Impact Factor ISSN: 1449-1907
Int J Med Sci 2015; 12(12):946-951. doi:10.7150/ijms.13184 This issue Cite
Research Paper
Visceral Fat Area Determined Using Bioimpedance Analysis Is Associated with Hearing Loss
1. Division of Nephrology, Department of Internal Medicine, Yeungnam University Hospital, Daegu, Republic of Korea;
2. Department of Otorhinolaryngology-Head and Neck Surgery, School of Medicine, Kyungpook National University Hospital, Daegu, Republic of Korea.
*These authors contributed equally to this work.
Received 2015-7-8; Accepted 2015-10-11; Published 2015-11-4
Abstract
Background: Visceral fat area (VFA) using bioimpedance analysis (BIA) as a simple analyzer can be used to assess VFA, which may be associated with HL. The aim of the present study was to evaluate the clinical relevance and usefulness of VFA using BIA as a predictor of HL.
Patients and Methods: In total, 18,415 patients were recruited into our study. VFAs were measured using multi-frequency BIA. VFAs were normalized by body mass index (BMI). Participants were divided into 3 tertiles based on their VFA/BMI for both sexes. For both ears of each participant, the low-frequency (Low-Freq), mid-frequency (Mid-Freq), and high-frequency (High-Freq) values were obtained calculating the pure tone averages at 0.5 and 1 kHz, 2 and 3 kHz, and 4 and 6 kHz, respectively. The average hearing threshold (AHT) was calculated as the pure tone average at the 4 frequencies (i.e., 0.5, 1, 2, and 3 kHz). HL was defined as AHT >40 dB.
Results: The VFA/BMI had the greatest AUROC among VFA, BMI, and VFA/BMI in both sexes in this study. In both univariate and multivariate analyses, VFA/BMI tertiles were associated with all 4 hearing thresholds (i.e., Low-Freq, Mid-Freq, High-Freq, and AHT). The 4 hearing thresholds were positively correlated with VFA/BMI as a continuous variable. The odds ratio for HL increased as the VFA/BMI tertile increased.
Conclusion: VFA/BMI was associated with hearing impairment in the Asian population. The participants with high VFA/BMI should be closely monitored for hearing impairment.
Keywords: Visceral fat area, body mass index, hearing threshold, bioimpedance analysis.
Introduction
Hearing loss (HL) is one of the most common public health problems. Obesity is a well-known risk factor for HL in the general population [1-3]. Obesity is associated with the development of cardiovascular disease, including cochlear vasculopathies. Body mass index (BMI) has been known as a classic obesity indicator. However, BMI cannot be used to differentiate fat from free-fat mass. The reliability of BMI as a representation of fat mass has been questioned [4]. During previous studies, conflicting results regarding the association between hearing impairment and BMI have been observed [3,5,6]. Fat, a constituent of body mass, is a biologically active tissue associated with the development of cardiometabolic complications. There are significant clinical and prognostic differences between subcutaneous and visceral fat tissues. Visceral fat tissue is more biologically active than subcutaneous fat tissue [7]. Kim et al. showed an association between visceral fat are (VFA) using computed tomography and hearing thresholds [8].
Computed tomography and magnetic resonance imaging at the umbilical level have been known as the gold standard for assessing visceral fat accumulation [9]. However, these methods involve x-ray irradiation or require an expensive equipment and relatively long imaging time. Multi-frequency bioimpedance analysis (BIA) is a simple method for assessing body compositions, including VFA. BIA uses electrical current resistance and impedance to measure body compositions. In our previous study, we showed that VFA measured using BIA was significantly correlated with that acquired by the computed tomography [10]. Therefore, VFA using BIA as a simple analyzer can be used to assess VFA, which may be associated with HL. The aim of the present study was to evaluate the clinical relevance and usefulness of VFA using BIA as a predictor of HL.
Methods
Study population
We identified 20,726 adults (≥40 years old) who underwent voluntary routine health checkups at Yeungnam University Hospital between June 2008 and April 2014. Data acquired during the first visit were used when patients underwent multiple examinations. Among these patients, 2,311 were excluded due to a lack of laboratory findings or BIA data. In total, 18,415 patients were recruited into our study. This study was approved by the Institutional Review Board of Yeungnam University Hospital. The board waived the need for informed consent because the patients' records and information were anonymized and de-identified prior to analysis.
Study variables
Clinical and laboratory data collected from the participants during the health examination included the following variables: age, sex, body mass index (BMI; kg/m2), VFA (cm2), systolic blood pressure (SBP; mmHg), diastolic blood pressure (DBP; mmHg), serum creatinine level (mg/dL), estimated glomerular filtration rate (eGFR; mL/min/1.73m2), fasting blood glucose level (FBG; mg/dL), total cholesterol level (mg/dL), triglyceride level (TG; mg/dL), high-density lipoprotein (HDL) cholesterol level (mg/dL), and hearing thresholds. BMI was calculated as body weight divided by height squared [body weight (kg)/height (m2)]. VFAs were measured using multi-frequency BIA (In-Body 720; Biospace, Seoul, Korea). VFAs were normalized by BMI. Participants were divided into 3 tertiles based on their VFA/BMI for both sexes: low, middle, and high tertiles. Serum creatinine levels were measured using a Hitachi Automatic Analyzer (alkaline picrate, Jaffé kinetic reaction). The eGFR was calculated using the Chronic Kidney Disease Epidemiology Collaboration equation [11]. Chronic kidney disease was defined as an eGFR <60 mL/min/1.73 m2. Diabetes mellitus (DM) was defined as a self-reported history of DM diagnosis or a fasting glucose level ≥126 mg/dL. Hypertension was defined as an SBP ≥140 mmHg, DBP ≥90 mmHg, a self-reported history of hypertension or use of anti-hypertensive drugs.
The hearing thresholds were measured using an automatic audiometer at 0.5, 1, 2, 3, 4, and 6 kHz. None of the participants had been receiving medication associated with ototoxicity or had ear disease (i.e., chronic otitis media). For both ears of each participant, the low-frequency (Low-Freq), mid-frequency (Mid-Freq), and high-frequency (High-Freq) values were obtained calculating the pure tone averages at 0.5 and 1 kHz, 2 and 3 kHz, and 4 and 6 kHz, respectively. In the present study, the average hearing threshold (AHT) was calculated as the pure tone average at the 4 frequencies (i.e., 0.5, 1, 2, and 3 kHz). HL was defined as AHT >40 dB.
Statistical analyses
Data were analyzed using SPSS version 21 (SPSS, Chicago, IL, USA). Categorical variables were expressed as numbers and percentages. Continuous variables were expressed as mean ± standard deviation or standard error. The Pearson's χ2 test or Fisher's exact test was used to analyze categorical variables. For continuous variables, means were compared using one-way analysis of variance. Linear regression analysis was performed to assess the independent predictors of AHT. Logistic regression analyses were used to estimate odds ratios and 95% confidence intervals (CI) to determine the relationship between VFA/BMI tertiles and HL. Multivariate analysis was adjusted for age, DM, HTN, and CKD. Variance inflation factor (VIF) was used to identify multicollinearity for the multivariate linear regression model. VIF greater than 10 was not accepted.
Discrimination, the ability of the model to differentiate between participants with and without HL, was examined using the area under the receiver operating characteristic curve (AUROC). AUROC analysis was also performed to calculate cutoff values, sensitivity, and specificity. The cutoff risk point was defined as the highest sensitivity (100 - specificity) value in the AUROC. The statistical significance between AUROC values was calculated by the DeLong method. The AUROC was calculated using MedCalc version 11.6.1.0 (Medcalc, Mariakerke, Belgium). The level of statistical significance was set at P <0.05.
Results
Clinical characteristics of the participants
There were 3,410, 3,406, and 3,401 men and 2,733, 2,735, and 2,730 women in the low, middle, and high VFA/BMI tertiles, respectively. The mean VFA/BMI in the low, middle, and high tertiles were 3.37 ± 0.48 (0.23-3.89), 4.19 ± 0.16 (3.90-4.46), and 4.95 ± 0.41 (4.47-7.02), respectively, in men and 3.12 ± 0.38 (1.20-3.59), 3.92 ± 0.19 (3.60-4.24), and 4.78 ± 0.41 (4.25-6.64), respectively, in women (Table 1). In both sexes, the portions of participants with DM, hypertension, and chronic kidney disease increased with increasing VFA/BMI tertiles. Furthermore, age, BMI, and TG levels increased with increasing VFA/BMI tertiles in both sexes.
Clinical characteristics of the participants according to visceral fat are/body mass index tertiles.
| Men | Women | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| Low | Middle | High | P | Low | Middle | High | P | ||
| Age (years) | 48.6 ± 6.7 | 52.5 ± 6.7 | 61.2 ± 9.3 | <0.001 | 46.7 ± 5.3 | 53.2 ± 6.3 | 64.3 ± 8.3 | <0.001 | |
| BMI (kg/m2) | 23.6 ± 2.6 | 25.0 ± 2.6 | 25.2 ± 3.0 | <0.001 | 21.7 ± 2.2 | 23.6 ± 2.6 | 24.9 ± 3.2 | <0.001 | |
| VFA (cm2) | 79.7 ± 15.7 | 104.7 ± 11.8 | 124.6 ± 17.3 | <0.001 | 68.3 ± 12.7 | 92.8 ± 11.6 | 119.0 ± 18.6 | <0.001 | |
| Diabetes mellitus | 215 (6.3%) | 372 (10.9%) | 604 (17.8%) | <0.001 | 53 (1.9%) | 156 (5.7%) | 405 (14.8%) | <0.001 | |
| Hypertension | 499 (14.6%) | 738 (21.7%) | 1042 (30.6%) | <0.001 | 158 (5.8%) | 357 (13.1%) | 758 (27.8%) | <0.001 | |
| eGFR (mL/min/1.73m2) | 67.8 ± 12.0 | 66.8 ± 11.8 | 63.7 ± 15.1 | <0.001 | 98.7 ± 25.3 | 95.9 ± 25.7 | 89.0 ± 30.5 | <0.001 | |
| CKD (%) | 821 (24.0%) | 984 (28.9%) | 1335 (39.4%) | <0.001 | 20 (0.7%) | 36 (1.3%) | 104 (3.8%) | <0.001 | |
| FBG (mg/dL) | 93.6 ± 21.4 | 97.5 ± 23.7 | 101.4 ± 28.1 | <0.001 | 88.3 ± 15.0 | 91.6 ± 16.6 | 98.6 ± 24.9 | <0.001 | |
| TC (mg/dL) | 197.8 ± 35.1 | 203.6 ± 34.9 | 202.8 ± 40.1 | <0.001 | 188.3 ± 32.4 | 204.2 ± 35.3 | 211.7 ± 38.4 | <0.001 | |
| TG (mg/dL) | 134.4 ± 99.7 | 161.7 ± 112.6 | 156.8 ± 118.4 | <0.001 | 84.2 ± 50.6 | 108.8 ± 71.0 | 129.0 ± 96.9 | <0.001 | |
| HDL (mg/dL) | 54.3 ± 14.2 | 51.3 ± 13.1 | 51.2 ± 13.4 | <0.001 | 64.3 ± 15.4 | 60.1 ± 14.6 | 57.8 ± 14.8 | <0.001 | |
The data are expressed as numbers (percentages) for categorical variables and means ± standard deviations for continuous variables. The P values were tested using one-way analysis of variance for continuous variables and Pearson χ2 test or Fisher exact test for categorical variables.
Abbreviations: BMI, body mass index; VFA, Visceral fat area; eGFR, estimated glomerular filtration rate; CKD, chronic kidney disease; FBG, fasting blood glucose; TC, total cholesterol; TG, triglyceride; HDL, high-density lipoprotein.
Association between VFA/BMI and hearing loss
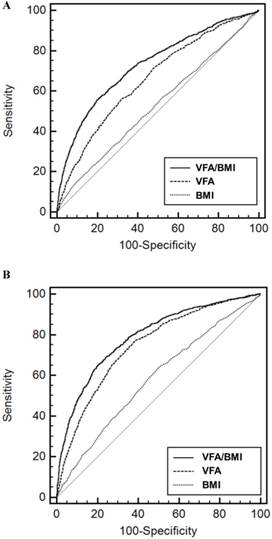
The AUROCs of VFA/BMI, VFA, and BMI for HL were 0.731 (95% CI, 0.722-0.739), 0.661 (95% CI, 0.652-0.670), and 0.530 (95% CI, 0.520-0.540) in men and 0.791 (95% CI, 0.782-0.799), 0.747 (95% CI, 0.737-0.756), and 0.583 (95% CI, 0.572-0.594) in women, respectively (Figure 1). The VFA/BMI had the highest AUROC and was the best predictor of HL (P <0.001). The sensitivity and specificity for predicting HL were 56.5% and 79.3% in men and 69.0% and 75.9% in women, respectively. According to univariate and multivariate analyses, the mean of the 4 hearing thresholds in both sexes increased as the VFA/BMI tertiles increased (Figure 2). In addition, a positive association of VFA/BMI with AHT was observed in the univariate and multivariate linear regression analyses (Table 2). The correlation coefficients between age and VFA/BMI were 0.594 in men and 0.796 in women (P < 0.001 in both sexes). However, VIFs for all explanatory variables, including age and VFA/BMI, were lesser than 10. These results show that age was positively associated with VFA/BMI, but multivariate models waived the risk of collinearity in both sexes.
Receiver operating characteristic curves of variable indices for the prediction of hearing loss in men (A) and women (B). Abbreviations: VFA, visceral fat area; BMI, body mass index.
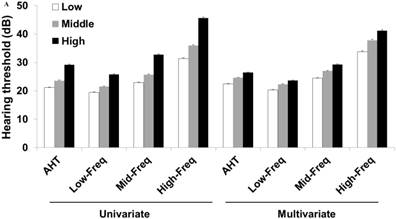
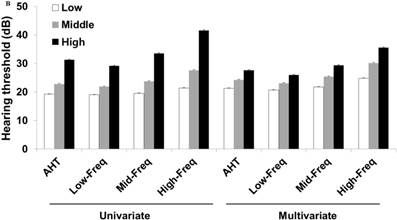
Hearing thresholds according to visceral fat area/body mass index tertiles in men (A) and women (B). The multivariate analysis was adjusted for age, diabetes mellitus, hypertension, and chronic kidney disease (P < 0.05 for trends in all analyses). The data are expressed as mean and standard error values. Abbreviations: AHT, average hearing threshold; Low-Freq, low frequency; Mid-Freq, middle frequency; High-Freq, high frequency.
Linear regression analyses of average hearing threshold according to visceral fat area/body mass index.
| Independent variables | Univariate | Multivariate | |||||
|---|---|---|---|---|---|---|---|
| Standardized β ± SE | P-value* | Standardized β ± SE | P-value* | VIF | |||
| Men | |||||||
| Age | 0.508 ± 0.010 | <0.001 | 0.490 ± 0.013 | <0.001 | 1.682 | ||
| Diabetes mellitus | 0.084 ± 0.339 | <0.001 | -0.003 ± 0.297 | 0.760 | 1.035 | ||
| Hypertension | 0.070 ± 0.262 | <0.001 | 0.000 ± 0.228 | 0.974 | 1.031 | ||
| eGFR | -0.081 ± 0.008 | <0.001 | 0.070 ± 0.007 | <0.001 | 1.098 | ||
| VFA/BMI | 0.347 ± 0.137 | <0.001 | 0.066 ± 0.157 | <0.001 | 1.576 | ||
| Women | |||||||
| Age | 0.569 ± 0.011 | <0.001 | 0.517 ± 0.018 | <0.001 | 2.941 | ||
| Diabetes mellitus | 0.159 ± 0.488 | <0.001 | 0.021 ± 0.419 | 0.026 | 1.070 | ||
| Hypertension | 0.185 ± 0.353 | <0.001 | 0.025 ± 0.308 | 0.008 | 1.095 | ||
| eGFR | -0.118 ± 0.005 | <0.001 | 0.033 ± 0.004 | 0.001 | 1.087 | ||
| VFA/BMI | 0.479 ± 0.151 | <0.001 | 0.062 ± 0.234 | <0.001 | 2.761 | ||
*The dependent variable was average hearing threshold, and the multivariate analysis was adjusted for age, diabetes mellitus, hypertension, and eGFR.
Abbreviations: SE, standard error; VIF, variance inflation factor; eGFR, estimated glomerular filtration rate; VFA, visceral fat area; BMI, body mass index.
Men in the middle and high VFA/BMI tertiles had a 1.760 (95% CI, 1.403-2.209; P <0.001) and 5.746 (95% CI, 4.705-7.016; P <0.001) fold increased risk of HL compared to participants in the low VFA/BMI tertile according to the univariate logistic regression analysis (Table 3). Men in the middle and high VFA/BMI tertiles had a 1.630 (95% CI, 1.294-2.053; P <0.001) and 2.341 (95% CI, 1.869-2.933; P <0.001) fold increased risk of HL compared to those in the low VFA/BMI tertile according to the multivariate analysis. Women in the middle and high VFA/BMI tertiles had a 2.663 (95% CI, 1.976-3.590; P <0.001) and 11.976 (95% CI, 9.162-15.653; P <0.001) fold increased risk of HL compared to those in the low VFA/BMI tertile according to the univariate logistic regression analysis. Women in the middle and high VFA/BMI tertiles had a 2.317 (95% CI, 1.715-3.130; P <0.001) and 4.425 (95% CI, 3.277-5.977; P <0.001) fold increased risk of HL compared to those in the low VFA/BMI tertile according to multivariate analysis.
Univariate and multivariate odds ratios for the hearing loss according to visceral fat area/body mass index tertiles.
| Men | Women | ||||||
|---|---|---|---|---|---|---|---|
| Low tertile | Middle tertile | High tertile | Low tertile | Middle tertile | High tertile | ||
| Univariate | |||||||
| Odds ratio | 1.0 | 1.760 | 5.746 | 1.0 | 2.663 | 11.976 | |
| 95% CI | ref | 1.403-2.209 | 4.705-7.016 | ref | 1.976-3.590 | 9.162-15.653 | |
| P-value | <0.001 | <0.001 | <0.001 | <0.001 | |||
| Multivariate | |||||||
| Odds ratio | 1.0 | 1.630 | 2.341 | 1.0 | 2.317 | 4.425 | |
| 95% CI | ref | 1.294-2.053 | 1.869-2.933 | ref | 1.715-3.130 | 3.277-5.977 | |
| P-value | <0.001 | <0.001 | <0.001 | <0.001 |
The multivariate analysis was adjusted for age, diabetes mellitus, hypertension, and chronic kidney disease.
Abbreviations: CI, confidence interval.
Discussion
The VFA/BMI had the greatest AUROC among VFA, BMI, and VFA/BMI in both sexes in this study. In both univariate and multivariate analyses, VFA/BMI tertiles were associated with all 4 hearing thresholds (i.e., Low-Freq, Mid-Freq, High-Freq, and AHT). The 4 hearing thresholds were positively correlated with VFA/BMI as a continuous variable. The odds ratio for HL increased as the VFA/BMI tertile increased. According to these results, VFA/BMI was associated with HL in the Asian population.
Obesity is an important risk factor for the development of hearing impairment in the general population [1-3]. Adiponectin is inversely associated with obesity. VFA induces insulin resistance, overproduction of offensive adipokines (i.e., plasminogen activator inhibitor-1), and underproduction of defensive adipokines (i.e., adiponectin) [12]. The change in adiponectin level with obesity plays a key role in the development of cardiometabolic complications or HL [12-15]. The change in circulating adiponectin levels is greater among participants with high VFA than in high BMI or subcutaneous fat area [16]. Circulating adiponectin levels in both sexes are increased by reduced VFA [17,18]. Based on a study by Hwang et al., there was an association between plasma adiponectin levels and hearing thresholds [13].
According to some studies, there was no association between BMI and hearing impairment, and that may be due to the inability to discriminate between VFA and subcutaneous fat area or muscle mass [5,6]. Kim et al. investigated the association between VFA and hearing thresholds in 662 adults using computed tomography [8]. They observed a significant association between the 2 variables in only 206 women. In addition, they analyzed the data using only linear regression analyses and divided the participants into only 2 threshold groups (low and high frequencies). To the best of our knowledge, there has been only one previous study to evaluate the association between VFA and hearing thresholds. The main technical advancement achieved in the current study was the method for VFA measurement. Although computed tomography and magnetic resonance imaging are the gold standard for assessing VFA, these are not proper methods in routine health-checkup settings. VFA measurement using BIA is simple, relatively accurate, and requires short imaging time. In present study, VFA measurements were normalized by BMI. The normalized VFA/BMI values had the highest AUROCs compared to the non-normalized VFA or BMI values. HL was defined as AHT >40 dB according to the serviceable hearing level. An association of VFA/BMI with all 4 variables and HL as a categorical variable was observed in both sexes.
There were a few limitations in this study. Firstly, the study was limited by its retrospective nature. We were unable to evaluate ethnic differences, and establishing possible causality. Secondly, we did not evaluate the sensitive components of hearing impairments (i.e., speech discrimination or auditory brainstem response test). Thirdly, habitual parameters (i.e., alcohol consumption and smoking), and history of exposure to noise (i.e., occupational or explosive noise) were not evaluated in the present study. However, the impact of these limitations was reduced by the large sample size in this study. Further prospective analysis, including follow-up data and sensitive methods, is needed to evaluate a possible strong correlation between VFA/BMI and hearing impairment.
In conclusion, VFA/BMI was associated with hearing impairment in the Asian population. Therefore, the participants with high VFA/BMI should be closely monitored for hearing impairment.
Acknowledgements
This work was supported by the Medical Research Center Program (2015R1A5A2009124) through the National Research Foundation of Korea (NRF) funded by the Ministry of Science, ICT and Future Planning. The funder had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
Competing Interests
The authors have declared that no competing interest exists.
References
1. Gates GA, Cobb JL, D'Agostino RB, Wolf PA. The relation of hearing in the elderly to the presence of cardiovascular disease and cardiovascular risk factors. Arch Otolaryngol Head Neck Surg. 1993;119:156-61
2. Barrenäs ML, Jonsson B, Tuvemo T, Hellström PA, Lundgren M. High risk of sensorineural hearing loss in men born small for gestational age with and without obesity or height catch-up growth: a prospective longitudinal register study on birth size in 245,000 Swedish conscripts. J Clin Endocrinol Metab. 2005;90:4452-6
3. Fransen E, Topsakal V, Hendrickx JJ, Van Laer L, Huyghe JR, Van Eyken E. et al. Occupational noise, smoking, and a high body mass index are risk factors for age-related hearing impairment and moderate alcohol consumption is protective: a European population-based multicenter study. J Assoc Res Otolaryngol. 2008;9:264-76
4. Gupta PP, Fonarow GC, Horwich TB. Obesity and the obesity paradox in heart failure. Can J Cardiol. 2015;31:195-202
5. Hwang JH, Wu CC, Hsu CJ, Liu TC, Yang WS. Association of central obesity with the severity and audiometric configurations of age-related hearing impairment. Obesity. 2009;17:1796-801
6. Shargorodsky J, Curhan SG, Eavey R, Curhan GC. A prospective study of cardiovascular risk factors and incident hearing loss in men. Laryngoscope. 2010;120:1887-91
7. Fox CS, Massaro JM, Hoffmann U, Pou KM, Maurovich-Horvat P, Liu CY. et al. Abdominal visceral and subcutaneous adipose tissue compartments: association with metabolic risk factors in the Framingham Heart Study. Circulation. 2007;116:39-48
8. Kim TS, Park SW, Kim DY, Kim EB, Chung JW, So HS. Visceral adipose tissue is significantly associated with hearing threholds in adult women. Clin Endocrinol (Oxf). 2014;80:368-75
9. Ryo M, Kishida K, Nakamura T, Yoshizumi T, Funahashi T, Shimomura I. Clincal significance of visceral adiposity assessed by computed tomography: A Japanese perspective. World J Radiol. 2014;6:409-16
10. Kang SH, Cho KH, Park JW, Yoon KW, Do JY. Association of Visceral Fat Area with Chronic Kidney Disease and Metabolic Syndrome Risk in the General Population: Analysis Using Multi-Frequency Bioimpedance. Kidney Blood Press Res. 2015;40:223-30
11. Levey AS, Stevens LA, Schmid CH, Zhang YL, Castro AF 3rd, Feldman HI. et al. A new equation to estimate glomerular filtration rate. Ann Intern Med. 2009;150:604-12
12. Kishida K, Funahashi T, Shimomura I. Adiponectin as a routine clinical biomarker. Best Pract Res Clin Endocrinol Metab. 2014;28:119-30
13. Hwang JH, Hsu CJ, Liu TC, Yang WC. Association of plasma adiponectin levels with hearing thresholds in adults. Clin Endocrinol. 2011;75:614-20
14. [14] Tanigawa T, Shibata R, Ouchi N, Kondo KL, Ishii M, Katahira N. et al. Adiponectin deficiency exacerbates age-related hearing impairment. Cell Death Dis. 2014;5:e1189
15. Wu CC, Tsai CH, Lu YC, Lin HC, Hwang JH, Lin YH. et al. Contribution of adiponectin and its type 1 receptor to age-related hearing impairment. Neurobiol Aging. 2015;36:2085-93
16. Kishida K, Kim KK, Funahashi T, Matsuzawa T, Kang HC, Shimomura I. Relationships between circulating adiponectin levels and fat distribution in obese subjects. J Atheroscler Thromb. 2011;18:592-5
17. Hotta K, Funahashi T, Arita Y, Takahashi M, Matsuda M, Okamoto Y. et al. Plasma concentrations of a novel, adipose-specific protein, adiponectin, in type 2 diabetic patients. Arterioscler Thromb Vasc Biol. 2000;20:1595-9
18. Ng TW, Watts GF, Barrett PH, Rye KA, Chan DC. Effect of weight loss on LDL and HDL kinetics in the metabolic syndrome: associations with changes in plasma retinol-binding protein-4 and adiponectin levels. Diabetes Care. 2007;30:2945-50
Author contact
Corresponding author: Jun-Young Do, MD, Department of Internal Medicine, Yeungnam University Hospital, 317-1 Daemyung-Dong, Nam-Ku, Daegu 705-717, Korea. Fax: +82-53-654-8386, Phone: +82-53-680-3844, E-mail: kangkang00001net

 Global reach, higher impact
Global reach, higher impact