Impact Factor ISSN: 1449-1907
Int J Med Sci 2008; 5(5):285-291. doi:10.7150/ijms.5.285 This issue Cite
Research Paper
EGFR Expression in Gallbladder Carcinoma in North America
1. Long Island Jewish Medical Center, New Hyde Park, NY 11040, USA
2. Hartford Hospital, Hartford, CT, USA
Received 2008-2-18; Accepted 2008-9-19; Published 2008-9-22
Abstract
BACKGROUND: Increased epidermal growth factor receptor (EGF receptor) expression has been noted in various cancers and has become a useful target for therapeutic interventions. Small studies from Asia and Australia have demonstrated EGFR over-expression in gallbladder cancer. We sought to evaluate the expression of EGFR in a series of 16 gallbladder cancer patients from North America.
METHODS: Using tumor registry data, we identified 16 patients diagnosed with gall bladder carcinoma at our medical center between the years of 1998 and 2005. We performed a retrospective review of these patients' charts, obtained cell blocks from pathology archives and stained for EGFR and Her2/neu.
RESULTS: Fifteen of sixteen patients were noted to over-express EGFR. Three were determined 1+, nine were 2+ and three were 3+. Eight patients had poorly differentiated adenocarcinoma, six had moderately differentiated and two had well-differentiated tumors. In this small series, there was a trend toward shorter survival and more poorly differentiated tumors in patients with greater intensity of EGFR expression. One patient was EGFR negative but 3+ for erb-2/Her 2-neu expression. No patient co-expressed EGFR and Her-2-neu. Median survival of patients in this series was 17 months.
CONCLUSION: In view of our observations confirming the over-expression of EGFR in our patient population in North America, and the recent success of EGFR targeted therapies in other solid tumors that over-express EGFR, it may now be appropriate to evaluate agents targeting this pathway either as single agents or in combination with standard chemotherapy.
Keywords: gallbladder cancer, endothelial growth factor receptor (EGFR), differentiation, survival, her-2-neu
Introduction
Approximately 5000 cases of gallbladder cancer are diagnosed in the United States per year. Higher rates are seen in Latin American countries such as Mexico, Chile and Bolivia, roughly correlating with the higher incidence of cholelithiasis. Various chemotherapy agents, including 5-FU and Gemcitabine, have been evaluated for the management advanced disease but thus far results have been disappointing [1-4].
5 FU plus LV has been the backbone of randomized clinical trials done in the past, demonstrating a RR of 32% and OS of 6months.[5] Combination therapy with 5FU and cisplatin have shown RRs of 10%–40% and median OS better than those observed with 5-FU alone.[5-12] Single agent gemcitabine has been extensively evaluated in patients with metastatic biliary tract tumors with RRs in the range of 0%–30%, with median OS times in the range of 5–14 months. [13-18]Gemcitabine combinations with cisplatin, oxaliplatin or capecitabine have been tested in several clinical trials, which have demonstrated RRs 21%–53% and median OS times 5–15 months; these results are somewhat better than those from single-agent gemcitabine studies.[19-23] A pooled analysis of 112 trial using gemcitabine-based combination regimens confirmed superiority to single agent therapy. However the outcomes are still dismal with the pressing need for development of newer therapies.[1, 24, 25]
Increased epidermal growth factor receptor (EGF receptor) expression has been noted in various cancers such as colon, squamous cell of the head and neck, non-small cell lung and breast cancers. Several small studies from Asia, Europe and Australia have examined the expression of EGFR in gallbladder cancer.[26-30] The epidermal growth factor receptor is one of many transmembrane protein kinases that are involved in signal transduction affecting cellular activities such as metabolism, transcription, cell-cycle progression, apoptosis and differentiation.[31] These processes are tightly controlled, but when protein kinase activity is deregulated, malignant transformation may occur. [32] Among the various mechanisms of increased EGFR activation, is receptor over-expression, gene amplification and the loss of inhibitory signals. Activation of EGFR results in phosphorylation of intracellular substrates downstream and the subsequent activation of mitotic pathways. [32]
The improved understanding of EGFR's role in oncogenesis has made it an attractive target for therapeutic intervention in several cancers. Clinical and preclinical data exist utilizing this target in colon cancer, squamous cell carcinoma of the head and neck, non-small cell lung cancer and breast cancer. [33-41] Likewise, the over-expression of EGFR on gallbladder carcinoma may have direct clinical implications with an alternative management strategy for the management of this difficult disease. [42, 43] In our study, we have gathered the data showing over-expression of EGFR in gallbladder cancer cases in North America.
Materials and Methods
Data Retrieval
Institutional Review Board approval was obtained. Tumor registry data identified patients diagnosed with gall bladder carcinoma at a single institution between the years of 1998 and 2005. We performed a retrospective review of these consecutive patients' charts and obtained the following information: biopsy site, stage at diagnosis, treatment modalities, survival, and tumor grade. Cell blocks were then obtained from pathology archives and stained for EGFR and Her2/neu as described below.
Methods for EGFR and Her 2/neu staining
Serial 4µm sections were cut from the cell block. Slides were then placed in xylene for 15 minutes for deparaffinization. Dehydration was performed by steps of graded alcohol. Tap water was used for rehydration. Slides stained with Her-2/neu (prediluted, monoclonal, clone CB11, Carpinteria, CA) were then pretreated for antigen retrieval by microwaving for 30 minutes using citrate buffer, pH 6. They were then stained using the Ventana Nexus autostainer. Slides stained with EGFR (prediluted, monoclonal, clone 2-18C9, Carpinteria, CA) were not pretreated for antigen retrieval and were stained using the Ventana autostainer.
Two observers who were blinded to the histologic diagnosis interpreted the slides. Cell membrane staining was used to assess positivity for EGFR and Her 2/neu. In each case, the intensity of the staining (0- negative to 3- strong) was determined. (Figure 1a-c).
EGFR Staining.
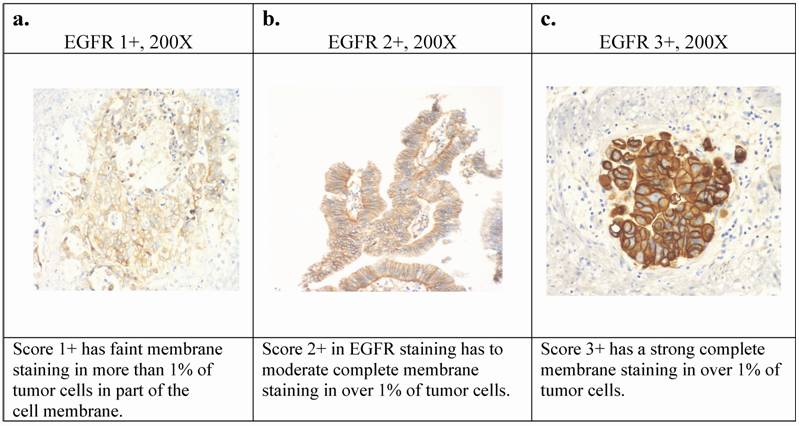
The staining pattern for Her2-neu was determined as follows: Score 0= no staining is observed; Score 1+= faint membrane staining in more than 10% of tumor cells in part of the cell membrane; score 2+= weak to moderate complete membrane staining in over 10% of tumor cells; score 3+= strong complete membrane staining in over 10% of tumor cells.
The staining pattern for EGFR was determined as follows: Score 0= no staining is observed; Score 1+= faint membrane staining in more than 1% of tumor cells in part of the cell membrane; score 2+= weak to moderate complete membrane staining in over 1% of tumor cells; Score 3+=strong complete membrane staining in over 1% of tumor cells.
Results
In our series of sixteen patients, fifteen were noted to over-express EGFR (Table 1). Three were determined 1+, nine were 2+ and three were 3+. Eight patients had poorly differentiated adenocarcinoma, six had moderately differentiated and two had well-differentiated tumors. One patient was EGFR negative but 3+ for erb-2/Her 2-neu expression. Nine of 16 patients underwent surgical intervention alone, three underwent chemotherapy alone, two underwent both surgery and chemotherapy and two underwent surgery, chemotherapy and radiation therapy. Staging distribution was as follows: stage I: 12.5%(n=2); stage II: 37.5%(n=6); stage III: 12.5% (n=2); stage IV: 37.5%(n=6).
Results
| Patient | Age | Sex | Stage | Biopsy Site | Rx modality | Survival | differentiation/Grad | Erb-B-2/Her 2-neu | EGFR |
|---|---|---|---|---|---|---|---|---|---|
| 1 | 83 | M | II | gallbladder | S | 40 months | poor diff adenocarcinom | 3+ | negative |
| 2 | 76 | F | II | gallbladder | S | 12 months | mod diff adenocarcinom | negative | 1+ |
| 3 | 61 | F | IV | gallbladder | S | 17 months | well-diff adenocarcinom | negative | 1+ |
| 4 | 62 | F | IV | peritoneum | C | 9 months | mod diff adenocarcinom | negative | 1+ |
| 5 | 54 | F | IV | liver | C | 10.5 months | mod diff adenocarcinom | negative | 2+ |
| 6 | 77 | F | I | gallbladder | S | 28 months (alive) | poor diff adenocarcinom | negative | 2+ |
| 7 | 65 | F | II | gallbladder | S | 11 months | mod diff adenocarcinom | negative | 2+ |
| 8 | 70 | F | II | gallbladder | S,C,R | 25 months (alive) | well-diff mucinous adenocarc | negative | 2+ |
| 9 | 55 | F | IV | omentum | C | 4 months | poor diff adenocarcinom | negative | 2+ |
| 10 | 63 | M | II | gallbladder | S | 33 months(alive) | mod diff adenocarcinom | negative | 2+ |
| 11 | 71 | F | III | gallbladder | S | 50 months (alive) | poor diff adenocarcinom | negative | 2+ |
| 12 | 68 | F | II | gallbladder | S,R,C | 19 months | poor diff adenocarcinom | negative | 2+ |
| 13 | 75 | F | IV | peritoneum | S,C | 27 months | mod diff adenocarcinom | negative | 2+ |
| 14 | 80 | F | I | gallbladder | S | 17 months | poor diff adenocarcinom | negative | 3+ |
| 15 | 74 | F | IV | gallbladder | S,C | 3.5 months | poor diff adenocarcinom | negative | 3+ |
| 16 | 46 | F | III | gallbladder | S | 2.5 months | poor diff adenocarcinom | negative | 3+ |
S=surgery, C=chemotherapy, R=radiation
We evaluated a possible correlation between the level of differentiation and intensity of EGFR expression. The three patients with 1+ expression had well-differentiated (one patient) and moderately differentiated (two patients) adenocarcinoma. Conversely, all three of the 3+ EGFR patients had tumors of the poorly differentiated type. The nine patients with 2+ EGFR was a mix of the former groups (one well-differentiated, four moderately differentiated and four poorly differentiated). This suggests an inverse relationship between differentiation and EGFR expression. Median survival of the 3+ patients was 3.5 months compared to 17 months overall.
Although our sample size is small, our data above also suggests an inverse relationship between EGFR expression intensity and survival. The patient with stage I disease with 3+ EGFR staining had a survival of 17 months versus the other stage I patient in our sample, who had 2+ EGFR staining, and is alive at 28 months follow-up. The patient with stage IV disease expressing 3+EGFR, had a survival of 3.5 months compared to the median survival of 10.5 months for stage IV patients with 1+ and 2+ staining. In summary, the 3+ patients had a substantially shorter survival when compared with less intense EGFR expression patients of similar stage.
Discussion
Background of EGFR
Epidermal growth factor receptor is a protein kinase receptor involved in the signal transduction affecting cellular activities such as metabolism, transcription, cell-cycle progression, apoptosis and differentiation. The two major subsets of drugs that inhibit EGF receptors are monoclonal antibodies and small molecules. The monoclonal antibodies prevent ligand binding and activation of the EGFR. One agent of this type is cetuximab, which has shown clinical efficacy in colon, [31, 32] and head and neck cancers. [31] Small molecules that target EGFR compete with ATP binding to the tyrosine kinase domain, thereby blocking signaling pathways.[32] Examples of drugs of this type are gefitinib and erlotinib. Erlotinib has shown activity against non-small cell lung and pancreatic cancers.
EGFR Expression in our sample of Gallbladder cancer patients
As with the available published data from Asia and Australia, we found a predominance of EGFR over-expression in our gallbladder cancer specimens. In our sample of 16 patients, only one patient (6.3%) did not have over-expression of EGFR. Nine patients(56.3%) were 2+ and three(18.3%) were 3+ in immunohistochemical staining. All fifteen of the patients expressing EGFR were negative for Erb-B-2/Her 2-neu. Conversely, the single patient that expressed Erb-B-2/Her 2-neu was 3+ intensity, and was negative for EGFR. We found it interesting that these two receptors, both of the erb-B family, have no co-expression in any of our patients.
As shown in the results, a disproportionate number of patients with 3+ EGFR expression had poorly differentiated tumors. Conversely, the patients with 1+ EGFR expression seemed to have proportionally higher numbers of patients with moderate or well-differentiated tumors. This suggests an inverse relationship between differentiation and EGFR expression. Assuming that poorly differentiated tumors behave more aggressively, intensity of EGFR expression may correlate with aggressiveness of disease.
This hypothesis is further supported by the examining the EGFR expression relating to survival. Stage for stage, the patients with greater EGFR intensity had shorter survival therefore suggesting an inverse relationship between EGFR expression intensity and survival. Although the number of patients is few, this is a consistent pattern throughout our sample. The patient with stage I disease with 3+ EGFR staining had a survival of 17 months versus the other stage I patient in our sample, who had 2+ EGFR staining, and is alive at 28 months follow-up. The patient with stage IV disease expressing 3+EGFR, had a survival of 3.5 months compared to the median survival of 10.5 months for stage IV patients with 1+ and 2+ staining. In summary, the 3+ patients had a substantially shorter survival when compared with less intense EGFR expression patients of similar stage.
Previous studies of EGFR Expression in Biliary Tumors
A study from MD Anderson demonstrated that constitutive expression of ErbB-2 in mice resulted in development of gallbladder cancer. [44]Several small studies, mostly from Asia, have complemented this work by examining the level of expression of EGFR in biliary tumors (Table 2). These few studies demonstrated a significant and consistent over-expression of epithelial growth factor receptor in biliary tumors. The largest such study was published by Zhou et al from China.[26] Zhou compared EGFR expression in normal gallbladder specimens (10 specimen) with gallbladder carcinoma specimen (41 specimens) and hyperplastic tissue specimens (26) using immunohistochemistry. EGFR over-expression was found to be 71% in the carcinoma specimens as compared to 0% of the normal gallbladder specimens. Lee et al performed immunohistochemistry stains for EGFR on 13 gallbladder cancer specimens from Australia.[29] 100% of the gallbladder cancer specimens were found to stain strongly positive for EGFR.
EGFR expression in Biliary Tumors.
| Study | N | Immunoreactivity(%) |
|---|---|---|
| Lee et al.[29] | Gallbladder-13 Biliary duct-7 | 100% 86% |
| Kim et al.[52] | Biliary duct-20 | 25% |
| Zhou et al.[26] | Gallbladder-41 | 71% |
Current studies in EGFR related therapy of Gallbladder Cancer
Several trials have been undertaken in the past investigating chemotherapy for advanced biliary cancers, including cancer of the gallbladder. Many of these trials involved gemcitabine, either as a single agent (table 3) or in combination with other chemotherapies (table 4).[1, 2] Other trials have looked at non gemcitabine based combination therapies (table 5). The response rates have been between 21%–53% and median OS times 5–15 months.[1] With limited improvement in responses and survival with the combination chemotherapies, the focus is now on evolution of newer targeted therapies.
Single agent Gemcitabine.
| Study | N | Response Rate (%) | Stable Disease | Time to Progression (months) | Median Overall Survival |
|---|---|---|---|---|---|
| Eng et al.[53] | 14 | 0% | 13% | 9 months | 5 months |
| Mehrotra et al[54] | 12 | 0% | 75% | 3 months | 6 months |
| Funakoshi et al.[55] | 40 | 17.5% | 2.6 months | 7.6 months | |
| Tsavaris et al [56] | 30 | 30% | 7 months | 17 months (Gallbladder) 11 months (biliary duct) | |
| Gallardo et al.[57] | 26 | 36% | 36.7% | ||
| Park et al.[58] | 23 | 26% | 39% | 8.1 months | 13.1 months |
| Kubicka et al.[17] | 23 | 30% |
Gemcitabine Combinations.
| Study | Treatment | N | Response Rate (%) | Median Time to Progression | Median Overall Survival |
|---|---|---|---|---|---|
| Doval et al [59] | Gemcitabine + cisplatin | 39 | 37% | 4.5 months | 5 months |
| Park et al.[58] | Gemcitabine + cisplatin | 35 | 17% | 3.5 months | 8.3 months |
| Malik et al.[60] | Gemcitabine + cisplatin | 11 | 64% | 6.5 months | 10 months |
| Reyes-Vidal et al [61] | Gemcitabine + cisplatin | 44 | 48% | 7 months | |
| Tan et al.[22] | Gemcitabine + carboplatin | 13 | 31% | ||
| Knox et al.[3] | Gemcitabine + Capecitabine | 45 | 31% | 7 months | 14 months |
| Chang et al [23] | Gemcitabine + Capecitabine | 34 | 12% | 2.6 months | 7.8 months |
| Verderame et al [24] | Gemcitabine + Oxaliplatin | 24 | 50% | 12 months | |
| Wagner et al [25] | Gemcitabine + Oxaliplatin + CI 5-FU | 35 | 9.9 months | ||
| NCCTG [26] | Gemcitabine + CI 5-FU/LV | 42 | 9.5% | 4.6 months | 9.7 months |
| Knox [27] | Gemcitabine + CI 5-FU/LV | 27 | 33% | 3.7 months | 5.3 months |
| Knox et al.[3] | Gemcitabine + Capecitabine | 45 | 31% | 7 months | 14 months |
Non-Gemcitabine Regimens.
| Study | Treatment | N | Response Rate (%) |
|---|---|---|---|
| Romano et al[62] | Cisplatin + irinotecan | 16 | 37% |
| Nehls et al.[63] | Capecitabine + Oxaliplatin | 27 | 27% |
| Glover et al.[64] | Capecitabine + Oxaliplatin | 21 | 19% |
| Sanz-Altamira[65] | Carboplatin + 5-FU/LV | 14 | 21% |
Several studies targeting the EGFR pathway have been undertaken. In a phase II study of 42 patients with biliary tract cancer treated with single-agent erlotinib, Philip et al. demonstrated a 17% 6-month progression-free survival (PFS) rate; three patients had partial responses (PRs) as determined by the Response Evaluation Criteria in Solid Tumors. Of these patients, 57% had received first line chemotherapy. [45] In this study, EGFR mutation status was not tested, and therefore it is unknown if the response correlated with EGFR mutation status. There is a possibility that the population of patients with the EGFR mutation might have a significant benefit from EGFR inhibition therapy, along the lines of non-small cell lung cancer patients. [2, 46]
Efficacy of cetuximab, in biliary tract and gallbladder cancers, in combination with either Gemcitabine or gemcitabine and oxaliplatin have been demonstrated in two studies [42, 43]. Lapatinib, a dual EGFR-1and humanepidermal growth factor receptor (HER)-2/Neu inhibitor, was tested in a phase I trial in seven patients with biliary tract cancer. [47]
In a recently completed phase II study, patients with locally advanced/metastatic cholangiocarcinoma or gallbladder cancer were given cetuximab 500 mg/m² on day 1 followed by 1,000mg/m² gemcitabine (day 1) and 100mg/m² oxaliplatin on day 2 every second week. The primary endpoint was response rate; secondary endpoints were toxicity, progression free and overall survival. The overall response rate of 19 evaluable patients was 58%, including one patient with a complete response. Six patients (32%) achieved stable disease and 2 patients (11%) progressed under chemotherapy after a median of 6.5 cycles (SD ± 2.8). The response significantly correlated with the grade of acne-like rash (p < 0.002). Six initially unresectable patients underwent a curative resection after major response was observed (32%). The median PFS was 9.0 months (95% CI 3.1-14.9). Four patients are currently without evidence of disease after a median follow-up of 6.3 months post-liver resection[42]. Bevacizumab and sorafenib are also under investigation for treatment of both these cancers.[48] [49]
The finding of over-expression of EGFR in our patient population in North America further strengthens the rationale in targeting this pathway in gallbladder cancer [35, 38, 40, 41]. Additional clinical trials are underway exploring the role of EGFR inhibition in this malignancy. Decreased response to EGFR inhibitors has been reported in Kras mutant patients in colorectal cancer[39-41]. In this context, Kras mutation status in patients with biliary tract and gallbladder warrants further investigation as use of EGFR inhibitors grows. [50, 51]
Conflict of Interest
The authors have declared that no conflict of interest exists.
References
1. Eckel F, Schmid RM. Chemotherapy in advanced biliary tract carcinoma: a pooled analysis of clinical trials. Br J Cancer. 2007;96(6):896-902
2. Hezel A.F, Zhu AX. Systemic therapy for biliary tract cancers. Oncologist. 2008;13(4):415-23
3. Iyer R.V. et al. A phase II study of gemcitabine and capecitabine in advanced cholangiocarcinoma and carcinoma of the gallbladder: a single-institution prospective study. Ann Surg Oncol. 2007;14(11):3202-9
4. Riechelmann R.P. et al. Expanded phase II trial of gemcitabine and capecitabine for advanced biliary cancer. Cancer. 2007;110(6):1307-12
5. Choi C.W. et al. Effects of 5-fluorouracil and leucovorin in the treatment of pancreatic-biliary tract adenocarcinomas. Am J Clin Oncol. 2000;23(4):425-8
6. Ducreux M. et al. Effective treatment of advanced biliary tract carcinoma using 5-fluorouracil continuous infusion with cisplatin. Ann Oncol. 1998;9(6):653-6
7. Ellis P.A. et al. Epirubicin, cisplatin and infusional 5-fluorouracil (5-FU) (ECF) in hepatobiliary tumours. Eur J Cancer. 1995;31A(10):1594-8
8. Lee M.A. et al. Epirubicin, cisplatin, and protracted infusion of 5-FU (ECF) in advanced intrahepatic cholangiocarcinoma. J Cancer Res Clin Oncol. 2004;130(6):346-50
9. Mitry E. et al. Combination of folinic acid, 5-fluorouracil bolus and infusion, and cisplatin (LV5FU2-P regimen) in patients with advanced gastric or gastroesophageal junction carcinoma. Ann Oncol. 2004;15(5):765-9
10. Glimelius B. et al. Chemotherapy improves survival and quality of life in advanced pancreatic and biliary cancer. Ann Oncol. 1996;7(6):593-600
11. Takada T. et al. Comparison of 5-fluorouracil, doxorubicin and mitomycin C with 5-fluorouracil alone in the treatment of pancreatic-biliary carcinomas. Oncology. 1994;51(5):396-400
12. Feisthammel J. et al. Irinotecan with 5-FU/FA in advanced biliary tract adenocarcinomas: a multicenter phase II trial. Am J Clin Oncol. 2007;30(3):319-24
13. Burris H.A3rd. et al. Improvements in survival and clinical benefit with gemcitabine as first-line therapy for patients with advanced pancreas cancer: a randomized trial. J Clin Oncol. 1997;15(6):2403-13
14. Oettle H. et al. Adjuvant chemotherapy with gemcitabine vs observation in patients undergoing curative-intent resection of pancreatic cancer: a randomized controlled trial. Jama. 2007;297(3):267-77
15. Raderer M. et al. Two consecutive phase II studies of 5-fluorouracil/leucovorin/mitomycin C and of gemcitabine in patients with advanced biliary cancer. Oncology. 1999;56(3):177-80
16. Gebbia V. et al. Treatment of inoperable and/or metastatic biliary tree carcinomas with single-agent gemcitabine or in combination with levofolinic acid and infusional fluorouracil: results of a multicenter phase II study. J Clin Oncol. 2001;19(20):4089-91
17. Kubicka S. et al. Phase II study of systemic gemcitabine chemotherapy for advanced unresectable hepatobiliary carcinomas. Hepatogastroenterology. 2001;48(39):783-9
18. Penz M. et al. Phase II trial of two-weekly gemcitabine in patients with advanced biliary tract cancer. Ann Oncol. 2001;12(2):183-6
19. Meyerhardt J.A. et al. Phase-II study of gemcitabine and cisplatin in patients with metastatic biliary and gallbladder cancer. Dig Dis Sci. 2008;53(2):564-70
20. Lee G.W. et al. Combination chemotherapy with gemcitabine and cisplatin as first-line treatment for immunohistochemically proven cholangiocarcinoma. Am J Clin Oncol. 2006;29(2):127-31
21. Kim S.T. et al. A Phase II study of gemcitabine and cisplatin in advanced biliary tract cancer. Cancer. 2006;106(6):1339-46
22. Andre T. et al. Gemcitabine combined with oxaliplatin (GEMOX) in advanced biliary tract adenocarcinoma: a GERCOR study. Ann Oncol. 2004;15(9):1339-43
23. Harder J. et al. Outpatient chemotherapy with gemcitabine and oxaliplatin in patients with biliary tract cancer. Br J Cancer. 2006;95(7):848-52
24. Wang S.J. et al. Prediction model for estimating the survival benefit of adjuvant radiotherapy for gallbladder cancer. J Clin Oncol. 2008;26(13):2112-7
25. Smith G. et al. A 10-year experience in the management of gallbladder cancer. HPB (Oxford). 2003;5(3):159-66
26. Zhou Y.M. et al. [Significance of expression of epidermal growth factor (EGF) and its receptor (EGFR) in chronic cholecystitis and gallbladder carcinoma]. Ai Zheng. 2003;22(3):262-5
27. Ariyama H. et al. Gefitinib, a selective EGFR tyrosine kinase inhibitor, induces apoptosis through activation of Bax in human gallbladder adenocarcinoma cells. J Cell Biochem. 2006;97(4):724-34
28. Nakazawa K. et al. Amplification and overexpression of c-erbB-2, epidermal growth factor receptor, and c-met in biliary tract cancers. J Pathol. 2005;206(3):356-65
29. Lee C.S. and A. Pirdas, Epidermal growth factor receptor immunoreactivity in gallbladder and extrahepatic biliary tract tumours. Pathol Res Pract. 1995;191(11):1087-91
30. Leone F. et al. Somatic mutations of epidermal growth factor receptor in bile duct and gallbladder carcinoma. Clin Cancer Res. 2006;12(6):1680-5
31. Manning G. et al. The protein kinase complement of the human genome. Science. 2002;298(5600):1912-34
32. Baselga J and ArteagaCL. Critical update and emerging trends in epidermal growth factor receptor targeting in cancer. J Clin Oncol. 2005;23(11):2445-59
33. Macarulla T. et al. Novel targets for anticancer treatment development in colorectal cancer. Clin Colorectal Cancer. 2006;6(4):265-72
34. Cunningham D. et al. Cetuximab monotherapy and cetuximab plus irinotecan in irinotecan-refractory metastatic colorectal cancer. N Engl J Med. 2004;351(4):337-45
35. Posner M.R. and L.J. Wirth, Cetuximab and radiotherapy for head and neck cancer. N Engl J Med. 2006;354(6):634-6
36. Nanda R. Targeting the human epidermal growth factor receptor 2 (HER2) in the treatment of breast cancer: recent advances and future directions. Rev Recent Clin Trials. 2007;2(2):111-6
37. Metro G. et al. Epidermal growth factor receptor (EGFR) targeted therapies in non-small cell lung cancer (NSCLC). Rev Recent Clin Trials. 2006;1(1):1-13
38. Pirker R. FLEX: A randomized, multicenter, phase III study of cetuximab in combination with cisplatin/vinorelbine (CV) versus CV alone in the first-line treatment of patients with advanced non-small cell lung cancer (NSCLC). J Clin Oncol. 2008;26:3
39. Cervantes A. Correlation of KRAS status (wild type [wt] vs mutant [mt]) with efficacy to first-line cetuximab in a study of cetuximab single agent followed by cetuximab + FOLFIRI in patients (pts) with metastatic colorectal cancer (mCRC). Abstract - No 4129 - 2008 ASCO Annual Meeting. 2008
40. Cutsem EV. KRAS status and efficacy in the first-line treatment of patients with metastatic colorectal cancer (mCRC) treated with FOLFIRI with or without cetuximab: The CRYSTAL experience. Abstract No 2 - 2008 ASCO Annual Meeting. 2008
41. Bokemeyer C. KRAS status and efficacy of first-line treatment of patients with metastatic colorectal cancer (mCRC) with FOLFOX with or without cetuximab: The OPUS experience. Abstract No 4000 - 2008 ASCO Annual Meeting. 2008
42. Paule B. et al. Cetuximab plus gemcitabine-oxaliplatin (GEMOX) in patients with refractory advanced intrahepatic cholangiocarcinomas. Oncology. 2007;72(1-2):105-10
43. Sprinzl M.F. et al. Gemcitabine in combination with EGF-Receptor antibody (Cetuximab) as a treatment of cholangiocarcinoma: a case report. BMC Cancer. 2006;6:190
44. Kiguchi K. et al. Constitutive expression of ErbB-2 in gallbladder epithelium results in development of adenocarcinoma. Cancer Res. 2001;61(19):6971-6
45. Philip P.A. et al. Phase II study of erlotinib in patients with advanced biliary cancer. J Clin Oncol. 2006;24(19):3069-74
46. Lynch T.J. et al. Activating mutations in the epidermal growth factor receptor underlying responsiveness of non-small-cell lung cancer to gefitinib. N Engl J Med. 2004;350(21):2129-39
47. Safran H. et al. Lapatinib/gemcitabine and lapatinib/gemcitabine/oxaliplatin: a phase I study for advanced pancreaticobiliary cancer. Am J Clin Oncol. 2008;31(2):140-4
48. Hida Y. et al. Vascular endothelial growth factor expression is an independent negative predictor in extrahepatic biliary tract carcinomas. Anticancer Res. 1999;19(3B):2257-60
49. El Khoueiry AB. et al. A phase II study of sorafenib (BAY 43-9006) as single agent in patients (pts) with unresectable or metastatic gallbladder cancer or cholangiocarcinomas. J Clin Oncol. 2007;25(suppl 18S):4639
50. Nakanuma Y. et al. Anatomic and molecular pathology of intrahepatic cholangiocarcinoma. J Hepatobiliary Pancreat Surg. 2003;10(4):265-81
51. Lapkus O. et al. Determination of sequential mutation accumulation in pancreas and bile duct brushing cytology. Mod Pathol. 2006;19(7):907-13
52. Kim H.J. et al. [Expression of epidermal growth factor receptor, ErbB2 and matrix metalloproteinase-9 in hepatolithiasis and cholangiocarcinoma]. Korean J Gastroenterol. 2005;45(1):52-9
53. Eng C. et al. A Phase II trial of fixed dose rate gemcitabine in patients with advanced biliary tree carcinoma. Am J Clin Oncol. 2004;27(6):565-9
54. Gupta N. et al. Gemcitabine-induced pulmonary toxicity: case report and review of the literature. Am J Clin Oncol. 2002;25(1):96-100
55. Okusaka T. et al. Phase II study of single-agent gemcitabine in patients with advanced biliary tract cancer. Cancer Chemother Pharmacol. 2006;57(5):647-53
56. Tsavaris N. et al. Weekly gemcitabine for the treatment of biliary tract and gallbladder cancer. Invest New Drugs. 2004;22(2):193-8
57. Gallardo J.O. et al. A phase II study of gemcitabine in gallbladder carcinoma. Ann Oncol. 2001;12(10):1403-6
58. Park J.S. et al. Single-agent gemcitabine in the treatment of advanced biliary tract cancers: a phase II study. Jpn J Clin Oncol. 2005;35(2):68-73
59. Doval D.C. et al. A phase II study of gemcitabine and cisplatin in chemotherapy-naive, unresectable gall bladder cancer. Br J Cancer. 2004;90(8):1516-20
60. Malik I.A. et al. Gemcitabine and Cisplatin is a highly effective combination chemotherapy in patients with advanced cancer of the gallbladder. Am J Clin Oncol. 2003;26(2):174-7
61. Reyes-Vidal J. et al. Gemcitabine and cisplatin in the treatment of patients with unresectable or metastatic gallbladder cancer: Results of the phase II Gocchi study. ASCO GI (abstr 87). 2000
62. Romano R. et al. A combination of cisplatin and irinotecan against advanced biliary tree carcinomas. Journal of Clinical Oncology. 2004;22:4145
63. Nehls O. et al. A prospective multicenter phase II trial of capecitabine plus oxaliplatin (CapOx) in advanced biliary system adenocarcinomas: The final results. Journal of Clinical Oncology. 2006;24:4136
64. Glover KY. et al. A Phase II Study of Oxaliplatin and Capecitabine (XELOX) in Patients with Unresectable Cholangiocarcinoma, including Carcinoma of the Gallbladder and Biliary Tract. Journal of Clinical Oncology. 2005;23:4123
65. Sanz-Altamira P.M. et al. A phase II trial of 5-fluorouracil, leucovorin, and carboplatin in patients with unresectable biliary tree carcinoma. Cancer. 1998;82(12):2321-5
Author contact
Correspondence to: Long Island Jewish Medical Center, Division of Hematology/Oncology, 270-05 76th Avenue, New Hyde Park, NY 11040. Telephone 718-470-8934; Facsimile 718-470-0169; Email: mkaufmanedu

 Global reach, higher impact
Global reach, higher impact