NLR, and DOR of overall miRNA for diagnosis of TBI were 0.89 (95% CI: 0.84-0.93), 0.92 (95% CI: 0.82-0.97), 11.8 (95% CI: 4.7-29.6), 0.11 (95% CI: 0.07-0.18) and 103 (95% CI: 30-355). Diagnostic accuracy was evaluated by plotting the summary receiver operating characteristic (SROC) curve (Fig. 3). The diagnostic accuracy of overall miRNAs was outstanding since the area under the Curve (AUC) was 0.95 (95% CI: 0.93-0.97). We performed subgroup analyses according to ethnicity, detected sample, miRNA profiling and type of TBI in order to find the heterogeneity (Fig. 4). The diagnostic value of single miRNAs was as follows: sensitivity, 0.88; specificity, 0.91; PLR, 9.7; NLR, 0.13; DOR, 74; and AUC, 0.94 (Fig. S1, Table 3). However, miRNA panels have a higher overall diagnostic accuracy: sensitivity, 0.93; specificity, 0.95; PLR, 18.3; NLR, 0.08; DOR, 239; and AUC, 0.97 (Fig. S2, Table 3). The sensitivity, specificity, PLR, NLR, DOR and AUC of saliva, brain tissue, and blood were 0.73, 0.17, 0.90, 1.59, 1.0 and 0.40; 0.88, 0.87, 7.0, 0.13, 52 and 0.94; 0.94, 0.98, 46.0, 0.06, 756 and 0.99, respectively (Fig. S3-S5 & Table 3). This result suggested that miRNAs detected in blood have the highest overall diagnostic accuracy. In the severe TBI patients, the results were 0.91 for sensitivity, 0.92 for specificity, 11.9 for PLR, 0.09 for NLR, 129 for DOR, and 0.97 for AUC (Fig. S6, Table 3). The diagnostic value of miRNAs in Caucasians was as follows: sensitivity, 0.88; specificity, 0.91; PLR, 9.7; NLR, 0.14; DOR, 70; and AUC, 0.93 (Fig. S7, Table 3).
Forest plots for studies on overall miRNAs used in the diagnosis of traumatic brain injury (TBI) among 44 experiments included in the meta-analysis.
Summary of diagnostic value of miRNAs for diagnosis of TBI
| miRNAs | sensitivity | specificity | PLR | NLR | DOR | AUC |
|---|---|---|---|---|---|---|
| overall | 0.89 (0.84-0.93) | 0.92 (0.82-0.97) | 11.8 (4.7-29.6) | 0.11 (0.07-0.18) | 103 (30-355) | 0.95 (0.93-0.97) |
| Single miRNA | 0.88 (0.81-0.93) | 0.91 (0.76-0.97) | 9.7 (3.4-27.9) | 0.13 (0.08-0.23) | 74 (18-308) | 0.94 (0.92-0.96) |
| miRNA panels | 0.93 (0.84-0.97) | 0.95 (0.74-0.99) | 18.3(3.0-111.9) | 0.08 (0.03-0.19) | 239(21-2691) | 0.97 (0.95-0.98) |
| Blood | 0.94 (0.85-0.98) | 0.98 (0.90-1.00) | 46.0 (8.9-238.1) | 0.06 (0.02-0.16) | 756 (79-7255) | 0.99 (0.98-1.00) |
| Brain tissue | 0.88 (0.79-0.94) | 0.87 (0.75-0.94) | 7.0 (3.5-14.1) | 0.13 (0.08-0.24) | 52 (20-124) | 0.94 (0.91-0.96) |
| Salivary | 0.73 (0.65-0.80) | 0.17 (0.09-0.28) | 0.9 (0.8-1.0) | 1.59 (0.85-3.00) | 1.0 (0.0-1.0) | 0.40 (0.36-0.45) |
| Caucasians | 0.88 (0.82-0.92) | 0.91 (0.80-0.96) | 9.7 (4.0-23.2) | 0.14 (0.09-0.21) | 70 (22-222) | 0.93 (0.90-0.95) |
| sTBI | 0.91 (0.85-0.95) | 0.92 (0.84-0.96) | 11.9 (5.6-25.6) | 0.09 (0.05-0.17) | 129 (45-374) | 0.97 (0.95-0.98) |
PLR: positive likelihood ratio; NLR: negative likelihood ratio; DOR: diagnostic odds ratio; AUC: area under the curve; sTBI: severe traumatic brain injury.
The summary receiver operating characteristic (SROC) curves based on overall miRNAs in the meta-analysis.
Univariable meta-regression and subgroup analysis for sensitivity and specificity of miRNAs for diagnosis of traumatic brain injury (TBI).
Sensitivity analysis, meta-regression analysis and subgroup analysis
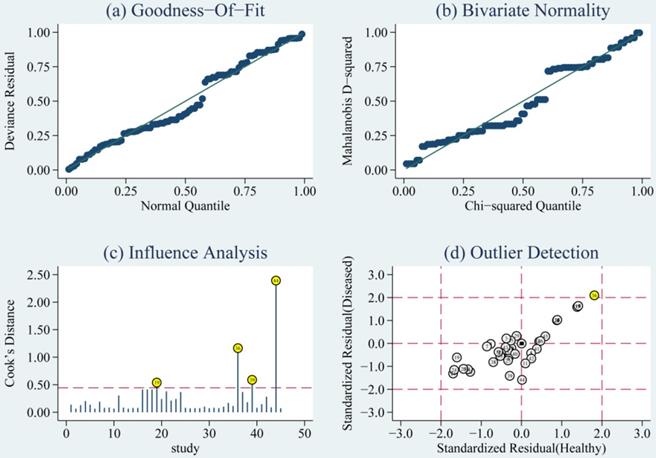
The goodness of fit and bivariate normality analyses revealed that the random effects bivariate model was best suited for sensitivity analysis (Fig. 5a & 5b). Influence analysis showed that studies of Di Pietro et al., Yang et al., and O'Connell et al. were the leading researches in weight (Fig. 5c). Outlier detection identified that no research would significantly affect the heterogeneity of our meta-analysis (Fig. 5d). Considering the bias of miRNAs, ethnicity, type of TBI and the detected sample, we conducted a meta-regression analysis and found that the detected sample may influence sensitivity and specificity. Results on subgroup analyses indicated that miRNA detected in blood exhibit the highest heterogeneity compared to brain tissue and saliva. We further conducted a subgroup analysis according to different types of TBI. In sTBI, no apparent heterogeneity was found because the I2 value was only 27.07% for sensitivity and 47.04% for specificity (Fig. S8). However, we did not do a subgroup analysis of mTBI due to the limitation of the number of mTBI studies.
Diagram of (a) Goodness-of-fit, (b) Bivariate normality, (c) Influence analysis, and (d) Outlier detection. Goodness-of-fit and Bivariate normality showed that random effects bivariate model is suitable. Influence analysis identified that studies of Pietro et al, Yang et al. and O'Connell et al were the most dominant studies in weight. Outlier detection implied that no research is the reason for heterogeneity.
Discussion
As potential biomarkers, miRNAs have been clinically tested for the diagnosis of diverse human diseases. In recent years, more researches have determined the diagnostic value of circulating miRNAs for TBI. However, the diagnostic performance of miRNAs in these studies remains controversial. For example, miR-135b acts as a biomarker in the diagnosis of sTBI, with specificity and sensitivity levels of 75% and 86% [11]. However, the sensitivity and specificity of mir-135b-5p from saliva samples were 73% and 20% [8]. Therefore, the reliability of miRNAs for the diagnosis of TBI remains to be confirmed, particularly with regards to sample type. We conducted this study to systematically assess the accuracy of circulating miRNAs in the diagnosis of TBI.
This meta-analysis involved seven articles, including 215 TBI patients and 152 controls. Our results implied that miRNAs had high sensitivity (0.89) and specificity (0.92) in TBI diagnosis. The pooled PLR was 11.8, suggesting that positive miRNA testing improved the diagnostic probability of TBI by 11.8-fold. Besides, the NLR was 0.11, indicating that negative miRNA testing increased the likelihood of TBI by 89%. A DOR of 1 indicates that miRNAs could not distinguish TBI from control, the DOR of 103 in our article indicated that miRNAs are distinguished biomarkers in the diagnosis of TBI.
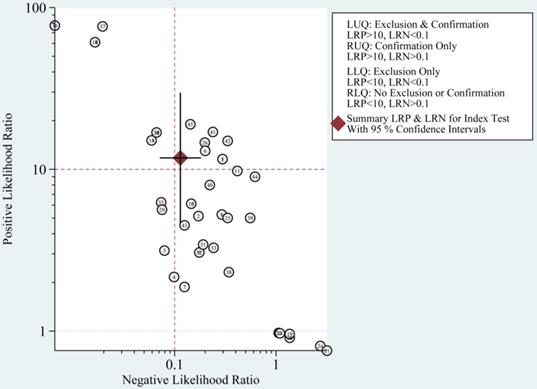
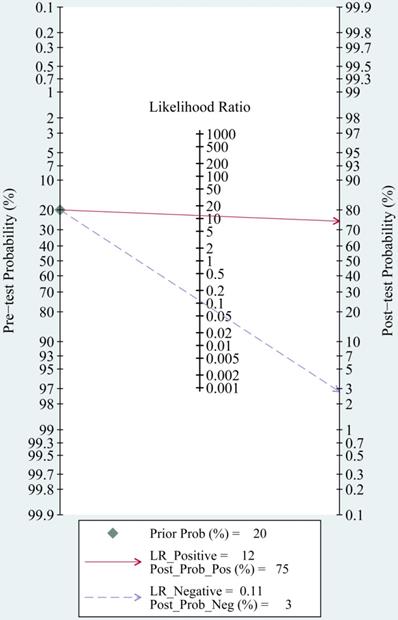
The most significant role of biomarkers is to help clinicians in clinical decision making. Through likelihood ratios and post-test probabilities, doctors can know the likelihood that a patient has TBI or not. Positive likelihood ratios and negative likelihood ratios were also summarized to assess diagnostic applicability of miRNAs (Fig. 6). NLR < 0.1 and PLR > 10 imply a high diagnostic accuracy [7]. The articles of Schober et al., Di Pietro et al., and Yang et al. revealed that some miRNAs had outstanding diagnostic accuracy, including single miRNA (miRNA-93, miRNA-425-5p, and miRNA-502) and a panel of miRNAs (miR-138 and miR-744; miR-195 and miR-324-5p). When we set the pretest probability at 20%, a positive likelihood ratio improves the post-test probability to 75%. When the clinician's accuracy rate for diagnosis of TBI based on the patient's symptoms and signs are 20%, the accuracy rate can be increased to 75% by combining miRNAs. When negative likelihood ratio was set at 0.11, the post-test probability for a negative test result is 3%, which means miRNAs can help clinicians reduce the false negative rate (Fig. 7).
Summary of positive likelihood ratio and negative likelihood ratio for diagnosis of traumatic brain injury (TBI).
Fagan nomogram of the miRNAs test for diagnosis of traumatic brain injury (TBI).
Notably, ideal biomarkers should be readily measurable in easily accessible samples such as blood or saliva. In our study, miRNAs in blood showed higher diagnostic accuracy, with a sensitivity of 0.94, a specificity of 0.98 and AUC of 0.99. We hypothesise the reason may be that brain specific miRNAs in exosomes can diffuse into the blood once TBI-induced disruption of blood brain barrier has occurred, which can maintain stability and replicability of miRNA from human blood [14]. However, miRNA detected in blood, including ten in plasma, twelve in serum, one in blood, showed the highest heterogeneity, we then did a subgroup analyses of plasma and found the I2 value in sensitivity and specificity only decreased 21.88% and 7.02% compared to other blood sample (Fig. S9 and Fig. S10). Thus, the heterogeneity could not be completely explained by this subgroup analyses. Compared to single miRNA, we demonstrated that multiple-miRNAs have higher diagnostic accuracy for TBI, which was consistent with the findings in other disorders [7, 15-17]. We also did a subgroup analysis of sTBI because we combined studies with different TBI severity. After excluding non-severe TBI studies, the I2 value in sensitivity and specificity dramatically decreased 53.15% and 39.84%. We thought that non-severe TBI studies could be part of the source of heterogeneity. Pooled sensitivity, pooled specificity and ROC revealed miRNAs have potential diagnostic value for sTBI. We failed to do a subgroup analysis of mTBI because only four of our included experiments reported on the diagnostic value of miRNAs in mTBI patients. We also intend to evaluate the clinical value of miRNAs expression in mTBI patients. Our results were, however, not conclusive because of the limited number of studies. We suggest that more future researchers could explore the clinical value of miRNAs in mTBI.
This meta-analysis has several limitations. Firstly, the results of subgroup analysis, such as blood sample and non-severe TBI, could only find a part of the source of heterogeneity in our study. Second, our meta-analysis had a small sample size. Third, the overall diagnostic accuracy may be exaggerated because studies with positive results have high possibility of publication. Finally, we included studies only written in English, which may bring some bias to our findings.
Conclusion
Our meta-analysis is the first to evaluate the clinical value of miRNAs expression in TBI patients. miRNAs have potential diagnostic value for TBI. Subgroup analysis demonstrated that miRNAs in blood could improve diagnostic accuracy. Compared to a single miRNA, panels of multiple miRNAs could more accurately identify TBI patients. However, we need include large-sizes researches in future to validate our results and confirm the clinical value of miRNAs in the diagnosis of TBI.
Abbreviations
miRNAs: MicroRNAs; AUC: area under the curve; CI: confidence interval; SROC: summary receiver operator characteristic; TP: true positive; FP: false positive; FN: false negative; TN: true negative; PLR: positive likelihood ratio; NLR: negative likelihood ratio; DOR: diagnostic odds ratio; TBI: traumatic brain injury; mTBI: mild traumatic brain injury; sTBI: severe traumatic brain injury; HRs: hazard ratios.
Supplementary Material
Supplementary figures.
Acknowledgements
This project was supported by The National Natural Science Foundation of China (No. 81971159). This fund supported data collection and literature download for this study.
Competing Interests
The authors have declared that no competing interest exists.
References
1. Maas AIR, Menon DK, Adelson PD, Andelic N, Bell MJ, Belli A. et al. Traumatic brain injury: integrated approaches to improve prevention, clinical care, and research. The Lancet Neurology. 2017;16:987-1048
2. Agoston DV, Shutes-David A, Peskind ER. Biofluid biomarkers of traumatic brain injury. Brain injury. 2017;31:1195-203
3. Pan YB, Sun ZL, Feng DF. The Role of MicroRNA in Traumatic Brain Injury. Neuroscience. 2017;367:189-99
4. Di Pietro V, Ragusa M, Davies D, Su Z, Hazeldine J, Lazzarino G. et al. MicroRNAs as Novel Biomarkers for the Diagnosis and Prognosis of Mild and Severe Traumatic Brain Injury. Journal of neurotrauma. 2017;34:1948-56
5. Redell JB, Moore AN, Ward NH 3rd, Hergenroeder GW, Dash PK. Human traumatic brain injury alters plasma microRNA levels. Journal of neurotrauma. 2010;27:2147-56
6. Qin X, Li L, Lv Q, Shu Q, Zhang Y, Wang Y. Expression profile of plasma microRNAs and their roles in diagnosis of mild to severe traumatic brain injury. The Journal of neuroscience: the official journal of the Society for Neuroscience. 2018;13:e0204051
7. Zhou Q, Liu J, Quan J, Liu W, Tan H, Li W. MicroRNAs as potential biomarkers for the diagnosis of glioma: A systematic review and meta-analysis. Cancer Sci. 2018;109:2651-9
8. Di Pietro V, Porto E, Ragusa M, Barbagallo C, Davies D, Forcione M. et al. Salivary MicroRNAs: Diagnostic Markers of Mild Traumatic Brain Injury in Contact-Sport. Frontiers in molecular neuroscience. 2018;11:290
9. Yang T, Song J, Bu X, Wang C, Wu J, Cai J. et al. Elevated serum miR-93, miR-191, and miR-499 are noninvasive biomarkers for the presence and progression of traumatic brain injury. Journal of Neurochemistry. 2016;137:122-9
10. Ko J, Hemphill M, Yang Z, Beard K, Sewell E, Shallcross J. et al. Multi-Dimensional Mapping of Brain-Derived Extracellular Vesicle MicroRNA Biomarker for Traumatic Brain Injury Diagnostics. Journal of neurotrauma. J Neurotrauma. 2019
11. Schober K, Ondruschka B, Dressler J, Abend M. Detection of hypoxia markers in the cerebellum after a traumatic frontal cortex injury: a human postmortem gene expression analysis. International journal of legal medicine. 2015;129:701-7
12. Pan P, Bai L, Hua X, Wang Y, Jiang X, Cheng X. et al. miR-155 Regulates claudin1 Expression in Humans With Intestinal Mucosa Dysfunction After Brain Injury. Transplant Proc. 2019;51:3474-80
13. O'Connell GC, Smothers CG, Winkelman C. Bioinformatic analysis of brain-specific miRNAs for identification of candidate traumatic brain injury blood biomarkers. Brain injury. 2020;34:965-74
14. Vickers KC, Palmisano BT, Shoucri BM, Shamburek RD, Remaley AT. MicroRNAs are transported in plasma and delivered to recipient cells by high-density lipoproteins. Nature cell biology. 2011;13:423-33
15. Gao Y, Zhao H, Lu Y, Li H, Yan G. MicroRNAs as potential diagnostic biomarkers in renal cell carcinoma. Tumour biology: the journal of the International Society for Oncodevelopmental Biology and Medicine. 2014;35:11041-50
16. Yang Y, Zhu R. Diagnostic value of circulating microRNAs for hepatocellular carcinoma. Molecular biology reports. 2014;41:6919-29
17. Zi Y, Yin Z, Xiao W, Liu X, Gao Z, Jiao L. et al. Circulating MicroRNA as Potential Source for Neurodegenerative Diseases Biomarkers. Molecular neurobiology. 2015;52:1494-503
Author contact
Corresponding author: Xiaofeng Yang, Department of Emergency and Trauma Center, The International Medical Center, The First Affiliated Hospital, College of Medicine, Zhejiang University, 79 Qingchun Road, Hangzhou 310003, Zhejiang Province, China. E-mail: zjcswkedu.cn.

 Global reach, higher impact
Global reach, higher impact