Impact Factor ISSN: 1449-1907
Int J Med Sci 2017; 14(9):798-803. doi:10.7150/ijms.19241 This issue Cite
Short Research Communication
High Efficiency Low Cost Fibroblast Nucleofection for GMP Compatible Cell-based Gene Therapy
1. Department of Orthopedics, Tongji Hospital, Tongji Medical College, Huazhong University of Science and Technology, Wuhan, China.
2. Department for Plastic Surgery and Hand Surgery; Klinikum rechts der Isar; Technical University Munich, Munich, Germany;
3. Technical University Munich, Faculty of Medicine, TUM Cells Interdisciplinary Center for Cellular Therapies, Munich, Germany;
4. Department of Plastic Surgery and Hand Surgery, University of Lübeck, Lübeck, Germany;
5. Klinik für Unfallchirurgie, Orthopädie und Plastische Chirurgie, Universitätsmedizin Göttingen, Göttingen, Germany.
Received 2017-1-17; Accepted 2017-4-23; Published 2017-7-19
Abstract
Background: Dermal fibroblast is a powerful tool for the study of ex vivo DNA delivery in development of both cell therapy and tissue engineering products. Using genetic modification, fibroblasts can be diversely adapted and made suitable for clinical gene therapy. In this study, we first compared several non-viral transfection methods including nucleofection in rat and human primary dermal fibroblast. In addition, the original protocol for nucleofection of primary mammalian fibroblasts was modified in order to achieve the highest possible transfection efficiency, as determined by flow cytometry analysis of the green fluorescent protein (GFP) expression.
Results: the results showed that transfection performance of Dulbecco's Modified Eagle Medium (DMEM) supplemented with 10% Fetal Calf Serum (FCS) yielded the best transfection efficiency with rat dermal fibroblasts and ITS (insulin, transferrin, and sodium selenite solution) was comparable to the standard nucleofection solution for human dermal fibroblasts.
Conclusion: Our results suggest a promising application of the modified nucleofection method for GMP compatible therapeutic translational medical research.
Keywords: Dermal fibroblast, nucleofection method, green fluorescent protein
Background
In the last decade, the gene therapy has opened new possibilities in the management of chronic wounds [1-3]. Divergent virus-based methods for manipulation of cells were effectively used in several non-clinical studies [4-6] including at least one reported clinical trial [6]. However, the possible adverse effects due to integration of the virus as well as the long-term persistence of the virus-coded transgene expression are factors which significantly limit the wider use of such applications [7]. Thus, non-viral gene delivery technologies deliver an attractive alternative approach in genetic modification of target cells, and importantly, show the efficacy in wound healing and tissue regeneration [1, 8]
Cultured dermal fibroblasts are used to support the tissue repair process in a variety of wound etiologies. Moreover, dermal fibroblasts are ideal candidates for large scale cell-based gene therapy since they are easy to isolate, robust and grow fast ex vivo [9, 10]. Nucleofection, an electroporation-based transfection method, has proved to be a very efficient method for genetic modification of many hard to transfect cell types [8, 11-13]. Several studies demonstrated that with nucleofection the greatest transfection efficiency was achieved compared to other commonly used non-viral methods for transfection of several hard-to-transfect cells [14-16]. In our study, we tested several different non-viral transfection methods in rat and human dermal fibroblasts and compared it with a commercial nucleofection method. Moreover, our aim was to further optimise the electroporation-based method taking into consideration its potential use in Good Manufacturing Practice (GMP) compatible large- scale fibroblasts-based gene therapy.
Methods
For rat dermal fibroblasts, rat skin samples were obtained from the back of Lewis inbred rats (weight 200-300 g, Charles River Laboratories, Germany) and cells were isolated as described before [17]. The study conforms the principles outlined in the Declaration of Helsinki and the Guiding Principles in the Care and Use of Animals and local animal protection regulations. Only the first 3 passages of the primary cells were used for experiments. The fibroblasts were cultivated in medium containing Dulbecco's Modified Eagle Medium (DMEM) + 10% Fetal Calf Serum (FCS) (further indicated as cell culture medium). Isolated fibroblasts were stained with phalloidin (Invitrogen, California, USA) and DAPI (4',6-diamidino-2-phenylindole; Invitrogen, California, USA) and the morphology was examined under the fluorescent microscope. The cells showed a typical spindle shape during the culture (Figure 1A upper panel: red fluorescence: Phalloidin; blue fluorescence: DAPI). Additionally, the cells were seeded on chamber slides for fibroblast characterization and stained with the antibody against beta subunit of prolyl-4-hydroxylase (P4Hβ: Acris, Herford, Germany). As shown in Figure 1A the cells were positive for this rat fibroblast marker (Figure 1A lower panel: green fluorescence: P4Hβ; red fluorescence: PI nuclear staining).
Analysis of transfection efficiency of rat dermal fibroblasts. Rat fibroblasts were isolated, shortly cultured (passage number did not exceed 3) and transfected with pmaxGFP plasmid. Transfection efficiency was analyzed by flow cytometry of GFP expression and was given as the percentage of GFP positive cells. A) Phenotypical characterization of rat dermal fibroblasts. The cells were evaluated with phalloidin/DAPI staining (upper panel) as well as by staining with rat fibroblast-specific antibody against beta subunit of prolyl-4-hydroxilase and propidium iodide (lower panel). B) Comparison of the transfection efficiencies of the four different non-viral transfection methods. Images of light and fluorescent microscopy are given in the upper panel and GFP transfected cells in the lower panel. C) Comparison of the transfection efficiencies of standard and modified nucleofection protocol (standard transfection solution was substituted with DMEM cell culture medium supplemented with 10% FCS). Images of light and fluorescent microscopy are given in the upper panel and GFP transfected cells in the lower panel. D) Time-course of the percentage of GFP positive fibroblasts transfected by using the modified nucleofection protocol. Images of fluorescent microscopy are given in the upper panel. Scale bar represents 100 μm in A upper panel, 50 μm in lower panel and 200 μm in others. The results are depicted as mean ± SD, t-test: *p<0.05, **p<0.01, ***p<0.001.
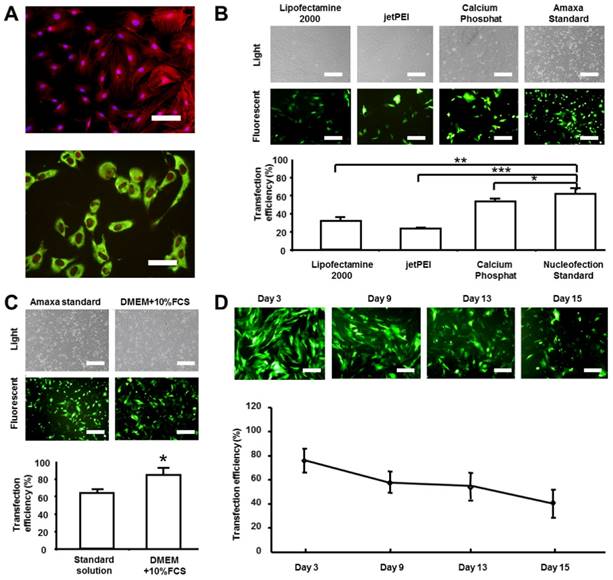
After cell isolation, four common non-viral transfection methods were used for the transfection of rat dermal fibroblasts: 1) Lipofectamine 2000 (Invitrogen, California, USA), 2) Jet PEI (Polyplus-transfection SA, Strasbourg, France), 3) Calcium Phosphate Transfection Kit (Invitrogen, California, USA) and 4) Transfection with the Nucleofector apparatus (later in the text referred as nucleofection) by using the Nucleofactor Kit for primary mammalian fibroblasts as described by the manufacturer (Lonza, Cologne, Germany). In addition, the modified nucleofection method was tested. Plasmid pmaxGFP (Lonza, Cologne, Germany) was used for all transfection experiments. Transfection efficiencies were monitored by GFP fluorescence using flow cytometry (Cytomation MoFlo® Flow Cytometer, Dako, Denmark). The transfection protocols were as follows:
- Lipofectamine 2000: 0.2 million cells were seeded one day before transfection in one well of a 24-well plate in 1 ml cell culture medium. The cells were transfected upon reaching the confluence of 80-90%. Medium was changed short time before the transfection. Two mixtures were prepared. One contained 4 µg GFP and 50 µl DMEM, and the other 2 µl Lipofectamine 2000 and 50 µl DMEM. They were incubated at RT (room temperature) for 5 min. Subsequently, both solutions were thoroughly mixed, followed by incubation at RT for 20 min. 100 µl of the complete solution was added into the well with cultured primary fibroblasts and incubated for 4 hours in the incubator under standard conditions (37°C, 5%CO2). After incubation time elapsed, the medium containing the transfection solution was discarded and the fresh cell culture medium added to the cells. The transfection efficiency was measured after 48 h.
- Jet PEI: 0.1 million cells were seeded one day before transfection in one well of a 24-well plate in 1 ml cell culture medium. The cells were transfected upon reaching the confluence of 80-90%. 1 µg pmaxGFP and 2 µl Jet PEI were resuspended in 100 µl of 150 mM NaCl and incubated for 15 min. The mixture was then added to the plated fibroblasts and incubated for 4 hours in the incubator under standard conditions. After incubation time elapsed, the medium containing the transfection solution was discarded and the fresh cell culture medium was added to the cells. The transfection efficiency was measured after 48 h.
- Calcium Phosphate Transfection Kit: 0.2 million cells were seeded one day before transfection in a 60 mm culture plate. The cells were transfected upon reaching the confluence of 80-90%. The medium was changed 4 hours before the transfection. Further, pmaxGFP plasmid (20 µg) was mixed with CaCl2 (resuspended in sterile distilled water) in a final volume of 150 µl and slowly added to 150 µl 2X HEPES buffer. The solution was then incubated at RT for 30 min, transferred to the cell culture plate and incubated overnight in the incubator under standard conditions. Medium was changed in the second day and transfection efficiency was detected after 2 days with FACS.
- Nucleofection standard method: For the transfection of rat primary fibroblasts the Basic Nucleofactor Kit for primary mammalian fibroblasts (Lonza, Cologne, Germany) was used. For the standard transfection method manufacturer's instructions were followed. Program U30 was applied.
We further investigated the influence of changes in the original nucleofection protocol on the transfection efficiency of dermal fibroblasts. In general, two factors are critical for successful nucleofection: cuvettes and transfection solution. The transfection solution is provided in the manufacturer's kit as ready-made solution. For the best transfection performance, the manufacturer recommends using cuvettes supplied with the kit. In order to test the performance of alternative cuvettes in the combination with Nucleofector apparatus and the Nucleofector Kit, electroporation cuvettes from Biorad (Munich, Germany) and Eppendorf (Hamburg, Germany) were compared with cuvettes supplied with the Lonza Nucleofector Kit. No significant differences were found in the transfection efficiency between used cuvettes as determined by flow cytometry analysis of GFP expression (data not shown). Furthermore, we tested an alternative transfection solution to the one supplied with the Nucleofector Kit. The conditions were as follows:
- Nucleofection modified method / DMEM +10% FCS: For the modified method, the standard transfection solution was substituted with DMEM+10% FCS. In addition, the Eppendorf cuvettes were used. Program U30 was applied.
We found that DMEM supplemented with 10% FCS showed a better transfection performance (85.35%±11.56%) than the standard Nucleofector Kit (68.34%±10.32%, Figure 1C P<0.05). The expression of GFP in the rat dermal fibroblasts genetically modified according to an adapted protocol for nucleofection was persistent at a high rate even 15 days after the transfection (Figure 1D).
The next step was to adapt the protocol for the nucleofection of human primary dermal fibroblasts. For human dermal fibroblasts nucleofection, the cells were isolated from split skin obtained from human subjects by using the procedure described below. The human skin biopsies were obtained from patients undergoing operation at the Department of Plastic and Hand Surgery of Lübeck University (after receiving signed informed control which had been approved by the Clinical Ethical Committee of the University of Lübeck). The fibroblasts were cultivated in medium containing DMEM+10%FCS. Isolated fibroblasts were stained with phalloidin (Invitrogen, California, USA) and DAPI (4',6-diamidino-2-phenylindole, California, Invitrogen) and the morphology was examined under the fluorescent microscope. The cells showed a typical spindle shape during the culture (Figure 2A upper panel: red fluorescence: Phalloidin; blue fluorescence: DAPI). Furthermore, the primary fibroblasts were characterized by applying the cytospin technique and stained with the anti-Thy-1 antibody (Dianova, Hamburg, Germany). The cells exhibited positive staining for the Thy-1 human fibroblast surface marker (Figure 2A lower panel: green fluorescence: Thy-1; red fluorescence: PI nuclear staining).
Analysis of transfection efficiency of human dermal fibroblasts. Human fibroblasts were isolated from split skin, cultured (passage number did not exceed 3) and transfected with pmaxGFP plasmid. Transfection efficiency was analyzed by flow cytometry of GFP expression and was given as the percentage of GFP positive cells. A) Phenotypical characterization of human dermal fibroblasts. The cells were evaluated with phalloidin/DAPI staining (upper panel) as well as by staining with human fibroblast-specific antibody against Thy-1 and propidium iodide (lower panel). (B) Transfection efficiency of three transfection solutions was compared by measuring the GFP expression (lower panel). Images of fluorescent microscopy are given in the upper panel. C) Time-course of the percentage of GFP positive fibroblasts transfected by using the modified nucleofection protocol and ITS liquid media supplement (right panel). Images of fluorescent microscopy are given in the left panel. Scale bar represents 100 μm in A upper panel, 50 μm in lower panel and 200 μm in others. The results are depicted as mean ± SD, t-test: ***p<0.001.

Three different transfection methods for the nucleofection of human dermal primary fibroblasts were evaluated:
- Nucleofection standard method: For the transfection of rat primary fibroblasts the Basic Nucleofactor Kit for primary mammalian fibroblasts (Lonza, Cologne, Germany) was used. For the standard transfection method manufacturer's instructions were followed. Program U24 was applied.
- Nucleofection modified method /DMEM +10% FCS: For the modified method, the standard transfection solution was substituted with DMEM+10% FCS. In addition, the Eppendorf cuvettes were used. Program U24 was applied.
- Nucleofection modified method /ITS liquid media supplement: For the modified method, the standard transfection solution was substituted with ITS liquid media supplement (Sigma Aldrich). In addition, the Eppendorf cuvettes were used. Program U24 was applied. The ITS liquid media supplement was chosen as serum alternative. It has defined composition which presents the advantage over the high complexity of animal sera, especially in the view of quality requirements of raw materials used for the production of cell-based and gene therapy medicinal products for human use.
Results
Our results demonstrate that nucleofection was the most suitable ex vivo transfection method for rat dermal fibroblasts, which is in the line with data published by other groups [12, 15]. As shown in Figure 1B, the transfection efficiency was the highest with standard nucleofection method (62.07%±9.49%) compare to Lipofectamine 2000 (32.22%±8.58% P<0.001), Jet PEI (23.47%±0.49% P<0.01) and Calcium Phosphate (53.87%±3.61% P<0.05) The results from human dermal fibroblasts transfection showed that transfection performance of DMEM supplemented with 10% FCS (57.88%±3.45%) was less efficient than ITS solution method (79.21%±1.62%, P<0.001, Figure 2B). However, the transfection efficiency with the ITS medium was comparable with the standard method (83.88%±9.67%, P>0.05). Finally, we evaluated the time-course of GFP expression in human primary fibroblasts transfected with pmaxGFP by using the ITS-based modified nucleofection method. At day 15, more than 40% GFP positive cells could still be detected (Figure 2C). At day 24, there are still more than 20% positive cells (data not shown).
Discussion
This study demonstrates a high efficiency of nucleofection technology as a useful tool for gene transfer of rat and human dermal fibroblasts. To our knowledge, the application of the modified nucleofection method described here yielded the highest transfection efficiency compared to other similar studies [15, 18, 19]. Thus, this optimized nucleofection technology for ex vivo gene delivery has a promising potential for clinical translation, in particular in skin-related health care [8, 17]. Adding growth factors in the form of plasmid DNA to the dermal fibroblasts could greatly improve wound repair process [20]. Moreover, from the perspective of drug development, the results presented here have notable impact on several safety and efficacy issues. Firstly, a high transfection rate of the cells ensures high expression of the therapeutic protein in transient but sustained manner. Local expression of the transgene will persist only as long as it is needed to promote wound healing [21]. Achieving the high level of therapeutic protein expression reduces the need for application of large number of genetically modified cells [22, 23]. Not only that the comparable efficacy could be obtained with less cells but also the higher level of safety. Secondly, the comparability of methods developed by using animal models and ex vivo systems and those intended for therapeutic applications in humans are of considerable importance from both safety and efficacy perspective [24]. In our institute, we have a Good Manufacturing Practice (GMP) facility for manipulating human cells in vitro and for biochemical manufacturing those cells. Our results confirmed the method transferability and we are currently working on the clinical transfer of such method. Finally, due to alternative components (nucleofection medium and cuvettes), the described method for transfection of dermal fibroblasts could significantly reduce the costs of manufacturing and is suitable for upscaling to clinical grade cell production.
Conclusions
In summary, the presented results suggest a promising application of the modified nucleofection method in therapeutic translational medical research.
Abbreviations
GMP: Good Manufacturing Practice; DMEM: Dulbecco's Modified Eagle Medium; FCS: Fetal Calf Serum; GFP: Green Fluorescent Protein; DAPI: 4',6-diamidino-2-phenylindole; ITS: insulin, transferrin, and sodium selenite solution.
Acknowledgements
This work was supported by grants from Innovations fund Schleswig-Holstein and University Hospital rechts der Isar, Technische Universität München to H-G Machens. Z. ZHANG was supported by a scholarship from the China Scholarship Council, a clinic research grant from Technische Universität München to Z. ZHANG (KKF. No. 8744556) and a grant to Z. Zhang from National Natural Science Foundation of China (Grant No. 81401538). The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript. Dr. Ziyang Zhang would thank Frau Dr. med. Zhang for helpful personal advices. The authors have declared that no competing interests exist.
Ethics approval
All procedures performed in this study involving human participants and animals were in accordance with the ethical standards of research committee of Luebeck Univeristy, Technical University of Munich and local research committees. All procedures performed in this study involving animals are in accordance with guidelines for the care and use of animals of research committee of Luebeck Univeristy, Technical University of Munich and local research committees.
Author contributions
Conceived and designed the experiments: Z. ZHANG, A. SLOBODIANSKI, H-G. MACHENS. Performed the experiments: Z. ZHANG, A. ARNOLD, J. NEHLSEN, T. PERISIC. Analyzed the data: Z. ZHANG, A. ARNOLD, J. NEHLSEN, T. PERISIC. Contributed reagents/materials/analysis tools: U. HOPFNER. Wrote the paper: Z. ZHANG, A. SLOBODIANSKI, T. PERISIC, A. F. SCHILLING.
Competing Interests
The authors have declared that no competing interests exist.
References
1. Branski LK, Pereira CT, Herndon DN, Jeschke MG. Gene therapy in wound healing: present status and future directions. Gene therapy. 2007;14:1-10
2. Eming SA, Krieg T, Davidson JM. Gene therapy and wound healing. Clinics in dermatology. 2007;25:79-92
3. Lohmeyer JA, Liu F, Kruger S, Lindenmaier W, Siemers F, Machens HG. Use of gene-modified keratinocytes and fibroblasts to enhance regeneration in a full skin defect. Langenbeck's archives of surgery. 2011;396:543-50
4. Deodato B, Arsic N, Zentilin L, Galeano M, Santoro D, Torre V. et al. Recombinant AAV vector encoding human VEGF165 enhances wound healing. Gene therapy. 2002;9:777-85
5. Liechty KW, Nesbit M, Herlyn M, Radu A, Adzick NS, Crombleholme TM. Adenoviral-mediated overexpression of platelet-derived growth factor-B corrects ischemic impaired wound healing. The Journal of investigative dermatology. 1999;113:375-83
6. Mulder G, Tallis AJ, Marshall VT, Mozingo D, Phillips L, Pierce GF. et al. Treatment of nonhealing diabetic foot ulcers with a platelet-derived growth factor gene-activated matrix (GAM501): results of a phase 1/2 trial. Wound repair and regeneration: official publication of the Wound Healing Society [and] the European Tissue Repair Society. 2009;17:772-9
7. Ortolano S, Spuch C, Navarro C. Present and future of adeno associated virus based gene therapy approaches. Recent patents on endocrine, metabolic & immune drug discovery. 2012;6:47-66
8. Zhang Z, Slobodianski A, Ito WD, Arnold A, Nehlsen J, Weng S. et al. Enhanced collateral growth by double transplantation of gene-nucleofected fibroblasts in ischemic hindlimb of rats. PloS one. 2011;6:e19192
9. Krueger GG. Fibroblasts and dermal gene therapy: a minireview. Human gene therapy. 2000;11:2289-96
10. Yan WF, Murrell DF. Fibroblast-based cell therapy strategy for recessive dystrophic epidermolysis bullosa. Dermatologic clinics. 2010;28:367-70 xii
11. Kim JH, Shin KH, Li TZ, Suh H. Potential of nucleofected human MSCs for insulin secretion. Journal of tissue engineering and regenerative medicine. 2011;5:761-9
12. Liu J, Gaj T, Yang Y, Wang N, Shui S, Kim S. et al. Efficient delivery of nuclease proteins for genome editing in human stem cells and primary cells. Nature protocols. 2015;10:1842-59
13. Steinbrunn T, Chatterjee M, Bargou RC, Stuhmer T. Efficient transient transfection of human multiple myeloma cells by electroporation-an appraisal. PloS one. 2014;9:e97443
14. Badakov R, Jazwinska A. Efficient transfection of primary zebrafish fibroblasts by nucleofection. Cytotechnology. 2006;51:105-10
15. Dickens S, Van den Berge S, Hendrickx B, Verdonck K, Luttun A, Vranckx JJ. Nonviral transfection strategies for keratinocytes, fibroblasts, and endothelial progenitor cells for ex vivo gene transfer to skin wounds. Tissue engineering Part C, Methods. 2010;16:1601-8
16. Nakayama A, Sato M, Shinohara M, Matsubara S, Yokomine T, Akasaka E. et al. Efficient transfection of primarily cultured porcine embryonic fibroblasts using the Amaxa Nucleofection system. Cloning and stem cells. 2007;9:523-34
17. Spanholtz TA, Theodorou P, Holzbach T, Wutzler S, Giunta RE, Machens HG. Vascular endothelial growth factor (VEGF165) plus basic fibroblast growth factor (bFGF) producing cells induce a mature and stable vascular network-a future therapy for ischemically challenged tissue. The Journal of surgical research. 2011;171:329-38
18. Mueller CK, Thorwarth MW, Schultze-Mosgau S. Angiogenic gene-modified fibroblasts for induction of localized angiogenesis. The Journal of surgical research. 2010;160:340-8
19. Stroh T, Erben U, Kuhl AA, Zeitz M, Siegmund B. Combined pulse electroporation-a novel strategy for highly efficient transfection of human and mouse cells. PloS one. 2010;5:e9488
20. Basu G, Downey H, Guo S, Israel A, Asmar A, Hargrave B. et al. Prevention of distal flap necrosis in a rat random skin flap model by gene electro transfer delivering VEGF(165) plasmid. The journal of gene medicine. 2014;16:55-65
21. Galiano RD, Tepper OM, Pelo CR, Bhatt KA, Callaghan M, Bastidas N. et al. Topical vascular endothelial growth factor accelerates diabetic wound healing through increased angiogenesis and by mobilizing and recruiting bone marrow-derived cells. The American journal of pathology. 2004;164:1935-47
22. Machens HG, Salehi J, Weich H, Munch S, Siemers F, Krapohl BD. et al. Angiogenic effects of injected VEGF165 and sVEGFR-1 (sFLT-1) in a rat flap model. The Journal of surgical research. 2003;111:136-42
23. Spanholtz T, Maichle A, Niedworok C, Stoeckelhuber BM, Kruger S, Wedel T. et al. Timing and targeting of cell-based VEGF165 gene expression in ischemic tissue. The Journal of surgical research. 2009;151:153-62
24. Yufit T, Carson P, Falanga V. Topical delivery of cultured stem cells to human non-healing wounds: GMP facility development in an academic setting and FDA requirements for an IND and human testing. Current drug delivery. 2014;11:572-81
Author contact
* Equal contributions
Corresponding author: Ziyang Zhang, M.D. Ph.D., Current address: Department of Orthopedics, Tongji Hospital, Tongji Medical College, Huazhong University of Science and Technology, 1095 Jiefang Avenue, 430030, Wuhan, China. Phone: (+086) 27-83665318; Fax: (+086) 27-83665338; E-Mail: zhangziyang776com

 Global reach, higher impact
Global reach, higher impact